Published online Nov 21, 2015. doi: 10.3748/wjg.v21.i43.12505
Peer-review started: May 11, 2015
First decision: July 10, 2015
Revised: July 20, 2015
Accepted: September 14, 2015
Article in press: September 15, 2015
Published online: November 21, 2015
Processing time: 196 Days and 4.6 Hours
Human cytomegalovirus (CMV) is a herpesvirus, which establishes lifelong latency after primary infection and leads to severe disease in immunocompromised patients. However, CMV infection in immunocompetent patients is usually asymptomatic and severe organ damage is rarely reported. We report a case of severe CMV hepatitis in an immunocompetent patient presenting with cholestasis, portal hypertension-related ascites and pancytopenia. The patient was asymptomatic with normal liver function and negative CMV DNA after two weeks of antiviral therapy. This case is an example of a common infection with an uncommon presentation, and suggests that testing for CMV should be carried out, even in patients with normal immune status, presenting with severe liver damage or cholestasis.
Core tip: Human cytomegalovirus (CMV) can establish lifelong latency after primary infection. It also leads to severe disease in immunocompromised patients. However, CMV infection in immunocompetent patients is usually asymptomatic and severe organ damage is rarely reported. Here, we report an immunocompetent patient with a unique presentation of severe CMV hepatitis manifested by cholestasis, ascites and pancytopenia. The patient was asymptomatic with normal liver function and negative CMV DNA after two weeks of antiviral therapy. It is suggested that testing for CMV should be carried out, even in patients with normal immune status, presenting with severe liver damage or cholestasis.
- Citation: Qian JY, Bai XY, Feng YL, Zhu WJ, Yao F, Li JN, Yang AM, Li F, Qian JM. Cholestasis, ascites and pancytopenia in an immunocompetent adult with severe cytomegalovirus hepatitis. World J Gastroenterol 2015; 21(43): 12505-12509
- URL: https://www.wjgnet.com/1007-9327/full/v21/i43/12505.htm
- DOI: https://dx.doi.org/10.3748/wjg.v21.i43.12505
Human cytomegalovirus (CMV), a herpesvirus, is a double-stranded DNA virus which can establish lifelong latency after primary infection. The infection can be transmitted by body fluid contact, placental transfer and transplantation[1]. The primary CMV infection may arise at an early age and the seroprevalence in developed countries is reported to be 30%-70%[2]. In immunocompetent patients, primary and long-term immunity are induced after the initial infection, preventing reactivation of the virus; while in immunocompromised patients, such as transplant recipients and patients with acquired immune deficiency syndrome, viral replication cannot be controlled and therefore can cause severe diseases. This explains the significant morbidity and mortality in CMV infected immunocompromised patients[3,4]. CMV infection in immunocompetent hosts is commonly asymptomatic, or in some cases, presents as mononucleosis syndrome. Severe organ involvement after CMV infection in immunocompetent adults is rare[5]. We report a unique case of severe CMV hepatitis in an immunocompetent patient presenting with cholestasis, portal hypertension-related ascites and pancytopenia.
A previously healthy 66-year-old female presented with a one-month history of yellow coloration of the eyes and skin, abdominal distension, dark colored urine and pale stool. Fever, abdominal pain, nausea, vomiting and diarrhea were absent. The patient denied alcohol consumption, toxic exposure and a medication history of glucocorticoids, immunosuppressive agents or herbal medicine. No significant history of traveling and animal contact or similar cases in the community were reported. Moderate mucocutaneous jaundice and shifting dullness were revealed on physical examination which was otherwise unremarkable.
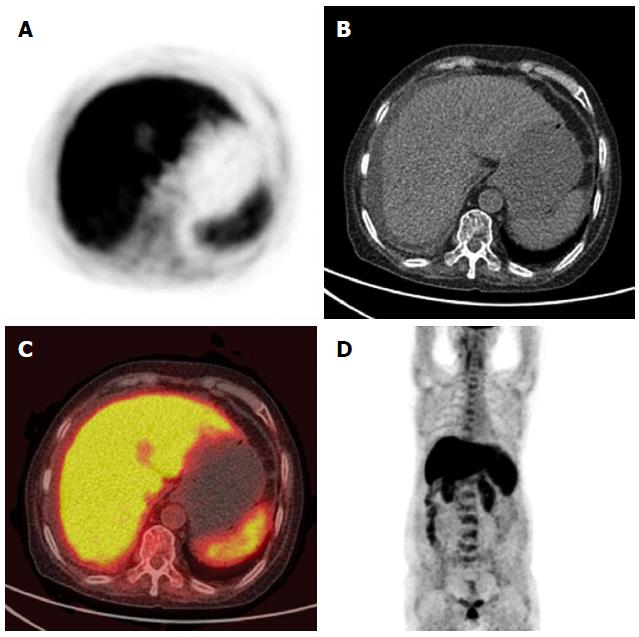
Complete blood count (CBC) showed progressive pancytopenia (hemoglobin of 113→95 g/L, platelet count of 154→54 × 109/L and white blood cell count of 6.74→4.48 × 109/L), and bone marrow biopsy revealed infrequent hemopoietic precursors and increased adipose tissue. Liver function tests were abnormal with alanine aminotransferase 164 U/L (normal value 7-40 U/L), aspartate aminotransferase 191 U/L (normal value 13-35 U/L), gamma glutamyl transpeptidase 83 U/L (normal value 7-45 U/L), alkaline phosphatase 420 U/L (normal value 50-135 U/L), total bilirubin 179.5 μmol/L (normal value 5.1-22.2 μmol/L) and direct bilirubin 145.6 μmol/L (normal value 0-6.8 μmol/L). Coagulation tests showed elevated prothrombin time and partial thromboplastin time of 15.7 s (normal value 10.4-12.6 s) and 65.5 s (normal value 22.7-31.8 s), respectively. Acute phase reactants and immunoglobulin A/G/M were normal. Autoantibodies including antinuclear antibodies, antimitochondrial antibodies, anti-smooth muscle antibodies, anti-liver-kidney microsome-1 antibodies, and anti-liver cytosol antibody-1 indicated that autoimmune hepatitis and primary biliary cirrhosis were negative. Abdominal paracentesis was performed and the results suggested portal hypertension-related ascites which was confirmed by a serum-ascites albumin gradient of 26 g/L. No malignant cells were found in the ascites. Contrast-enhanced computed tomography (CT) revealed homogeneous low density of the liver and slight dilatation of the intrahepatic bile ducts. No dilatation of the extrahepatic bile duct, cirrhosis or splenomegaly was found. Positron emission tomography (PET)/CT showed diffusely increased fluorodeoxyglucose uptake in the liver (Figure 1). Three-dimensional CT angiography revealed no evidence of portal vein thrombosis. A lymphocyte subset test showed a CD4+ cell to CD8+ cell ratio of 1.2 (normal 0.95-2.13). Activation of functional CD8+ T cells was shown by CD8+HLA-DR+/CD8+% of 60.2% (normal 6.3%-23.8%) and CD8+CD38+/CD8+% of 90.2% (normal 32.4%-57.4%). Tests for hepatitis A, hepatitis B, hepatitis C, hepatitis E, Epstein-Barr virus and human herpes virus were negative, however, quantitative PCR for CMV DNA showed 7800 copies/mL (normal < 500 copies/mL). Liver biopsy was not performed due to coagulopathy.
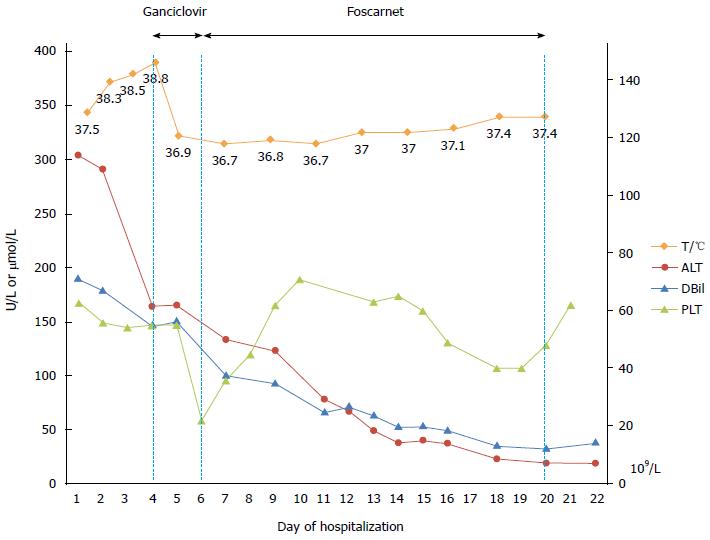
Following admission, the patient developed recurrent episodes of fever (Tmax of 38.7 °C) without other associated symptoms. CMV hepatitis was considered to be the most likely diagnosis in this case, although liver biopsy could not be carried out, given the fact that other causes of liver damage including alcohol, drug, toxicant, tumor, and other common causes of viral hepatitis had been reasonably excluded. Anti-CMV treatment with intravenous ganciclovir was commenced due to progressive deterioration of her conditions, especially pancytopenia and coagulopathy. Her body temperature normalized within a day indicating a positive response to the antiviral therapy, however, the platelet count progressively decreased to 22 × 109/L. As thrombocytopenia may have been an adverse effect of ganciclovir, intravenous foscarnet was started instead. Jaundice, ascites, liver function tests and CBC gradually improved (Figure 2). After two weeks of antiviral therapy, the patient became asymptomatic and discharged with normal liver function and negative CMV DNA.
Single or multi-organ disorders as complications of CMV infection in immunocompetent hosts are rarely reported. Of these complications, gastrointestinal manifestations are thought to be the most common. Other complications include hepatic, neurological, pulmonary, ocular, and cardiovascular. Asymptomatic elevation of transaminases is the most common hepatic manifestation in immunocompetent patients, while hepatic dysfunction with significant symptoms and signs is extremely rare[5]. In the present report, an immunocompetent adult presenting with features consistent with portal hypertension and pancytopenia was found to have a significantly increased CMV DNA level. The diagnosis of severe CMV hepatitis was made after other possible causes were excluded, although liver biopsy was not performed due to coagulopathy. Fever and liver function improved with initial ganciclovir treatment, however, a decrease in the platelet count was observed. We considered the worsening of thrombocytopenia to be a side effect of ganciclovir in this case as other symptoms associated with CMV primary infection improved. It has been reported that cancer chemotherapy, decreased creatinine clearance (< 20 mL/min) and high ganciclovir dose (≥ 12 mg/kg daily) were independent risk factors for thrombocytopenia in CMV patients receiving ganciclovir[6]. Two weeks after switching from ganciclovir to foscarnet, the patient’s liver function was back to normal. This case is an example of a common infection with an uncommon presentation, and suggests that testing for CMV is necessary in patients presenting with severe liver damage, even if they are immunocompetent. The decision to perform a liver biopsy should be based on the condition of the patient, even though it is essential to achieve a definitive diagnosis. Antiviral therapy is beneficial in severe and life-threatening conditions, but should be administered after weighing the benefits and risks.
The authors thank Yanyan Wu, M.D. from Launceston General Hospital, Tasmania, Australia for her kind help in language editing.
A 66-year-old female presented with a one-month history of yellow coloration of the eyes and skin, abdominal distension, dark colored urine and pale stool.
Physical examination showed moderate mucocutaneous jaundice and shifting dullness.
Virus infection [hepatitis A, hepatitis B, hepatitis C, hepatitis E, Epstein-Barr virus, cytomegalovirus (CMV) and human herpes virus], autoimmune hepatitis, primary biliary cirrhosis and periampullary carcinoma.
Laboratory tests revealed severe liver injury, pancytopenia, coagulopathy, and quantitative PCR showed CMV DNA of 7800 copies/mL.
Positron emission tomography-computed tomography showed diffusely increased fluorodeoxyglucose uptake in the liver.
As thrombocytopenia may be an adverse effect of ganciclovir, treatment was switched to intravenous foscarnet.
Single or multi-organ disorders as complications of CMV infection in immunocompetent hosts are rarely reported.
This case is an example of a common infection with an uncommon presentation, and suggests that testing for CMV should be carried out, even in patients with normal immune status who have severe liver damage or cholestasis.
The authors presented the common infection of CMV in an immunocompetent patient with a unique presentation of severe cytomegalovirus hepatitis manifested with cholestasis, ascites and pancytopenia.
P- Reviewer: ElBaz AA, Kanda T S- Editor: Ma YJ L- Editor: A E- Editor: Liu XM
| 1. | Gandhi MK, Khanna R. Human cytomegalovirus: clinical aspects, immune regulation, and emerging treatments. Lancet Infect Dis. 2004;4:725-738. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 384] [Cited by in RCA: 399] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 2. | Pass RF. Epidemiology and transmission of cytomegalovirus. J Infect Dis. 1985;152:243-248. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 107] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 3. | Gallant JE, Moore RD, Richman DD, Keruly J, Chaisson RE. Incidence and natural history of cytomegalovirus disease in patients with advanced human immunodeficiency virus disease treated with zidovudine. The Zidovudine Epidemiology Study Group. J Infect Dis. 1992;166:1223-1227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 369] [Cited by in RCA: 358] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 4. | Brennan DC. Cytomegalovirus in renal transplantation. J Am Soc Nephrol. 2001;12:848-855. [PubMed] |
| 5. | Rafailidis PI, Mourtzoukou EG, Varbobitis IC, Falagas ME. Severe cytomegalovirus infection in apparently immunocompetent patients: a systematic review. Virol J. 2008;5:47. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 332] [Cited by in RCA: 374] [Article Influence: 22.0] [Reference Citation Analysis (0)] |
| 6. | Matsumoto K, Shigemi A, Ikawa K, Kanazawa N, Fujisaki Y, Morikawa N, Takeda Y. Risk factors for ganciclovir-induced thrombocytopenia and leukopenia. Biol Pharm Bull. 2015;38:235-238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 1.8] [Reference Citation Analysis (0)] |