Published online Jan 7, 2018. doi: 10.3748/wjg.v24.i1.5
Peer-review started: October 3, 2017
First decision: October 18, 2017
Revised: November 7, 2017
Accepted: November 21, 2017
Article in press: November 21, 2017
Published online: January 7, 2018
Processing time: 97 Days and 8.5 Hours
Ulcerative colitis (UC) is an inflammatory disease that mainly affects the colon and rectum. It is believed that genetic factors, host immune system disorders, intestinal microbiota dysbiosis, and environmental factors contribute to the pathogenesis of UC. However, studies on the role of intestinal microbiota in the pathogenesis of UC have been inconclusive. Studies have shown that probiotics improve intestinal mucosa barrier function and immune system function and promote secretion of anti-inflammatory factors, thereby inhibiting the growth of harmful bacteria in the intestine. Fecal microbiota transplantation (FMT) can reduce bowel permeability and thus the severity of disease by increasing the production of short-chain fatty acids, especially butyrate, which help maintain the integrity of the epithelial barrier. FMT can also restore immune dysbiosis by inhibiting Th1 differentiation, activity of T cells, leukocyte adhesion, and production of inflammatory factors. Probiotics and FMT are being increasingly used to treat UC, but their use is controversial because of uncertain efficacy. Here, we briefly review the role of intestinal microbiota in the pathogenesis and treatment of UC.
Core tip: As we all know, genetic factors, host immune system disorders, intestinal microbiota dysbiosis, and environmental factors contribute to the pathogenesis of ulcerative colitis (UC). In this review, we explore the mechanism and clinical application of intestinal microbiota such as probiotics and fecal microbiota transplantation in UC so that we can use these tools to cure more diseases. Enteric microbiota leads to new therapeutic strategies for UC.
- Citation: Shen ZH, Zhu CX, Quan YS, Yang ZY, Wu S, Luo WW, Tan B, Wang XY. Relationship between intestinal microbiota and ulcerative colitis: Mechanisms and clinical application of probiotics and fecal microbiota transplantation. World J Gastroenterol 2018; 24(1): 5-14
- URL: https://www.wjgnet.com/1007-9327/full/v24/i1/5.htm
- DOI: https://dx.doi.org/10.3748/wjg.v24.i1.5
The etiology and pathogenesis of ulcerative colitis (UC) are complex. UC is believed to be caused by an imbalance between intestinal microbiota and mucosal immunity, resulting in excessive intestinal inflammation[1]. Thus, dysbiosis of intestinal microbiota contributes to the pathogenesis of UC. In UC patients, the intestinal microbial population and functional diversity and stability of intestinal bacteria are impaired, with reductions in specific Firmicutes bacteria and increased Bacteroidetes bacteria and facultative anaerobes[2].
Probiotics are increasingly being used to treat UC. Probiotics are living nonpathogenic bacteria, such as Lactobacillus, Bifidobacterium, and Enterococcus[3]. The bacteria in probiotics can benefit the intestinal and immune systems[4]. Probiotics can restore the function of disturbed mucosal barrier[5], correct intestinal microbiota imbalance, inhibit competition of potential pathogens, improve local and systemic immunity, and enhance intestinal barrier function[6]. Probiotics can effectively induce and maintain remission in UC patients, suggesting that optimizing intestinal microbiota is key in the treatment of UC.
Fecal microbiota transplantation (FMT) also shows promise in UC. FMT aims to quickly restore the normal function and composition of intestinal microbiota, which can help treat intestinal and parenteral diseases. How FMT improves inflammatory bowel disease (IBD) is not completely understood. Studies have shown that after FMT, the intestinal microbiota composition of recipients was consistent with that of the donors[7]. The mechanisms of action of FMT might be the competitive inhibition of pathogenic microorganisms and improvement of immunity and metabolism.
The pathogenesis of UC is complex and it is believed to be mediated by genetic susceptibility, microbial dysregulation, and environmental factors. In UC, mucosal permeability increases and the inflammatory reaction is caused by excessive reaction below the lymphoid tissue[8]. There exists impaired ileum barrier function, the reduction of mucin and goblet cells which produce mucin, and the decrease of epithelial NLRP6 in UC patients[9].
The intestinal microbiota and the bacteria that compose it, such as Firmicutes, Bacteroidetes, and Actinomycetes, are still not well understood[10,11]. According to aerobiosis or anaerobiosis, the intestinal microorganisms are divided into anaerobic bacteria, facultative anaerobic bacteria, and aerobic bacteria; anaerobic bacteria are the most abundant intestinal bacteria[12]. These bacteria are mainly distributed in the colon and distal small intestine. Most of the bacteria live on the surface of the intestinal mucosa. They attach to the surface of the intestinal epithelial cells to form a layer of bacterial biofilm, which ultimately affects the intestinal metabolism of nutrients, intestinal permeability, and intestinal immune system function.
According to their role in the host, intestinal microbiota can be divided into three categories. The first category contains the physiologic bacteria that are symbiotic with the host. They attach to the deep mucosal epithelial cells, and most are anaerobic bacteria. They are the dominant microbiota of the intestine (e.g., Bifidobacterium, Bacteroides, and Peptococcus) and play key roles in nutrition and immune regulation. The second category contains conditional pathogens that inhabit the host. They are mainly facultative aerobic bacteria and intestinal nondominant bacteria (e.g., Enterococcus and Enterobacter). These organisms are harmless when intestinal microecological balance is maintained but can be harmful to humans under certain conditions. The third category contains mostly pathogens (e.g., Proteus and Pseudomonas). When microecology is in balance, long-term colonization of pathogens is rare, and the number of these organisms is small and nonpathogenic. If changes in the enteral and external environments lead to a decline of intestinal-dominant microbiota, then intestinal microbiota imbalance will occur, with pathogens or conditional pathogens increasing to the point of causing disease.
Intestinal mucosal symbiotic bacteria promote intestinal stability. Symbiotic bacteria help inhibit intestinal colonization by pathogens. Highly evolved bacteria occupy the intestine and prevent pathogens from invading the lamina propria layer. Intestinal bacteria stimulate the intestinal mucosa by causing a low level of inflammation. This inflammation contributes to the formation and improvement of the intestinal immune system, which monitors and removes harmful bacteria to maintain the body health. Intestinal bacteria also participate in important physiologic metabolic activities. Propionibacterium freudenreichii ET-3 can produce large amounts of vitamin K2 precursor material, which can activate aroma receptors, participate in substance metabolism, and detoxify and inhibit dextran sodium sulfate-induced colitis in vivo[13]. When the composition of intestinal symbiotic bacteria changes, the intestinal microbiota becomes unbalanced, causing an intestinal immune response[14].
Microorganisms release many biologically active substances, such as intestinal short-chain fatty acids (SCFAs), which have a strong immunomodulatory effect. Acetic acid, butyric acid, and propionic acid are the most abundant SCFAs[15]. SCFAs act as the main energy substrates and directly influence the host digestive tract through phenotypic alteration of colonic epithelial cells. They can also act as tumor suppressors and have recently been shown to be modulators of the intestinal neuroendocrine system. In addition, SCFAs are involved in anti-inflammatory gene regulation processes both in vitro and in vivo[16]. Faecalibacterium (F.) prausnitzii and Roseburia are butyric acid-producing bacteria that are usually considered as probiotic microorganisms. Butyrate can protect the integrity of the intestinal epithelium, promote the intestinal immune response, inhibit the growth of tumor cells, and reduce the activity of cancer-promoting enzymes, thus protecting the intestinal wall and reducing intestinal inflammation and colorectal cancer incidence[17].
There is close relationship between the pathogenesis of UC and intestinal microbiota. The steady state of the intestinal microbiota is important in preventing the excessive growth of certain microorganisms. Dysbiosis of the intestinal microbiota may be a contributing factor in some diseases and conditions, such as obesity, metabolic syndrome, autoimmune diseases, necrotizing enterocolitis, skin disease, UC, Crohn’s disease (CD), and irritable bowel syndrome[18]. When the balance of intestinal microbiota is broken, the intestinal defense function and immunoregulatory function are decreased, the immunity of the body is reduced, and the relative pathogenic factors are increased so as to cause the intestinal mucosal invasion or aggravate the diseases. The pathogenesis of UC is complex, and the interaction between the host and intestinal microbiota may be a key factor. Under normal circumstances, the host’s innate and adaptive immunity prevents the invasion of harmful bacteria while tolerating the normal microbiota. However, if the microbiota is imbalanced, immunity is compromised. The intestinal mucosal immune response is overstimulated, which can lead to disease[19].
Disrupted intestinal microbiota can cause intestinal inflammatory responses. Disruption in the microbiota results in a rapid increase in harmful bacteria in the intestine. In addition, release of enterotoxin increases intestinal mucosal permeability, and production of immunosuppressive protein results in immune dysfunction. Growing populations of harmful bacteria directly invade and damage the intestinal epithelial cells, resulting in damage to the intestinal mucosal barrier. Excessive growth of some bacteria affects metabolic and energy metabolism, triggering intestinal inflammation and damage of the intestinal mucosa. Intestinal mucosal barrier function declines, the shield function of the intestinal wall diminishes, and intestinal microbiota is translocated, which further damages the intestinal mucosal barrier, causing a vicious cycle and aggravating the intestinal inflammatory response. Animal experiments have found that colitis can be induced in animal models by disrupting intestinal bacteria. Sterile animals with IL-10 or HLA-B27 knockout did not develop colitis[20], suggesting that intestinal microbiota is essential for the occurrence of UC.
A large number of bacteria are adhered to intestinal epithelial cells of CD patients compared with healthy people. Microbiota might be one of the key factors of activating the intestinal immune system and inducing CD. In addition, the dysregulation of resident microbiota may be the major factor of inducing IBD. The occurrence of intestinal inflammation might cause the production of a variety of different types of instant cell factors.
Many studies have demonstrated that the composition and function of intestinal microbiota in UC patients are compromised. Some bacteria such as Akkermansia (A.) muciniphila are decreased. A. muciniphila is one of the most abundant members of the human gut microbiota, representing between 1% and 5% of human intestinal microbes. Several studies have shown a relationship between UC and A. muciniphila. A. muciniphila was decreased in UC patients, along with Roseburia bacteria[21]. It was also decreased in a mouse model of carrageenan-induced colitis[22]. Therefore, A. muciniphila might be a new target of UC. Abnormal microbiota reduced the complexity of the intestinal microbial ecosystem, which is a common feature of patients with UC and CD[23]. Several microbial pathogens are possibly related to intestinal inflammation and thus UC patients may harbor Mycobacterium avium paratuberculosis[24,25], adherent-invasive Escherichia coli[26], Clostridium (C.) difficile[27-29], Helicobacter species[30-32], Salmonella species[33], Yersinia species[34], Fusobacterium species[35], norovirus[36], and Listeria species[37]. However, the exact pathogenesis is unclear.
In one study, qualitative and quantitative changes were demonstrated in the composition of enteric bacteria in patients with UC[38]. The diversity of bacteria in UC patients was less than that of the control group. The number of dominant bacteria was decreased, the number of pathogens was increased, and the proportions of each strain of the dominant microbiota were not balanced. Studies have found that the diversity of intestinal microbiota in UC patients is decreased by approximately 25% compared with that of healthy controls. Firmicutes and Bacteroidetes were decreased, while Proteobacteria and Actinomycetes were increased. Machiels et al[39] found that two important butyrate-producing Firmicutes bacteria, Roseburia hominis and F. prausnitzii, were significantly decreased in UC patients. Varela et al[40] found that the number of F. prausnitzii was significantly increased in the remission period of UC, indicating that F. prausnitzii may play a vital role in the treatment of UC.
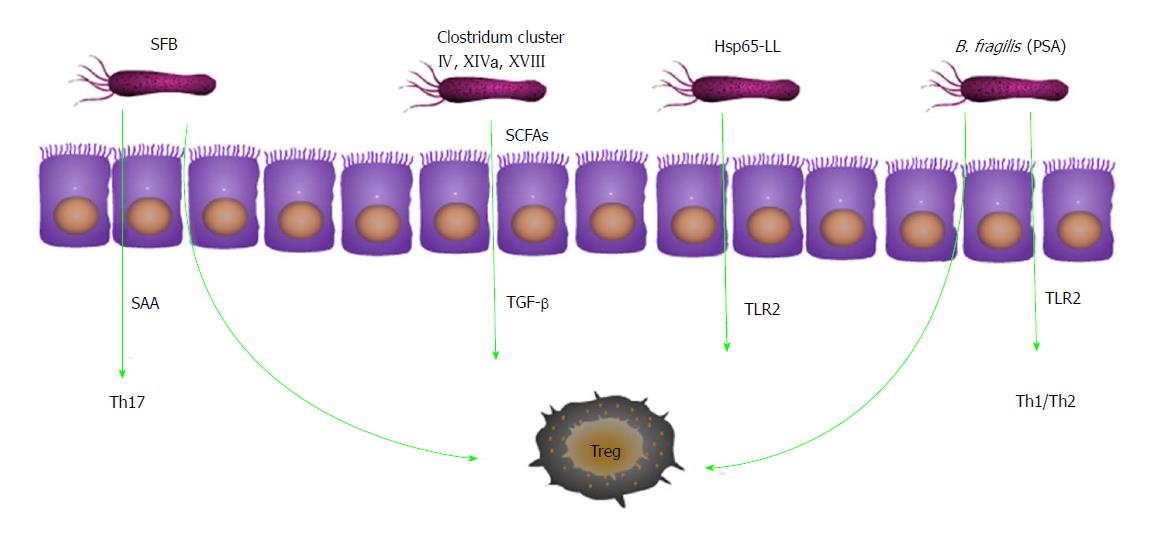
Some bacteria affect the differentiation of T-cell subsets and thus influence the occurrence and development of inflammation. Bacteroides (B.) fragilis and capsular lipopolysaccharide A improved the balance of Th1/Th2 cells in mice by activating NF-κB via TLR2 and regulating the secretion of TNF-α and IL-12[41]. Other studies have found that B. fragilis affected the steady state of mucosal T cells by inducing the production of regulatory T (Treg) cells in a 2,4,6-trinitrobenzene sulfonic acid-induced colitis mouse model[42]. In the small intestine and colon, segmented filamentous bacteria (SFB) can increase the number of Treg cells[43] and can also regulate the differentiation of Th17 cells and promote the production of IL-22-producing CD4+ T cells. The mechanism might be that SFB stimulates the production of serum amyloid A (SAA), which is an early inflammatory marker, and SAA induces a specific Th17 response in vivo[44]. These findings suggest that lipopolysaccharide A may play a dual role in intestinal homeostasis. Mice treated with Lactobacillus reuteri had a higher percentage of Treg cells[45]. Clostridium clusters IV, XIVa, and XVIII secrete SCFAs to stimulate the production of TGF-β by intestinal epithelial cells and induce Treg cell differentiation to promote intestinal mucosal immune tolerance[46]. Hsp65-producing Lactococcus lactis inhibited experimental autoimmune encephalomyelitis, and the mechanism might be related to the induction of CD4+/Foxp3+ and CD4+/LAP+ Treg cells and the TLR2 signaling pathway in vivo[47] (Figure 1).
Probiotics can promote the secretion of anti-inflammatory factors by improving intestinal mucosal barrier and immune system function and thus inhibiting the growth of harmful bacteria in the intestine. Pathogens can weaken the barrier function of the intestinal mucosa and pass through or damage the mucosal wall. Probiotics can prevent or repair such damage.
The main mechanisms of probiotics include competing for adhesion sites and nutrients, maintaining the balance of normal intestinal microbiota, enhancing intestinal mucosal barrier function, promoting immune tolerance of the intestinal mucosa, interfering with intestinal inflammatory response, and inhibiting intestinal epithelial cell apoptosis. Studies have indicated that probiotics might be useful in the treatment of UC. Through the PI3K/Akt and NF-κB signaling pathway, probiotics decreased proinflammatory cytokines such as TNF-α and IL-1β and increased anti-inflammatory factor IL-10, thus improving the symptoms of UC.
Bifidobacterium is one of the first colonizers of the baby intestine and the main microbial organism of the adult intestine[48]. It is beneficial microbiota that promotes antitumor immunity and prevents recurrence of UC. Bifidobacterium breve reduces the shedding of apoptotic epithelial cells in an extracellular polysaccharides- and MyD88-dependent manner[49].
A meta-analysis containing a total of 1763 cases in 32 clinical trials showed that probiotics significantly increased the clinical remission rate of patients with active UC (P = 0.01; risk ratio = 1.51). Probiotics and aminosalicylic acid had similar clinical effects. Subgroup analysis showed that VSL#3 (Alfasigma) resulted in the most significant improvement in UC, followed by lactic acid bacteria and E coli[50]. Another study on UC patients aged 12 to 16 years showed that the recurrence rate was lower in the group treated with probiotics for 1 year than in the control group (21% vs 73%)[51]. One study showed that the addition of Lactobacillus species to the usual therapies reduced relapse in UC patients[52]. Oliva et al[53] also found that local administration of L. reuteri ATCC 5573 with a standard dose of mesalazine reduced inflammation of the rectal mucosa in children with UC. In addition, E. coli strain Nissle promoted long-term maintenance of remission in UC patients. Administration of Saccharomyces boulardii during treatment with mesalazine induced clinical remission in 71% of patients with active mild to moderate UC[54,55]. In 2013, Varela and colleagues[40] investigated the fecal bacteria of 116 UC patients in remission and 16 healthy subjects and found that F. prausnitzii colonization was significantly reduced in UC patients. In addition, oral administration of F. prausnitzii was found to help induce remission in UC patients[56].
However, the results with probiotics in UC have not been consistent. Studies have shown that probiotics can increase gastrointestinal peristalsis, induce diarrhea in IBD patients, change stool frequency, and increase disease activity[57]. A study by Wildt et al[58] demonstrated that there was no difference in UC remission between patients administered with Lactobacillus acidophilus LA-5 and Bifidobacterium and the control group. Larger sample sizes and more randomized controlled clinical trials are needed to clarify the role of probiotics in the treatment of UC.
Along with probiotics, FMT is also being investigated as a treatment for UC. FMT is the process of transplanting fecal bacteria from a donor into a recipient. Currently, no standard criteria exist for FMT donor screening[59]. Donors are often selected from relatives, spouses, friends, or healthy volunteers. The collection of feces is usually conducted on the day of transplantation. The feces are dissolved in saline or water, homogenized, and filtered to form a homogeneous solution. Stools are usually transplanted in 6 to 8 h[60]. Stool banks exist in many countries and may serve many clinical functions in the future[61,62]; however, internationally standardized stool banks have not yet been established.
The essence of FMT is to reconstruct intestinal microbiota and cure disease by normalizing abnormal immune and inflammatory responses, the numbers and activities of neurotransmitters and vasoactive substances, and energy metabolism. FMT is used to treat chronic gastrointestinal infections and IBD[60]. FMT can improve intestinal microecology and the permeability of the intestinal mucosa. It can activate the intestinal humoral immune response to induce synthesis of IgA, IgG, and IgM through the TLR pathway, thus protecting the intestinal mucosa. FMT can also reduce the pH value of the intestine and increase the adhesion of bacteria and H2O2 to competitively inhibit the adhesion and translocation of pathogens[63]. Finally, FMT can treat immune disorders by inhibiting the secretion of proinflammatory cytokines and promoting Th1 differentiation, T-cell activity, leukocyte adhesion, and immune-stimulatory factors.
In gastrointestinal homeostasis, the diversity of the microbiota prevents colonization and overgrowth of pathogens. FMT can reduce intestinal permeability by increasing the production of SCFAs, thereby reducing the severity of disease. Increased SCFAs, especially butyrate, which is the main source of energy in colonic epithelial cells, maintain the integrity of the epithelial barrier by reducing intestinal permeability. FMT can also restore the dysbiosis of microbiota. The proportions of beneficial bacteria are increased and the diversity is also increased. FMT can make the composition of microbiota be more likely to be similar to the donor for a long time. Recently, a study by Dutta et al[64] found that FMT increased the diversity of fecal microbiota and increased the proportion of Lachnospiraceae, which are butyrate-producing bacteria. These findings not only confirm the speculated mechanism of action of FMT, but also indicate that Lachnospiraceae might be the key bacteria in the success of FMT. This finding could lead to targeted and standardized FMT.
FMT is successful in treating recurrent C. difficile infection. FMT could also potentially be used to treat UC, although limited evidence exists. Only a few small case series have been reported, and the efficacy is inconsistent, ranging from 20% to 92% (Table 1). There are currently only three randomized controlled trials of FMT in UC (Table 2). Clinical trials have shown differences in efficacy of FMT, which might be due to differences in individual factors, severity of disease, sites of disease, FMT donors, infusion paths, infusion doses, and so on. A recently reported meta-analysis[65] included 11 studies (containing two randomized controlled trials, one open case-control study, and eight cohort studies) and 133 patients with UC. The results showed that the clinical remission rate was 30.4% in UC patients treated with FMT. There were no significant differences in the remission rates between single and multiple transplantations and between upper digestive tract and lower digestive tract transplantations. In contrast, some reports have indicated that repeated FMT may be better than single transplantation[66]. Although the remission rate of UC was not ideal, patients’ quality of life improved[67], and FMT is easy to perform and safe. Another meta-analysis of FMT showed that the clinical response rate of IBD was 22%[68]. After FMT, the intestinal microbiota diversity of UC patients increased, and the microbiota species converged with those of the donors[69-71]. However, in time, intestinal bacteria will revert back to the pretreatment state. Therefore, repeated FMT may be needed; thus, the ideal number of treatments and interval between transplantations need to be explored[72].
| Ref. | UC (n) | Stool material | Volume infusion | Infusion route | Frequency | Donor relationship | Characteristics of outcomes |
| Kump et al[92], 2013 | 15 | Fresh | 100 to 150 g | Rectosigmoidoscopy | 1 | Unrelated | None of the patients achieved CR |
| 300-500 mL | |||||||
| Kunde et al[75], 2013 | 10 | Fresh | 70 to 130 g 60 mL | Enemas | 5 | Related | 33% patients achieved CR at 1 wk |
| Angelberger et al[72], 2013 | 5 | Fresh | 60 g | Nasojejunal tube | 5 | Related or unrelated | (0/5) patients demonstrated a remission |
| 250 mL | |||||||
| Suskind et al[93], 2015 | 4 | NR | 30 g | Nasogastric Tube | 1 | Related or unrelated | (0/4) patients demonstrated a remission |
| 100 mL | |||||||
| Kellermayer et al[86], 2015 | 3 | Frozen | NR | Colonoscopy | 22-30 | A single donor | (3/3) patients demonstrated a remission |
| Damman et al[94], 2015 | 7 | Fresh | 175-290 mL | Colonoscopy | 1 | Related or unrelated | (1/7) patients demonstrated a temporary remission |
| Wei et al[67], 2015 | 11 | Fresh | 60 g | Colonoscopy | 5 | Unrelated | The Mayo scores of all patients decreased at 4 wk |
| 300 mL | |||||||
| Cui et al[66], 2015 | 15 | NR | NR | Gastroscope channel | 1 | 1 (73.3%) related; 2 (26.7%) unrelated | 28.6% patients achieved CR |
| Vermeire et al[95], 2016 | 8 | Fresh | 100 g | Nasojejunal tube or colonoscopy | 2 | Related | (2/8) demonstrated a remission at week 8 |
| 200 mL |
| Ref. | Rossen et al[69], 2015 | Moayyedi et al[70], 2015 | Paramsothy et al[96], 2017 |
| n (UC/placebo) | 48 (23/25) | 75 (38/37) | 85 (42/43) |
| UC arm | 50 mL, nasoduodenal, healthy donors | 50 mL enema, healthy donors | 150 mL, colonoscopic, unrelated donors |
| Placebo arm | Autologous FMT | 50 mL enema, water | 150 mL, colonoscopic, isotonic saline |
| Frequency | At weeks 0 and 3 | Once weekly for 6 wk | 5 d per week for 8 wk |
| Evaluation criterion | Remission (SCCAI ≤ 2 + ≥ 1-point decrease in the Mayo endoscopic score) at week 12 | Remission (a Mayo score ≤ 2 with an endoscopic Mayo score of 0) at week 7 | Remission (Mayo score ≤ 2, all subscores ≤ 1, and ≥ 1 point reduction in endoscopic subscore) at week 8. |
| Results | 30% with FMT vs 20% controls (P = 0.51) | 24% with FMT vs 5% placebo (P = 0.03) | 27% with FMT vs 8% placebo (P = 0.021) |
Few adverse reactions have been reported with probiotics[73,74], and FMT has also been regarded as a safe treatment strategy. Adverse events or serious complications of FMT are rarely reported but include abdominal cramps, bloating, constipation, and diarrhea. Fever and increased C-reactive protein level are common, but both are transient and self-limiting[75]. Other rare adverse reactions include sore throat and headache[76], abnormally low blood pressure[77], shingles[78], the occurrence of UC[79], norovirus infection[76], weight gain[80], peritonitis or enteritis[81,82], perforation of the colon, pneumonia[83], and cytomegalovirus infection[84]. Bacteremia has been reported after FMT[85]. After FMT, changes in the composition of intestinal microbiota may lead to changes in gene expression of the mucosal cells of the recipients[86], alteration of intestinal mucosal immune function[87], changes in the intestinal ecological environment[88], and differences in body metabolism[89]. FMT can affect gastrointestinal diseases as well as extra-gastrointestinal disorders[90]. Therefore, patients who undergo FMT require long-term follow-up to observe pathologic and physiologic effects of treatment.
In addition, because of the diversity and uncertainty of intestinal microbiota, the current treatment criteria for FMT are difficult to formulate. Controversy concerns whether intestinal microbiota should be managed as a drug or as a bodily organ. These problems hamper the standardization and specific application of FMT. Determining the effective bacteria and the ideal treatment subjects will help to standardize FMT and make treatment more consistent[91].
The pathogenesis of UC is closely related to intestinal microbiota, although the exact bacteria that contribute to UC have not been determined. The use of probiotics in UC is currently being investigated. Probiotics may help normalize the imbalance of intestinal microbiota, improve the microecological environment, enhance intestinal mucosal barrier function, and reduce gastrointestinal infections. However, determination of the specific mechanism and types of probiotics, the best concentrations, and ideal treatment methods requires further study. The treatment of UC with FMT is also promising, but many questions remain regarding this treatment as well. Clearly, more research is needed regarding the use of probiotics and FMT in UC.
Manuscript source: Unsolicited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: China
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C, C, C
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Bashashati M, Manguso F, Pellicano R, Sergi CM S- Editor: Gong ZM L- Editor: Wang TQ E- Editor: Huang Y
| 1. | Ahmed J, Reddy BS, Mølbak L, Leser TD, MacFie J. Impact of probiotics on colonic microflora in patients with colitis: a prospective double blind randomised crossover study. Int J Surg. 2013;11:1131-1136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 27] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 2. | Hansen J, Gulati A, Sartor RB. The role of mucosal immunity and host genetics in defining intestinal commensal bacteria. Curr Opin Gastroenterol. 2010;26:564-571. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 125] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 3. | Oelschlaeger TA. Mechanisms of probiotic actions - A review. Int J Med Microbiol. 2010;300:57-62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 406] [Cited by in RCA: 408] [Article Influence: 25.5] [Reference Citation Analysis (0)] |
| 4. | Bron PA, van Baarlen P, Kleerebezem M. Emerging molecular insights into the interaction between probiotics and the host intestinal mucosa. Nat Rev Microbiol. 2011;10:66-78. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 421] [Cited by in RCA: 444] [Article Influence: 31.7] [Reference Citation Analysis (0)] |
| 5. | Dicksved J, Schreiber O, Willing B, Petersson J, Rang S, Phillipson M, Holm L, Roos S. Lactobacillus reuteri maintains a functional mucosal barrier during DSS treatment despite mucus layer dysfunction. PLoS One. 2012;7:e46399. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 88] [Cited by in RCA: 99] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 6. | Sartor RB. Mechanisms of disease: pathogenesis of Crohn’s disease and ulcerative colitis. Nat Clin Pract Gastroenterol Hepatol. 2006;3:390-407. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1158] [Cited by in RCA: 1270] [Article Influence: 66.8] [Reference Citation Analysis (2)] |
| 7. | Bakken JS, Borody T, Brandt LJ, Brill JV, Demarco DC, Franzos MA, Kelly C, Khoruts A, Louie T, Martinelli LP. Treating Clostridium difficile infection with fecal microbiota transplantation. Clin Gastroenterol Hepatol. 2011;9:1044-1049. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 650] [Cited by in RCA: 681] [Article Influence: 48.6] [Reference Citation Analysis (0)] |
| 8. | Actis GC, Pellicano R. The pathologic galaxy modulating the genotype and phenotype of inflammatory bowel disease: comorbidity, contiguity, and genetic and epigenetic factors. Minerva Med. 2016;107:401-412. [PubMed] |
| 9. | Alipour M, Zaidi D, Valcheva R, Jovel J, Martínez I, Sergi C, Walter J, Mason AL, Wong GK, Dieleman LA. Mucosal Barrier Depletion and Loss of Bacterial Diversity are Primary Abnormalities in Paediatric Ulcerative Colitis. J Crohns Colitis. 2016;10:462-471. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 179] [Article Influence: 19.9] [Reference Citation Analysis (0)] |
| 10. | Scaldaferri F, Gerardi V, Lopetuso LR, Del Zompo F, Mangiola F, Boškoski I, Bruno G, Petito V, Laterza L, Cammarota G. Gut microbial flora, prebiotics, and probiotics in IBD: their current usage and utility. Biomed Res Int. 2013;2013:435268. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 115] [Cited by in RCA: 143] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 11. | Seksik P. [Gut microbiota and IBD]. Gastroenterol Clin Biol. 2010;34 Suppl 1:S44-S51. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 27] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 12. | Sommer F, Bäckhed F. The gut microbiota--masters of host development and physiology. Nat Rev Microbiol. 2013;11:227-238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2011] [Cited by in RCA: 2386] [Article Influence: 198.8] [Reference Citation Analysis (0)] |
| 13. | Fukumoto S, Toshimitsu T, Matsuoka S, Maruyama A, Oh-Oka K, Takamura T, Nakamura Y, Ishimaru K, Fujii-Kuriyama Y, Ikegami S. Identification of a probiotic bacteria-derived activator of the aryl hydrocarbon receptor that inhibits colitis. Immunol Cell Biol. 2014;92:460-465. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 93] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 14. | Kaur N, Chen CC, Luther J, Kao JY. Intestinal dysbiosis in inflammatory bowel disease. Gut Microbes. 2011;2:211-216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 96] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 15. | Al-Lahham SH, Peppelenbosch MP, Roelofsen H, Vonk RJ, Venema K. Biological effects of propionic acid in humans; metabolism, potential applications and underlying mechanisms. Biochim Biophys Acta. 2010;1801:1175-1183. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 347] [Cited by in RCA: 384] [Article Influence: 25.6] [Reference Citation Analysis (0)] |
| 16. | Heerdt BG, Houston MA, Augenlicht LH. Short-chain fatty acid-initiated cell cycle arrest and apoptosis of colonic epithelial cells is linked to mitochondrial function. Cell Growth Differ. 1997;8:523-532. [PubMed] |
| 17. | Rose DJ, DeMeo MT, Keshavarzian A, Hamaker BR. Influence of dietary fiber on inflammatory bowel disease and colon cancer: importance of fermentation pattern. Nutr Rev. 2007;65:51-62. [PubMed] |
| 18. | Xu MQ, Cao HL, Wang WQ, Wang S, Cao XC, Yan F, Wang BM. Fecal microbiota transplantation broadening its application beyond intestinal disorders. World J Gastroenterol. 2015;21:102-111. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 161] [Cited by in RCA: 156] [Article Influence: 15.6] [Reference Citation Analysis (2)] |
| 19. | Zhang YJ, Li S, Gan RY, Zhou T, Xu DP, Li HB. Impacts of gut bacteria on human health and diseases. Int J Mol Sci. 2015;16:7493-7519. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 442] [Cited by in RCA: 589] [Article Influence: 58.9] [Reference Citation Analysis (0)] |
| 20. | Bamias G, Nyce MR, De La Rue SA, Cominelli F; American College of Physicians; American Physiological Society. New concepts in the pathophysiology of inflammatory bowel disease. Ann Intern Med. 2005;143:895-904. [PubMed] |
| 21. | Bajer L, Kverka M, Kostovcik M, Macinga P, Dvorak J, Stehlikova Z, Brezina J, Wohl P, Spicak J, Drastich P. Distinct gut microbiota profiles in patients with primary sclerosing cholangitis and ulcerative colitis. World J Gastroenterol. 2017;23:4548-4558. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 215] [Cited by in RCA: 261] [Article Influence: 32.6] [Reference Citation Analysis (3)] |
| 22. | Shang Q, Sun W, Shan X, Jiang H, Cai C, Hao J, Li G, Yu G. Carrageenan-induced colitis is associated with decreased population of anti-inflammatory bacterium, Akkermansia muciniphila, in the gut microbiota of C57BL/6J mice. Toxicol Lett. 2017;279:87-95. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 123] [Article Influence: 15.4] [Reference Citation Analysis (0)] |
| 23. | Manichanh C, Borruel N, Casellas F, Guarner F. The gut microbiota in IBD. Nat Rev Gastroenterol Hepatol. 2012;9:599-608. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 771] [Cited by in RCA: 894] [Article Influence: 68.8] [Reference Citation Analysis (0)] |
| 24. | Autschbach F, Eisold S, Hinz U, Zinser S, Linnebacher M, Giese T, Löffler T, Büchler MW, Schmidt J. High prevalence of Mycobacterium avium subspecies paratuberculosis IS900 DNA in gut tissues from individuals with Crohn’s disease. Gut. 2005;54:944-949. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 126] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 25. | Sechi LA, Scanu AM, Molicotti P, Cannas S, Mura M, Dettori G, Fadda G, Zanetti S. Detection and Isolation of Mycobacterium avium subspecies paratuberculosis from intestinal mucosal biopsies of patients with and without Crohn’s disease in Sardinia. Am J Gastroenterol. 2005;100:1529-1536. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 155] [Cited by in RCA: 149] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 26. | Mei L, Targan SR, Landers CJ, Dutridge D, Ippoliti A, Vasiliauskas EA, Papadakis KA, Fleshner PR, Rotter JI, Yang H. Familial expression of anti-Escherichia coli outer membrane porin C in relatives of patients with Crohn’s disease. Gastroenterology. 2006;130:1078-1085. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 64] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 27. | Reddy SS, Brandt LJ. Clostridium difficile infection and inflammatory bowel disease. J Clin Gastroenterol. 2013;47:666-671. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 36] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 28. | Deshpande A, Pasupuleti V, Pant C, Rolston DD, Sferra TJ. Diagnostic testing for Clostridium difficile infection in patients with inflammatory bowel disease. J Clin Gastroenterol. 2013;47:737-738. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 29. | Markowitz JE, Brown KA, Mamula P, Drott HR, Piccoli DA, Baldassano RN. Failure of single-toxin assays to detect clostridium difficile infection in pediatric inflammatory bowel disease. Am J Gastroenterol. 2001;96:2688-2690. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 23] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 30. | Laharie D, Asencio C, Asselineau J, Bulois P, Bourreille A, Moreau J, Bonjean P, Lamarque D, Pariente A, Soulé JC. Association between entero-hepatic Helicobacter species and Crohn’s disease: a prospective cross-sectional study. Aliment Pharmacol Ther. 2009;30:283-293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 57] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 31. | Sonnenberg A, Genta RM. Low prevalence of Helicobacter pylori infection among patients with inflammatory bowel disease. Aliment Pharmacol Ther. 2012;35:469-476. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 84] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 32. | Thomson JM, Hansen R, Berry SH, Hope ME, Murray GI, Mukhopadhya I, McLean MH, Shen Z, Fox JG, El-Omar E. Enterohepatic helicobacter in ulcerative colitis: potential pathogenic entities? PLoS One. 2011;6:e17184. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 73] [Cited by in RCA: 71] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 33. | Gradel KO, Nielsen HL, Schønheyder HC, Ejlertsen T, Kristensen B, Nielsen H. Increased short- and long-term risk of inflammatory bowel disease after salmonella or campylobacter gastroenteritis. Gastroenterology. 2009;137:495-501. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 317] [Cited by in RCA: 315] [Article Influence: 19.7] [Reference Citation Analysis (0)] |
| 34. | Saebo A, Vik E, Lange OJ, Matuszkiewicz L. Inflammatory bowel disease associated with Yersinia enterocolitica O:3 infection. Eur J Intern Med. 2005;16:176-182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 47] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 35. | Ohkusa T, Okayasu I, Ogihara T, Morita K, Ogawa M, Sato N. Induction of experimental ulcerative colitis by Fusobacterium varium isolated from colonic mucosa of patients with ulcerative colitis. Gut. 2003;52:79-83. [PubMed] |
| 36. | Chamaillard M, Cesaro A, Lober PE, Hober D. Decoding norovirus infection in Crohn’s disease. Inflamm Bowel Dis. 2014;20:767-770. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 37. | Huijsdens XW, Linskens RK, Taspinar H, Meuwissen SG, Vandenbroucke-Grauls CM, Savelkoul PH. Listeria monocytogenes and inflammatory bowel disease: detection of Listeria species in intestinal mucosal biopsies by real-time PCR. Scand J Gastroenterol. 2003;38:332-333. [PubMed] |
| 38. | Chassaing B, Darfeuille-Michaud A. The commensal microbiota and enteropathogens in the pathogenesis of inflammatory bowel diseases. Gastroenterology. 2011;140:1720-1728. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 326] [Cited by in RCA: 353] [Article Influence: 25.2] [Reference Citation Analysis (0)] |
| 39. | Machiels K, Joossens M, Sabino J, De Preter V, Arijs I, Eeckhaut V, Ballet V, Claes K, Van Immerseel F, Verbeke K. A decrease of the butyrate-producing species Roseburia hominis and Faecalibacterium prausnitzii defines dysbiosis in patients with ulcerative colitis. Gut. 2014;63:1275-1283. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1036] [Cited by in RCA: 1312] [Article Influence: 119.3] [Reference Citation Analysis (3)] |
| 40. | Varela E, Manichanh C, Gallart M, Torrejón A, Borruel N, Casellas F, Guarner F, Antolin M. Colonisation by Faecalibacterium prausnitzii and maintenance of clinical remission in patients with ulcerative colitis. Aliment Pharmacol Ther. 2013;38:151-161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 184] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 41. | Wang Q, McLoughlin RM, Cobb BA, Charrel-Dennis M, Zaleski KJ, Golenbock D, Tzianabos AO, Kasper DL. A bacterial carbohydrate links innate and adaptive responses through Toll-like receptor 2. J Exp Med. 2006;203:2853-2863. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 233] [Cited by in RCA: 217] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 42. | Round JL, Mazmanian SK. Inducible Foxp3+ regulatory T-cell development by a commensal bacterium of the intestinal microbiota. Proc Natl Acad Sci USA. 2010;107:12204-12209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1484] [Cited by in RCA: 1665] [Article Influence: 111.0] [Reference Citation Analysis (0)] |
| 43. | Gaboriau-Routhiau V, Rakotobe S, Lécuyer E, Mulder I, Lan A, Bridonneau C, Rochet V, Pisi A, De Paepe M, Brandi G. The key role of segmented filamentous bacteria in the coordinated maturation of gut helper T cell responses. Immunity. 2009;31:677-689. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1045] [Cited by in RCA: 1127] [Article Influence: 70.4] [Reference Citation Analysis (0)] |
| 44. | Ivanov II, Atarashi K, Manel N, Brodie EL, Shima T, Karaoz U, Wei D, Goldfarb KC, Santee CA, Lynch SV. Induction of intestinal Th17 cells by segmented filamentous bacteria. Cell. 2009;139:485-498. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3529] [Cited by in RCA: 3507] [Article Influence: 219.2] [Reference Citation Analysis (0)] |
| 45. | Karimi K, Inman MD, Bienenstock J, Forsythe P. Lactobacillus reuteri-induced regulatory T cells protect against an allergic airway response in mice. Am J Respir Crit Care Med. 2009;179:186-193. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 269] [Cited by in RCA: 285] [Article Influence: 16.8] [Reference Citation Analysis (0)] |
| 46. | Atarashi K, Tanoue T, Oshima K, Suda W, Nagano Y, Nishikawa H, Fukuda S, Saito T, Narushima S, Hase K. Treg induction by a rationally selected mixture of Clostridia strains from the human microbiota. Nature. 2013;500:232-236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1875] [Cited by in RCA: 2126] [Article Influence: 177.2] [Reference Citation Analysis (2)] |
| 47. | Gomes-Santos AC, de Oliveira RP, Moreira TG, Castro-Junior AB, Horta BC, Lemos L, de Almeida LA, Rezende RM, Cara DC, Oliveira SC. Hsp65-Producing Lactococcus lactis Prevents Inflammatory Intestinal Disease in Mice by IL-10- and TLR2-Dependent Pathways. Front Immunol. 2017;8:30. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 36] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 48. | Ventura M, Turroni F, Lugli GA, van Sinderen D. Bifidobacteria and humans: our special friends, from ecological to genomics perspectives. J Sci Food Agric. 2014;94:163-168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 81] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 49. | Hughes KR, Harnisch LC, Alcon-Giner C, Mitra S, Wright CJ, Ketskemety J, van Sinderen D, Watson AJ, Hall LJ. Bifidobacterium breve reduces apoptotic epithelial cell shedding in an exopolysaccharide and MyD88-dependent manner. Open Biol. 2017;. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 55] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 50. | Shen J, Zuo ZX, Mao AP. Effect of probiotics on inducing remission and maintaining therapy in ulcerative colitis, Crohn’s disease, and pouchitis: meta-analysis of randomized controlled trials. Inflamm Bowel Dis. 2014;20:21-35. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 230] [Cited by in RCA: 230] [Article Influence: 20.9] [Reference Citation Analysis (0)] |
| 51. | Sarowska J, Choroszy-Król I, Regulska-Ilow B, Frej-Mądrzak M, Jama-Kmiecik A. The therapeutic effect of probiotic bacteria on gastrointestinal diseases. Adv Clin Exp Med. 2013;22:759-766. [PubMed] |
| 52. | Cain AM, Karpa KD. Clinical utility of probiotics in inflammatory bowel disease. Altern Ther Health Med. 2011;17:72-79. [PubMed] |
| 53. | Oliva S, Di Nardo G, Ferrari F, Mallardo S, Rossi P, Patrizi G, Cucchiara S, Stronati L. Randomised clinical trial: the effectiveness of Lactobacillus reuteri ATCC 55730 rectal enema in children with active distal ulcerative colitis. Aliment Pharmacol Ther. 2012;35:327-334. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 234] [Cited by in RCA: 203] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 54. | Schultz M. Clinical use of E. coli Nissle 1917 in inflammatory bowel disease. Inflamm Bowel Dis. 2008;14:1012-1018. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 217] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 55. | Kruis W, Fric P, Pokrotnieks J, Lukás M, Fixa B, Kascák M, Kamm MA, Weismueller J, Beglinger C, Stolte M. Maintaining remission of ulcerative colitis with the probiotic Escherichia coli Nissle 1917 is as effective as with standard mesalazine. Gut. 2004;53:1617-1623. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 851] [Cited by in RCA: 817] [Article Influence: 38.9] [Reference Citation Analysis (0)] |
| 56. | Siaw YH, Hart A. Commentary: is Faecalibacterium prausnitzii a potential treatment for maintaining remission in ulcerative colitis? Aliment Pharmacol Ther. 2013;38:551. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 57. | Agathou CL, Beales IL. Factors associated with the use of probiotics in patients with inflammatory bowel disease. F1000Res. 2013;2:69. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 58. | Wildt S, Nordgaard I, Hansen U, Brockmann E, Rumessen JJ. A randomised double-blind placebo-controlled trial with Lactobacillus acidophilus La-5 and Bifidobacterium animalis subsp. lactis BB-12 for maintenance of remission in ulcerative colitis. J Crohns Colitis. 2011;5:115-121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 103] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 59. | Di Bella S, Drapeau C, García-Almodóvar E, Petrosillo N. Fecal microbiota transplantation: the state of the art. Infect Dis Rep. 2013;5:e13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 20] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 60. | Smits LP, Bouter KE, de Vos WM, Borody TJ, Nieuwdorp M. Therapeutic potential of fecal microbiota transplantation. Gastroenterology. 2013;145:946-953. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 421] [Cited by in RCA: 470] [Article Influence: 39.2] [Reference Citation Analysis (1)] |
| 61. | Terveer EM, van Beurden YH, Goorhuis A, Seegers JFML, Bauer MP, van Nood E, Dijkgraaf MGW, Mulder CJJ, Vandenbroucke-Grauls CMJE, Verspaget HW. How to: Establish and run a stool bank. Clin Microbiol Infect. 2017;23:924-930. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 122] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 62. | Kazerouni A, Burgess J, Burns LJ, Wein LM. Optimal screening and donor management in a public stool bank. Microbiome. 2015;3:75. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 34] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 63. | Yang YX, Chen X, Gan HT. Toll-like receptor 2 regulates intestinal inflammation by controlling integrity of the enteric nervous system: why were TLR3’s roles not tested? Gastroenterology. 2014;146:1428. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 64. | Dutta SK, Girotra M, Garg S, Dutta A, von Rosenvinge EC, Maddox C, Song Y, Bartlett JG, Vinayek R, Fricke WF. Efficacy of combined jejunal and colonic fecal microbiota transplantation for recurrent Clostridium difficile Infection. Clin Gastroenterol Hepatol. 2014;12:1572-1576. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 65] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 65. | Sun D, Li W, Li S, Cen Y, Xu Q, Li Y, Sun Y, Qi Y, Lin Y, Yang T. Fecal Microbiota Transplantation as a Novel Therapy for Ulcerative Colitis: A Systematic Review and Meta-Analysis. Medicine (Baltimore). 2016;95:e3765. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 26] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 66. | Cui B, Li P, Xu L, Zhao Y, Wang H, Peng Z, Xu H, Xiang J, He Z, Zhang T. Step-up fecal microbiota transplantation strategy: a pilot study for steroid-dependent ulcerative colitis. J Transl Med. 2015;13:298. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 123] [Cited by in RCA: 123] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 67. | Wei Y, Zhu W, Gong J, Guo D, Gu L, Li N, Li J. Fecal Microbiota Transplantation Improves the Quality of Life in Patients with Inflammatory Bowel Disease. Gastroenterol Res Pract. 2015;2015:517597. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 73] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 68. | Colman RJ, Rubin DT. Fecal microbiota transplantation as therapy for inflammatory bowel disease: a systematic review and meta-analysis. J Crohns Colitis. 2014;8:1569-1581. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 346] [Cited by in RCA: 334] [Article Influence: 30.4] [Reference Citation Analysis (1)] |
| 69. | Rossen NG, Fuentes S, van der Spek MJ, Tijssen JG, Hartman JH, Duflou A, Löwenberg M, van den Brink GR, Mathus-Vliegen EM, de Vos WM. Findings From a Randomized Controlled Trial of Fecal Transplantation for Patients With Ulcerative Colitis. Gastroenterology. 2015;149:110-118.e4. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 616] [Cited by in RCA: 678] [Article Influence: 67.8] [Reference Citation Analysis (0)] |
| 70. | Moayyedi P, Surette MG, Kim PT, Libertucci J, Wolfe M, Onischi C, Armstrong D, Marshall JK, Kassam Z, Reinisch W. Fecal Microbiota Transplantation Induces Remission in Patients With Active Ulcerative Colitis in a Randomized Controlled Trial. Gastroenterology. 2015;149:102-109.e6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 930] [Cited by in RCA: 1076] [Article Influence: 107.6] [Reference Citation Analysis (1)] |
| 71. | Berg D, Clemente JC, Colombel JF. Can inflammatory bowel disease be permanently treated with short-term interventions on the microbiome? Expert Rev Gastroenterol Hepatol. 2015;9:781-795. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 45] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 72. | Angelberger S, Reinisch W, Makristathis A, Lichtenberger C, Dejaco C, Papay P, Novacek G, Trauner M, Loy A, Berry D. Temporal bacterial community dynamics vary among ulcerative colitis patients after fecal microbiota transplantation. Am J Gastroenterol. 2013;108:1620-1630. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 252] [Cited by in RCA: 259] [Article Influence: 21.6] [Reference Citation Analysis (0)] |
| 73. | Fujiya M, Ueno N, Kohgo Y. Probiotic treatments for induction and maintenance of remission in inflammatory bowel diseases: a meta-analysis of randomized controlled trials. Clin J Gastroenterol. 2014;7:1-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 52] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 74. | Mardini HE, Grigorian AY. Probiotic mix VSL#3 is effective adjunctive therapy for mild to moderately active ulcerative colitis: a meta-analysis. Inflamm Bowel Dis. 2014;20:1562-1567. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 101] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 75. | Kunde S, Pham A, Bonczyk S, Crumb T, Duba M, Conrad H Jr, Cloney D, Kugathasan S. Safety, tolerability, and clinical response after fecal transplantation in children and young adults with ulcerative colitis. J Pediatr Gastroenterol Nutr. 2013;56:597-601. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 212] [Cited by in RCA: 227] [Article Influence: 18.9] [Reference Citation Analysis (0)] |
| 76. | Schwartz M, Gluck M, Koon S. Norovirus gastroenteritis after fecal microbiota transplantation for treatment of Clostridium difficile infection despite asymptomatic donors and lack of sick contacts. Am J Gastroenterol. 2013;108:1367. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 115] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 77. | Solari PR, Fairchild PG, Noa LJ, Wallace MR. Tempered enthusiasm for fecal transplant. Clin Infect Dis. 2014;59:319. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 60] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 78. | Kleger A, Schnell J, Essig A, Wagner M, Bommer M, Seufferlein T, Härter G. Fecal transplant in refractory Clostridium difficile colitis. Dtsch Arztebl Int. 2013;110:108-115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 79. | De Leon LM, Watson JB, Kelly CR. Transient flare of ulcerative colitis after fecal microbiota transplantation for recurrent Clostridium difficile infection. Clin Gastroenterol Hepatol. 2013;11:1036-1038. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 158] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 80. | Alang N, Kelly CR. Weight gain after fecal microbiota transplantation. Open Forum Infect Dis. 2015;2:ofv004. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 270] [Cited by in RCA: 298] [Article Influence: 29.8] [Reference Citation Analysis (1)] |
| 81. | Gough E, Shaikh H, Manges AR. Systematic review of intestinal microbiota transplantation (fecal bacteriotherapy) for recurrent Clostridium difficile infection. Clin Infect Dis. 2011;53:994-1002. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 808] [Cited by in RCA: 719] [Article Influence: 51.4] [Reference Citation Analysis (0)] |
| 82. | Gregory JC, Buffa JA, Org E, Wang Z, Levison BS, Zhu W, Wagner MA, Bennett BJ, Li L, DiDonato JA. Transmission of atherosclerosis susceptibility with gut microbial transplantation. J Biol Chem. 2015;290:5647-5660. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 319] [Cited by in RCA: 376] [Article Influence: 34.2] [Reference Citation Analysis (0)] |
| 83. | Kelly CR, Kahn S, Kashyap P, Laine L, Rubin D, Atreja A, Moore T, Wu G. Update on Fecal Microbiota Transplantation 2015: Indications, Methodologies, Mechanisms, and Outlook. Gastroenterology. 2015;149:223-237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 429] [Cited by in RCA: 420] [Article Influence: 42.0] [Reference Citation Analysis (1)] |
| 84. | Hohmann EL, Ananthakrishnan AN, Deshpande V. Case Records of the Massachusetts General Hospital. Case 25-2014. A 37-year-old man with ulcerative colitis and bloody diarrhea. N Engl J Med. 2014;371:668-675. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 60] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 85. | Quera R, Espinoza R, Estay C, Rivera D. Bacteremia as an adverse event of fecal microbiota transplantation in a patient with Crohn’s disease and recurrent Clostridium difficile infection. J Crohns Colitis. 2014;8:252-253. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 121] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 86. | Kellermayer R, Nagy-Szakal D, Harris RA, Luna RA, Pitashny M, Schady D, Mir SA, Lopez ME, Gilger MA, Belmont J. Serial fecal microbiota transplantation alters mucosal gene expression in pediatric ulcerative colitis. Am J Gastroenterol. 2015;110:604-606. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 61] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 87. | Pamer EG. Fecal microbiota transplantation: effectiveness, complexities, and lingering concerns. Mucosal Immunol. 2014;7:210-214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 95] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 88. | Khoruts A, Weingarden AR. Emergence of fecal microbiota transplantation as an approach to repair disrupted microbial gut ecology. Immunol Lett. 2014;162:77-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 42] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 89. | Woting A, Blaut M. The Intestinal Microbiota in Metabolic Disease. Nutrients. 2016;8:202. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 163] [Cited by in RCA: 208] [Article Influence: 23.1] [Reference Citation Analysis (0)] |
| 90. | Konturek PC, Haziri D, Brzozowski T, Hess T, Heyman S, Kwiecien S, Konturek SJ, Koziel J. Emerging role of fecal microbiota therapy in the treatment of gastrointestinal and extra-gastrointestinal diseases. J Physiol Pharmacol. 2015;66:483-491. [PubMed] |
| 91. | Ianiro G, Bibbò S, Scaldaferri F, Gasbarrini A, Cammarota G. Fecal microbiota transplantation in inflammatory bowel disease: beyond the excitement. Medicine (Baltimore). 2014;93:e97. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 74] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 92. | Kump PK, Gröchenig HP, Lackner S, Trajanoski S, Reicht G, Hoffmann KM, Deutschmann A, Wenzl HH, Petritsch W, Krejs GJ. Alteration of intestinal dysbiosis by fecal microbiota transplantation does not induce remission in patients with chronic active ulcerative colitis. Inflamm Bowel Dis. 2013;19:2155-2165. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 177] [Cited by in RCA: 188] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 93. | Suskind DL, Singh N, Nielson H, Wahbeh G. Fecal microbial transplant via nasogastric tube for active pediatric ulcerative colitis. J Pediatr Gastroenterol Nutr. 2015;60:27-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 78] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 94. | Damman CJ, Brittnacher MJ, Westerhoff M, Hayden HS, Radey M, Hager KR, Marquis SR, Miller SI, Zisman TL. Low Level Engraftment and Improvement following a Single Colonoscopic Administration of Fecal Microbiota to Patients with Ulcerative Colitis. PLoS One. 2015;10:e0133925. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 65] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 95. | Vermeire S, Joossens M, Verbeke K, Wang J, Machiels K, Sabino J, Ferrante M, Van Assche G, Rutgeerts P, Raes J. Donor Species Richness Determines Faecal Microbiota Transplantation Success in Inflammatory Bowel Disease. J Crohns Colitis. 2016;10:387-394. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 204] [Cited by in RCA: 234] [Article Influence: 26.0] [Reference Citation Analysis (0)] |
| 96. | Paramsothy S, Kamm MA, Kaakoush NO, Walsh AJ, van den Bogaerde J, Samuel D, Leong RWL, Connor S, Ng W, Paramsothy R. Multidonor intensive faecal microbiota transplantation for active ulcerative colitis: a randomised placebo-controlled trial. Lancet. 2017;389:1218-1228. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 710] [Cited by in RCA: 868] [Article Influence: 108.5] [Reference Citation Analysis (0)] |