Published online Nov 21, 2016. doi: 10.3748/wjg.v22.i43.9562
Peer-review started: July 21, 2016
First decision: September 5, 2016
Revised: September 28, 2016
Accepted: October 27, 2016
Article in press: October 27, 2016
Published online: November 21, 2016
Processing time: 122 Days and 13.9 Hours
To investigate the type and timing of evolution of incidentally found branch-duct intraductal papillary mucinous neoplasms (bd-IPMN) of the pancreas addressed to magnetic resonance imaging cholangiopancreatography (MRCP) follow-up.
We retrospectively evaluated 72 patients who underwent, over the period 2006-2016, a total of 318 MRCPs (mean 4.4) to follow-up incidental, presumed bd-IPMN without signs of malignancy, found or confirmed at a baseline MRCP examination. Median follow-up time was 48.5 mo (range 13-95 mo). MRCPs were acquired on 1.5T and/or 3.0T systems using 2D and/or 3D technique. Image analysis assessed the rates of occurrence over the follow-up of the following outcomes: (1) imaging evolution, defined as any change in cysts number and/or size and/or appearance; and (2) alert findings, defined as worrisome features and/or high risk stigmata (e.g., thick septa, parietal thickening, mural nodules and involvement of the main pancreatic duct). Time to outcomes was described with the Kaplan-Meir approach. Cox regression model was used to investigate clinical or initial MRCP findings predicting cysts changes.
We found a total of 343 cysts (per-patient mean 5.1) with average size of 8.5 mm (range 5-25 mm). Imaging evolution was observed in 32/72 patients (44.4%; 95%CI: 32-9-56.6), involving 47/343 cysts (13.7%). There was a main trend towards small (< 10 mm) increase and/or decrease of cysts size at a median time of 22.5 mo. Alert findings developed in 6/72 patients (8.3%; 95%CI: 3.4-17.9) over a wide interval of time (13-63 mo). No malignancy was found on endoscopic ultrasound with fine-needle aspiration (5/6 cases) or surgery (1/6 cases). No clinical or initial MRCP features were significantly associated with changes in bd-IPMN appearance (P > 0.01).
Changes in MRCP appearance of incidental bd-IPNM were frequent over the follow-up (44.4%), with relatively rare (8.3%) occurrence of non-malignant alert findings that prompted further diagnostic steps. Changes occurred at a wide interval of time and were unpredictable, suggesting that imaging follow-up should be not discontinued, though MRCPs might be considerably delayed without a significant risk of missing malignancy.
Core tip: Branch-duct intraductal papillary mucinous neoplasms are a frequent incidental finding. The management of these lesions is challenging because of the need to balance low but concrete risk of developing malignancy over time versus costs of prolonged follow-up procedures. In this retrospective study, we showed high rate of imaging evolution (44.4%), with 8.3% of patients developing alert findings prompting further diagnostic steps, though there were no final diagnoses of malignancy. Since there were no clinical or imaging predictors of alert findings, our results suggest that prolonged follow-up is useful, but might be significantly delayed with minimal risk of missing malignancy.
- Citation: Girometti R, Pravisani R, Intini SG, Isola M, Cereser L, Risaliti A, Zuiani C. Evolution of incidental branch-duct intraductal papillary mucinous neoplasms of the pancreas: A study with magnetic resonance imaging cholangiopancreatography. World J Gastroenterol 2016; 22(43): 9562-9570
- URL: https://www.wjgnet.com/1007-9327/full/v22/i43/9562.htm
- DOI: https://dx.doi.org/10.3748/wjg.v22.i43.9562
Branch-duct intraductal papillary mucinous neoplasm (bd-IPMN) is a subtype of pancreatic cystic tumor developing from mucin-producing columnar cells lining the side branches of the main pancreatic duct (MPD). Accumulation of mucin typically associates with cystic or saccular dilatation of the involved ducts[1]. Despite incidence of IPMN has been estimated rare (0.48-2.04 per 100000 persons), most reports suggest that the frequency of disease increased over the last years, possibly because of the widespread use of sensitive imaging modalities such as computed tomography (CT) or Magnetic resonance imaging (MRI) with Magnetic resonance cholangiopancreatography (MRCP)[2]. Not surprisingly, most bd-IPMNs are discovered incidentally during examinations performed for causes unrelated to the pancreas[1,3,4]. Up to 70.6% of incidentally found pancreatic cysts show IPMN-like appearance on MRCP[5].
According to the Fukuoka consensus guidelines[1], bd-IPMN showing no symptoms, high-risk stigmata or worrisome morphological features can be follow-up with serial imaging if cysts are ≤ 3 cm in size. Suggested follow-up methods [CT, MRI or endoscopic ultrasound (EUS)] as well as the timing of surveillance are driven by cysts size. However, the management of non-operated, presumed bd-IPMN is challenging because of the small but concrete potential of harboring high-grade dysplasia or malignancy (overall cancer risk as high as 2% per year)[3,6], regardless of the size[1,7]. Additionally, a growing body of data suggests an associated risk of developing concomitant pancreatic duct adenocarcinoma (about 4%)[3] or extrapancreatic cancer[2] in patients with bd-IPMN.
Though a large amount of studies focused on the natural history of bd-IPMN[3,8], many of them did not clearly distinguish between symptomatic vs incidental lesion or suspicious vs not suspicious cysts at presentation, nor separated bd-IPMN from other cystic lesions of the pancreas. Moreover, little has been reported on the specific patterns of cysts evolution over time using MRCP, which is the examination of choice to evaluate bd-IPMN, given high contrast-resolution for fluid-containing structures[8]. This contributes to current uncertainty about the most appropriate scheduling and overall duration of imaging follow-up of incidental bd-IPMN, with impacts on costs, accessibility to examinations and patients’ quality of life.
The purpose of this study was to investigate MRCP patterns of evolution over time of incidental bd-IPMN showing no suspicious signs at the time of diagnosis.
This study was approved by local Ethical Committee and Hospital administration. Given the retrospective design, informed consent acquisition was waived, in accordance with laws and regulations of our country.
We searched our institutional database (period June 2006-May 2016) to identify: (1) patients showing incidental pancreatic cysts on a baseline MRCP performed to evaluate conditions clinically unrelated to the pancreas; and (2) patients who underwent a baseline MRCP to assess pancreatic cysts incidentally found with previous ultrasonography (US) and/or CT performed no more than one month before. A total of 153 subjects were found.
Of them, we included: (1) those with at least one cyst ≥ 5 mm in largest size or more than two cysts of any size, with at least one cyst ≥ 5 mm showing clear communication with the MPD; and (2) patients with a minimum of two follow-up MRCPs covering a whole period of at least 24 mo, unless the study outcomes have been reached before (see below). We excluded from the study fifty subjects showing the following conditions: recent onset or worsening of diabetes mellitus, cysts with no definite communication with the MPD and/or showing MRCP appearance typical for other cysts type (e.g., mucinous cystic neoplasm), signs of chronic pancreatitis and, more importantly, patients showing incidental bd-IPMN- or mixed-IPMN-like lesions with high risk stigmata and/or worrisome features (see below) at baseline examination (for which further diagnostic steps rather than imaging follow-up were required). We also excluded patients presenting with two or less cysts < 5 mm in size (n = 5), < 2 follow-up MRCPs (n = 17) and < 24 mo of follow-up period without cysts evolution (n = 9). Included patients showed a variety of indications to MRCP, mainly related to the biliary tract. None of them underwent MRI examinations prior to the baseline MRCP.
Baseline and follow-up examinations were performed on one of two 1.5T systems [Avanto (1) and Aera (2), Siemens Medical Systems, Erlangen, Germany] and/or a 3.0T magnet (Achieva, Philips HealthCare, Best, Netherlands) using a 8-channel, 18-channel and 16-channel surface body coil, respectively. To minimize the overlap of fluid signal from the stomach and small bowel, we administered orally 1 mL of Gd-based contrast agent diluted into 9 mL of water (directly on the magnet, immediately before MRCP acquisition) or, prior to 2010, 250 mL of blueberry juice 20 min before patients started the examination.
MRCP was performed with the 2D and/or 3D technique with the heavily T2-weighted sequences shown in Table 1. When using the 2D technique, eight single thick slices were placed along radial oblique coronal planes of the pancreas and biliary tree, at a distance of 20° each. In most cases, we acquired also 2D slices in the oblique transverse and oblique coronal planes of the pancreas. In the case of the 3D sequence, one single slab was positioned on the oblique coronal plane to cover both the biliary tree and the pancreas, based on individual anatomy of the patient. In a few cases, one or more 2D slices or the 3D sequence were repeated to cover the whole pancreas if not initially included entirely.
| 1.5T (1) | 1.5T (2) | 3.0T | ||||
| 2D | 3D | 2D | 3D | 2D | 3D | |
| Sequence | HASTE | TSE | HASTE | TSE | SS-FSE | FSE |
| Type of acquisition | Breath hold | Navigator gated | Breath hold | Navigator gated | Breath hold | Respiratory triggered |
| TR (ms) | 4000 | 2500 | 4500 | 2500 | 9449 | 1661.5 |
| TE (ms) | 735 | 685 | 725 | 700 | 740 | 401.2 |
| FOV (mm × mm) | 300 × 300 | 380 × 380 | 250 × 250 | 380 × 380 | 300 × 300 | 300 × 300 |
| Matrix (pixel × pixel) | 307 × 384 | 357 × 384 | 269 × 384 | 353 × 384 | 256 × 320 | 200 × 200 |
| Number of slices | 1 × 8 | 72 | 1 × 8 | 72 | 1 x 6 | 107 |
| Slice thickness (mm) | 40-50 | 1 | 50 | 1 | 40-50 | 0.75 |
| Number of excitations | 1 | 2 | 1 | 1.5 | 1 | 1 |
| Acquisition time | 4.5 s × 8 | 3 min 4 s (nominal) | 4.5 s × 8 | 5 min 1 s (nominal) | 9.3 s × 6 | 4 min 54 s (nominal) |
During baseline and follow-up examinations, MRCP was always associated, depending on the magnet, with the following sequences to provide anatomic representation of the upper abdomen and a reference for MRCP positioning: (1) transverse and coronal T2-weighted Half-Fourier single-shot turbo spin echo (HASTE) or equivalent Single shot fast spin echo (SS-FSE); (2) transverse single-shot Echo-planar Diffusion-weighted imaging; and (3) transverse T1-weighted 3D fat satured Volume Interpolatd Breath-hold Examination (VIBE) or T1 High Resolution Isotropic Volume Exicitation (THRIVE) imaging. Optional sequences included transverse T1-weighted Gradient echo in-phase/out of-phase and transverse T2-weighted Short tau inversion recovery (STIR) or Spectrally adiabatic inversion recovery (SPAIR). Multiphasic contrast-enhanced study was performed in selected cases of extrapancreatic or pancreatic findings (e.g., larger cysts, suspicious findings, cysts evolution) using 0.1 mmol/kg of gadobenate dimeglumine (Multihance, Bracco, Milan, Italy) at an injection rate of 2 mL/sec and a 3-4 mm thick transverse T1-weighted VIBE/THRIVE sequence.
Image analysis was performed in consensus by a senior radiologist (13 years of experience in abdominal imaging) and a radiology resident (2 years of experience), who reviewed baseline and follow-up examinations on two different workstations (Suite Estensa, Esaote, Genova, Italy; and Olea Sphere, Olea Medical, La Ciotat, France).
By having access to MRCP and additional MRI sequences for problem-solving interpretation, radiologists assessed the baseline pattern of cysts presentation, and then recorded any per-patient variation in terms of PC number (definite increase or decrease) and size (defined as any increase or decrease of at least 50% of the baseline largest diameter). They also assessed the occurrence during the follow-up of at least one of the following high-risk and/or worrisome findings as defined by Tanaka et al[1] in 2012: (1) cyst size ≥ 30 mm; (2) solid mural nodules with or without contrast enhancement; (3) wall thickening and/or thick/nodular internal septa; (4) dilatation of the MPD (caliber > 5 mm); and (5) abrupt change in the MPD caliber with distal pancreatic atrophy. Baseline size and subsequent variations were measured on HASTE/SS-FSE images and/or 2D MRCP images and/or thin source images in the case of the 3D MRCP using the same reference plane across different examinations. We followed the standards suggested by Dunn et al[9] to reduce variability in size measurement. When both techniques were available, 3D MRCP was preferred to assess changes in size. In the case of multilocular cysts, lesion diameter was measured as a whole. Readers recorded the time of occurrence from baseline MRCP (in mo) of any of the above variations, as well as the number of follow-up MRCPs and the interval of time (in mo) between them.
Radiologists were blinded to medical history and patients’ workup data, which were independently reviewed and collected by a resident in surgery. In particular, he recorded any MRCP-induced EUS or surgery, as well as final diagnosis as established by EUS-guided fine-needle aspiration or histological examination after surgery.
Data on cyst number and distribution of cysts at the time of baseline MRCPs and during the follow-up are presented with descriptive statistic, reporting mean values (± SD) and/or median values (together with the range of minimum and maximum values).
Concerning PC evolution, we assessed the prevalence of two outcomes over the whole period of follow-up, namely: (1) imaging evolution, defined as any change in number and/or size and/or appearance occurred in ≥ 1 follow-up examination, involving ≥ 1 cyst; (2) alert findings, defined as the occurrence of worrisome and/or high risk features prompting further investigation. According to our definitions, imaging evolution included cases of alert findings. Prevalence was calculated on a per-patient basis as the ratio between the cases of radiologic evolution or alert findings occurrence over the total number of patients × 100.
Time to radiological evolution or alert findings was described according to the Kaplan-Meier approach. Cox regression model was used to explore which clinical or imaging factor was associated with both outcomes. Variables included in the model were sex, age, type 2 diabetes mellitus, previous cholecystectomy for stones, autoimmune systemic disorders, alcoholic liver cirrhosis, history of extrapancreatic neoplasia (solid cancer and/or hematologic malignancy), and the pattern of bd-IPMN presentation on baseline MRCP, that is cysts size (< 10 mm, 10-19 mm and ≥ 20 mm) and cysts number (≤ 2, 2-5 and > 5).
Statistical analysis was performed by our referring biostatistician using a commercially available software (Stata/SE 14.1, Stata Corp LP, United States). Alfa level was sat 0.01.
Final study population included 72 subjects (17 male, 55 female), with a mean age of 63.0 ± 9.4 years at the time of baseline MRCP (range 44-83 years).
They showed a total of 343 PC arranged in a bd-IPMN pattern, with a per-patient mean of 5.1 ± 2.4 cysts (range 1-10). PC size spanned from 5 to 25 mm (mean size 8.5 ± 1.7 mm). Largest lesions’ diameter was lower than 10 mm in 277/343 cases (80.7%), between 10-19 mm in 58/343 (16.9%), and between 20-29 mm in the remaining 8/343 cases (2.4%).
None of the subjects developed symptoms related to the pancreas over the entire follow-up period.
After baseline examination, patients underwent a total of 318 follow-up MRCPs, corresponding to a per-patient mean of 4.4 ± 2.0 examinations (range 2-12). Mean and median time of follow-up were 52.6 ± 22.4 and 48.5 (range 13-95) mo, respectively, whereas mean and median interval of time between repeated MRCPs were 11.6 ± 6.7 and 17 (range 4-58) mo, respectively.
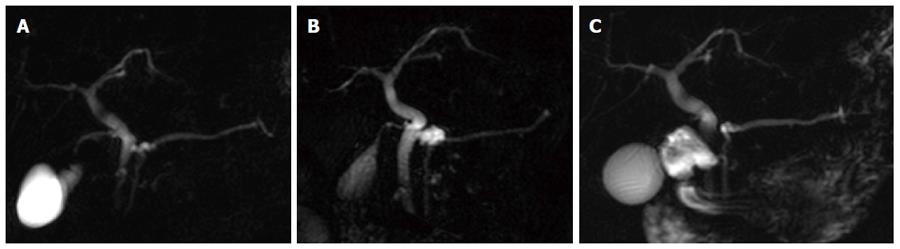
Over the entire follow-up, imaging evolution was observed in 32/72 patients (44.4%; 95%CI: 32.9-56.6), involving 47/343 cysts (13.7; 95%CI: 10.3-17.9) (per-patient mean 1.4 cysts). Table 2 details evolution patterns and the timing of changes we observed. Overall, PC tended to increase and/or decrease in size rather than in number (Figure 1), with mean variation in size less than 10 mm (around 5 mm for the majority evolution patterns), except for two 10 mm cysts in two patients which disappeared during the follow-up. In one patient (indicated as “other” in Table 2), a total of six cysts less than 10 mm on baseline MRCP showed minimal change in size in repeated examinations, with the largest variation occurring in one cyst moving from 5 mm to 12 mm, followed by subsequent decrease in size to 7 mm.
| Pattern of imaging evolution over the follow-up | Number of patients involved, n (95%CI) | Total number of cysts involved/per-patient mean number of cysts involved | Mean time of first change/mean time of last change1 (mo) | Median time of first change/median time of last change1 (mo), n (95%CI) | Mean size on baseline MRCP/Mean size at the end of the follow-up (mm) |
| Increase in size occurring in ≥ 1 examination | 14/72 | 17/1.2 | 34.2 ± 27.2/38.6 ± 29.6 | 23.5 (5-79)/25.5 (5-92) | 8.4 ± 5.7/13.5 ± 6.9 |
| 19.4% (11.4-30.8) | |||||
| Increase in size in ≥ 1 examination followed or alternating with a decrease in size and/or number in ≥ 1 examination | 5/72 | 8/1.6 | 21.0 ± 7.4/53.6 ± 23.0 | 21 (12-32)/48 (23-85) | 8.4 ± 3.2/13.6 ± 6.1 |
| 6.9% (2.6-16.1) | |||||
| Decrease in size occurring in ≥ 1 examination | 4/72 | 6/1.5 | 15.5 ± 16.8/17.7 ± 16.9 | 9 (4-40)/13.5 /(4-40) | 14.7 ± 7.6/10.5 ± 9.1 |
| 5.6% (1.8-14.3) | |||||
| Disappearance | 2/72 | 2/1.0 | 20.0 ± 11.3/20.0 ± 11.3 | 20 (12-28)/20 (12-28) | 10.0/0 |
| 1.4% (0.1-8.5) | |||||
| Other2 | 1/72 | 6/6.0 | 5/89 | 5/89 | 5.0/7.0 |
| 1.4% (0.1-8.5) | |||||
| Any type of evolution with development of alert findings | 6/72 | 8/1.3 | 34.5 ± 19.9/42.1 ± 19.4 | 34 (15-63)/45.5 (13-71) | 11.3 ± 6.4/15.8 ± 6.1 |
| 8.3% (3.4-17.9) | |||||
| Total | 32/72 | 47/1.4 | 28.1 ± 22.1/39.4 ± 26.9 | 22.5 (4-79)/32 (4-92) | 9.7 ± 5.7/12.5 ± 7.3 |
| 44.4% (32.9-56.6) |
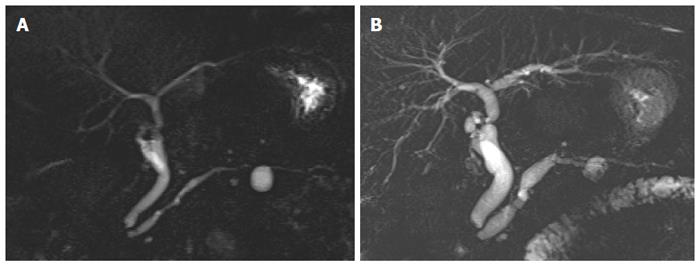
Alert findings developed in 6/72 patients (8.3%; 95%CI: 3.4-17.9) as detailed in Table 3 (Figure 2). Except for one case, the occurrence of alert findings was preceded by other imaging evolution.
| Patient/sex/age on baseline MRCP | Type and timing of alert finding occurrence | Type and timing of associated imaging evolution | Final diagnosis | Standard of reference | Notes |
| 1/female/58 yr | Non-enhancing small filling defect at 48 mo in an 8 mm cyst, increased from previous control | The same cyst showed initial increase in size (from 5 mm to 8 mm) at 12 mo, followed by decrease (from 8 mm to 5 mm) at 18 mo | Mucin plug; no dysplasia or invasive cancer | EUS with FNA: mucin plug, negative cytology | - |
| 2/male/59 yr | Thickened/nodular septa at 15 mo | Minimal increase of two cysts at 6 mo | No dysplasia or invasive cancer | EUS: negative examination | No further evolution over the whole follow-up (24 mo). Patient died for alcoholic liver cirrhosis |
| 3/female/68 yr | Non-enhancing filling defect at 43 mo | Disappearance of a 9 mm cyst at 33 mo | Mucin plug; no dysplasia or invasive cancer | EUS with FNA: negative cytology | - |
| 4/female/55 yr | Thickened/nodular septa at 63 and then 71 mo | Increase in size of the same cyst from 10 mm to 15 mm at 9 mo | Low-grade dysplasia bd-IPMN | EUS with FNA: low grade dysplasia bd-IPMN, negative CEA and CA 19.9 | Non further evolution over the whole follow-up (99 mo) |
| 5/female/60 yr | Dilation of the main pancreatic duct (head, caliber 9 mm) and mural filling defects in the larger cyst (15 mm) at 25 mo | No other evolution observed | Evolution from bd-IPMN to mixed-type IPMN | EUS with FNA (repeated 3 times): negative cytology, elevated CEA, CA 19.9 and amylase. No vascularization of filling defects on Color Doppler exam | Liver-transplanted patient (alcoholic cirrhosis) in whom pancreatic surgery was unfeasible. No further evolution over the whole follow up period (92 mo) |
| 6/female/72 yr | Increase in size of one cyst from 20 mm to 30 mm at 13 mo | Increase in size of the same cyst from 16 mm to 20 mm at 9 mo | No dysplasia or invasive cancer | Histological examination after surgical resection of the cyst | Occurrence of the alert finding preceded by negative EUS |
| No further evolution over the subsequent follow-up (39 mo) |
No clinical or baseline MRCP features were predictive of imaging evolution or alert findings development (P > 0.01) (Table 4). The only trend to statistical significance was observed for baseline cysts size > 10 mm as a predictor of imaging evolution.
| Clinical or imaging feature | Imaging evolution | Alert findings | ||||
| HR | P value | 95%CI | HR | P value | 95%CI | |
| Sex | 0.78 | 0.569 | 0.33-1.81 | 0.60 | 0.644 | 0.69-5.19 |
| Age | 0.98 | 0.488 | 0.95-1.02 | 1.00 | 0.967 | 0.92-1.08 |
| Previous cholecistectomy | 0.89 | 0.794 | 0.40-2.00 | -1 | ||
| Diabetes mellitus (type 2) | 0.92 | 0.873 | 0.34-2.44 | 2.39 | 0.316 | 0.43-13.14 |
| Autoimmune systemic disease | 0.58 | 0.218 | 0.25-1.36 | 0.51 | 0.543 | 0.59-4.39 |
| Alcoholic liver cirrhosis | 1.88 | 0.143 | 0.80-4.40 | 2.49 | 0.295 | 0.45-13.75 |
| History of extrapancreatic neoplasia | 1.79 | 0.109 | 0.87-3.68 | 0.70 | 0.694 | 0.12-3.93 |
| Cysts size > 10 mm on baseline MRCP | 2.13 | 0.055 | 0.98-4.64 | 4.02 | 0.205 | 0.46-34.70 |
| Number of cysts on baseline MRCP | 1.42 | 0.590 | 0.70-1.96 | 1.26 | 0.680 | 0.40-3.95 |
The proportion of patients without imaging evolution estimated at 12, 24 and 60 mo was 86.1% (95%CI: 75.7-92.2), 73.5% (95%CI: 61.6-82.2) and 52.9% (95%CI: 37.2-66.4), respectively; the proportion of patients without alert findings estimated at 12, 24 and 60 mo was 100%, 97.2% (95%CI: 89.3-99.3) and 91.0% (95%CI: 79.2-96.2), respectively.
MRCP has been advocated as the procedure of choice for evaluating bd-IPMN, given the capability of assessing subtle findings such as smaller cysts, communication with the MPD, septa and mural nodules[10]. MRCP is accurate in diagnosing presumed bd-IPMN at initial presentation and in detecting changes occurring over the follow-up[3]. However, most studies in literature evaluated cysts evolution by alternating or replacing MRCP with other imaging modalities, including CT, EUS and endoscopic retrograde cholangiopancreatography[3,8,11,12]. This suggest that not all morphological changes over time might have been detected, thus limiting information on natural history. Not surprisingly, the per-patient rate of imaging evolution we observed was higher (44.4%) compared to most clinical studies (5.6%-28.0%)[13-18], though initial size of all cysts (average 8.5 ± 1.7 mm) and the magnitude of changes in patients with evolution were comparably small (average size on baseline MRCP and at the end of the follow-up of 9.7 mm vs 12.5 mm, respectively, with changes involving 1.4 cysts per-patient). It should be pointed out that previous MRCP-based studies[19,20] found a lower imaging evolution rate (5%-10.8%) than ours in comparable patients populations. This discrepancy can be explained by our larger study population and/or by the fact we performed a cyst-by-cyst analysis rather than focusing on target ones, as supported by similar evolution rate (48.5% increase in size, 1.5% decrease in size) in a clinical study by Bae et al[11] with same type of analysis.
Imaging evolution included development of alert findings in 8.3% of patients, a value within the range of those previously reported in studies with MRCP (2.7%-10.8%)[19-22] or combined imaging modalities (4.9%-9%)[14,17]. None of them corresponded to “high-risk stigmata” according to Sendai guidelines definition, but rather to worrisome features[1]. Except for one patient who developed mixed-type IPMN, small changes preceded alert findings at a variable, unpredictable extent of time, involving both evolved cysts or not. Four out of 6 patients with alert findings showed no abnormalities on EUS with FNA (3 cases) and histological examination after surgery (1 case). Remaining EUS-based diagnoses were low-dysplasia in one patient refusing surgery and evolution to mixed-type IPMN in a liver-transplanted patient in whom surgery was unfeasible because of poor health status. Both showed lesion stability during the follow-up (92 and 99 mo, respectively). Similarly to previous MRCP-based studies on smaller populations[19,20], we observed no evolution to overt malignancy.
Our results suggest that small changes in the appearance of incidental bd-IPMN are common when the follow-up is performed with MRCP, likely reflecting a dynamic process of filling/empting of cystic mucin content and/or plug formation. In many cases, changes occurred repeatedly in different MRCP examinations, with a trend to progressive, small and slow progression that is in line with previous observations[21]. In particular, the median time of first and last occurrence was 22.5 and 32 mo, respectively, with about one-half of patients having manifested evolution at 60 mo, suggesting that imaging evolution can occur whenever during the follow-up. Furthermore, no clinical factors or cysts appearance on baseline MRCP were predictors of imaging evolution, with the partial, but not significant (P = 0.055) exception of size > 10 mm.
One can hypothesize some clinical consequences for the management of incidental, presumed bd-IPMN. First, imaging evolution on MRCP should not prompt interventions, additional diagnostic steps or change in scheduled follow-up unless alert findings occur. Second, since there was no definite proof for malignancy in most patients who develop alert findings, careful risk-to-benefit evaluation should be made before addressing them to invasive and costing diagnosis or treatment. In the case of subtle and ambiguous findings, interventions might be deferred, using MRCP to follow-up patients at a shorter time (e.g., every six mo from the alert findings for the first year) unless further evolution or stability are demonstrated. Crippa et al[23] have recently shown that conservative management in patients developing worrisome features is appropriate, with 5-year disease-survival of 96%. Our results showed that alert findings occurrence was quite variable in time (range 13-63 mo). More importantly, alert findings were unpredictable based on cysts pattern or clinical features. Given the low rate of occurrence (91% of alert findings-free patients at 60 mo) and risk they really correspond to malignancy (or rapidly evolving malignancy), one might hypothesize that follow-up should continue over time, possibly with longer intervals between examinations to avoid “oversuveillance”, e.g., every 2 years as previously suggested[24].
Our study shows some limitations. First, there was no histological confirmation of bd-IPMN in our series, as well as the majority of alert findings (5/6), though EUS with FNA was performed in those cases. However, our results reflects clinical practice, in which: (1) MRCP is considered the most reliable noninvasive tool to diagnose and follow-up presumed bd-IPMN[3]; and (2) final diagnosis cannot be frequently obtained in patients with alert findings because of age and comorbidities contraindicating surgery, mirroring the “incidental” setting. It would be unfeasible/unethical to address patients to unnecessary invasive procedures. On the other hand, none of the alert findings showed evolution to overt malignancy during the follow-up. Second, number and timing of follow-up examinations were inhomogeneous, given the retrospective design. Further studies with a prospective and more homogeneous follow-up scheduling may support our findings.
In conclusion, incidentally found cysts arranged in a bd-IPMN pattern showed 44.4% evolution rate when monitored with MRCP, mainly in terms of minimal increase in size. The rate of occurrence of alert findings prompting further diagnostic step was 8.3%, with no definite proof of malignancy nor significant impact on patients’ management. Time to evolution and/or alert findings was quite variable, whereas no clinical or baseline MRCP findings were found to predict both types of changes. Our results suggest that imaging follow-up of incidental bd-IPMN should not be discontinued, though examinations might be considerably delayed with a minimal risk of missing malignancy. Further studies should clarify whether rescheduling the interval of time between follow-up examinations is an effective strategy in managing patients who develop alert findings.
The Authors thank Dr. Iliana Bednarova from the Institute of Radiology, University of Udine, for her help in revising English language.
Branch-duct intraductal papillary mucinous neoplasm (bd-IPMN) is a frequent incidental finding in magnetic resonance cholangiography (MRCP) examinations performed for indications unrelated to the pancreas. To our knowledge, little is known on natural history of incidental bd-IPMN, and whether imaging follow-up should be prolonged in order not to miss rare but concrete cases harboring malignancy.
This research was clinical in nature. Together with other results, it might contribute to refine current guidelines on the management of IPMN. Moreover, our study provides a detailed description of how cysts evolve during follow-up.
Changes in MRCP appearance of incidental bd-IPNM are frequent and unpredictable over the follow-up, though with relatively rare (8.3%) occurrence of alert findings prompting further diagnostic steps (none of them was found malignant). The results suggest that imaging follow-up should be prolonged over time, though MRCPs might be considerably delayed without a significant risk of missing malignancy.
This study shows that MRCP can be safely used as a leading tool to monitor incidental bd-IPMN over time, in order to exclude the development of suspicious morphological findings prompting further diagnostic management.
MRCP: A magnetic resonance imaging-based technique which permits panoramic and elective representation of the biliary tract and/or pancreatic ductal system. bd-IPMN: A variant of IPMN that can be monitored over time in the absence of high risk stigmata or worrisome features easily diagnosed with MRCP.
The authors describe high rate of imaging evolution and developing 8.3% of alert findings during follow-up IPMN.
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: Italy
Peer-review report classification
Grade A (Excellent): A
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Kimura K, Peng SY, Souftas VD S- Editor: Yu J L- Editor: A E- Editor: Wang CH
| 1. | Tanaka M, Fernández-del Castillo C, Adsay V, Chari S, Falconi M, Jang JY, Kimura W, Levy P, Pitman MB, Schmidt CM. International consensus guidelines 2012 for the management of IPMN and MCN of the pancreas. Pancreatology. 2012;12:183-197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1714] [Cited by in RCA: 1613] [Article Influence: 124.1] [Reference Citation Analysis (0)] |
| 2. | Khan S, Sclabas G, Reid-Lombardo KM. Population-based epidemiology, risk factors and screening of intraductal papillary mucinous neoplasm patients. World J Gastrointest Surg. 2010;2:314-318. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 35] [Cited by in RCA: 34] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 3. | Marchegiani G, Fernández-del Castillo C. Is it safe to follow side branch IPMNs? Adv Surg. 2014;48:13-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 12] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 4. | Laffan TA, Horton KM, Klein AP, Berlanstein B, Siegelman SS, Kawamoto S, Johnson PT, Fishman EK, Hruban RH. Prevalence of unsuspected pancreatic cysts on MDCT. AJR Am J Roentgenol. 2008;191:802-807. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 724] [Cited by in RCA: 656] [Article Influence: 38.6] [Reference Citation Analysis (0)] |
| 5. | Girometti R, Intini S, Brondani G, Como G, Londero F, Bresadola F, Zuiani C, Bazzocchi M. Incidental pancreatic cysts on 3D turbo spin echo magnetic resonance cholangiopancreatography: prevalence and relation with clinical and imaging features. Abdom Imaging. 2011;36:196-205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 92] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 6. | Del Chiaro M, Verbeke C, Salvia R, Klöppel G, Werner J, McKay C, Friess H, Manfredi R, Van Cutsem E, Löhr M. European experts consensus statement on cystic tumours of the pancreas. Dig Liver Dis. 2013;45:703-711. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 324] [Cited by in RCA: 333] [Article Influence: 27.8] [Reference Citation Analysis (0)] |
| 7. | Sahani DV, Kambadakone A, Macari M, Takahashi N, Chari S, Fernandez-del Castillo C. Diagnosis and management of cystic pancreatic lesions. AJR Am J Roentgenol. 2013;200:343-354. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 88] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 8. | Scheiman JM, Hwang JH, Moayyedi P. American gastroenterological association technical review on the diagnosis and management of asymptomatic neoplastic pancreatic cysts. Gastroenterology. 2015;148:824-48.e22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 267] [Cited by in RCA: 282] [Article Influence: 28.2] [Reference Citation Analysis (0)] |
| 9. | Dunn DP, Brook OR, Brook A, Revah G, Jawadi S, Sun M, Lee KS, Mortele KJ. Measurement of pancreatic cystic lesions on magnetic resonance imaging: efficacy of standards in reducing inter-observer variability. Abdom Radiol (NY). 2016;41:500-507. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 24] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 10. | Berland LL, Silverman SG, Gore RM, Mayo-Smith WW, Megibow AJ, Yee J, Brink JA, Baker ME, Federle MP, Foley WD. Managing incidental findings on abdominal CT: white paper of the ACR incidental findings committee. J Am Coll Radiol. 2010;7:754-773. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 624] [Cited by in RCA: 512] [Article Influence: 34.1] [Reference Citation Analysis (0)] |
| 11. | Bae SY, Lee KT, Lee JH, Lee JK, Lee KH, Rhee JC. Proper management and follow-up strategy of branch duct intraductal papillary mucinous neoplasms of the pancreas. Dig Liver Dis. 2012;44:257-260. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 12. | Sahora K, Mino-Kenudson M, Brugge W, Thayer SP, Ferrone CR, Sahani D, Pitman MB, Warshaw AL, Lillemoe KD, Fernandez-del Castillo CF. Branch duct intraductal papillary mucinous neoplasms: does cyst size change the tip of the scale? A critical analysis of the revised international consensus guidelines in a large single-institutional series. Ann Surg. 2013;258:466-475. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 207] [Cited by in RCA: 224] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 13. | Salvia R, Crippa S, Falconi M, Bassi C, Guarise A, Scarpa A, Pederzoli P. Branch-duct intraductal papillary mucinous neoplasms of the pancreas: to operate or not to operate? Gut. 2007;56:1086-1090. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 201] [Cited by in RCA: 197] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 14. | Tanno S, Nakano Y, Nishikawa T, Nakamura K, Sasajima J, Minoguchi M, Mizukami Y, Yanagawa N, Fujii T, Obara T. Natural history of branch duct intraductal papillary-mucinous neoplasms of the pancreas without mural nodules: long-term follow-up results. Gut. 2008;57:339-343. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 190] [Cited by in RCA: 174] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 15. | Maguchi H, Tanno S, Mizuno N, Hanada K, Kobayashi G, Hatori T, Sadakari Y, Yamaguchi T, Tobita K, Doi R. Natural history of branch duct intraductal papillary mucinous neoplasms of the pancreas: a multicenter study in Japan. Pancreas. 2011;40:364-370. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 186] [Cited by in RCA: 169] [Article Influence: 12.1] [Reference Citation Analysis (0)] |
| 16. | Arlix A, Bournet B, Otal P, Canevet G, Thevenot A, Kirzin S, Carrere N, Suc B, Moreau J, Escourrou J. Long-term clinical and imaging follow-up of nonoperated branch duct form of intraductal papillary mucinous neoplasms of the pancreas. Pancreas. 2012;41:295-301. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 44] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 17. | Khannoussi W, Vullierme MP, Rebours V, Maire F, Hentic O, Aubert A, Sauvanet A, Dokmak S, Couvelard A, Hammel P. The long term risk of malignancy in patients with branch duct intraductal papillary mucinous neoplasms of the pancreas. Pancreatology. 2012;12:198-202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 79] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 18. | Shin SH, Han DJ, Park KT, Kim YH, Park JB, Kim SC. Validating a simple scoring system to predict malignancy and invasiveness of intraductal papillary mucinous neoplasms of the pancreas. World J Surg. 2010;34:776-783. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 51] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 19. | Guarise A, Faccioli N, Ferrari M, Salvia R, Mucelli RP, Morana G, Megibow AJ. Evaluation of serial changes of pancreatic branch duct intraductal papillary mucinous neoplasms by follow-up with magnetic resonance imaging. Cancer Imaging. 2008;8:220-228. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 20] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 20. | Carbognin G, Zamboni G, Pinali L, Chiara ED, Girardi V, Salvia R, Mucelli RP. Branch duct IPMTs: value of cross-sectional imaging in the assessment of biological behavior and follow-up. Abdom Imaging. 2006;31:320-325. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 39] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 21. | Manfredi R, Mehrabi S, Motton M, Graziani R, Ferrari M, Salvia R, Pozzi Mucelli R. MR imaging and MR cholangiopancreatography of multifocal intraductal papillary mucinous neoplasms of the side branches: MR pattern and its evolution. Radiol Med. 2008;113:414-428. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 24] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 22. | Castelli F, Bosetti D, Negrelli R, Di Paola V, Zantedeschi L, Ventriglia A, Manfredi R, Mucelli RP. Multifocal branch-duct intraductal papillary mucinous neoplasms (IPMNs) of the pancreas: magnetic resonance (MR) imaging pattern and evolution over time. Radiol Med. 2013;118:917-929. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 14] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 23. | Crippa S, Bassi C, Salvia R, Malleo G, Marchegiani G, Rebours V, Levy P, Partelli S, Suleiman SL, Banks PA. Low progression of intraductal papillary mucinous neoplasms with worrisome features and high-risk stigmata undergoing non-operative management: a mid-term follow-up analysis. Gut. 2016; Epub ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 151] [Article Influence: 18.9] [Reference Citation Analysis (0)] |
| 24. | Das A, Wells CD, Nguyen CC. Incidental cystic neoplasms of pancreas: what is the optimal interval of imaging surveillance? Am J Gastroenterol. 2008;103:1657-1662. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |