Published online Nov 14, 2015. doi: 10.3748/wjg.v21.i42.12059
Peer-review started: June 6, 2015
First decision: July 13, 2015
Revised: July 28, 2015
Accepted: September 14, 2015
Article in press: September 14, 2015
Published online: November 14, 2015
Processing time: 160 Days and 19.7 Hours
Sorafenib is the only and standard systematic chemotherapy drug for treatment of advanced hepatocellular carcinoma (HCC) at the current stage. Although sorafenib showed survival benefits in large randomized phase III studies, its clinical benefits remain modest and most often consist of temporary tumor stabilization, indicating that more effective first-line treatment regimens or second-line salvage therapies are required. The molecular pathogenesis of HCC is very complex, involving hyperactivated signal transduction pathways such as RAS/RAF/MEK/ERK and PI3K/AKT/mTOR and aberrant expression of molecules such as receptor tyrosine kinases and histone deacetylases. Simultaneous or sequential abrogation of these critical pathways or the functions of these key molecules involved in angiogenesis, proliferation, and apoptosis may yield major improvements in the management of HCC. In this review, we summarize the emerging sorafenib-based combined molecule targeting for HCC treatment and analyze the rationales of these combinations.
Core tip: Cancer is regarded as a heterogeneous disease, with no exception of hepatocellular carcinoma (HCC), which requires combined chemotherapy. HCC is not sensitive to most currently used conventional cytotoxic drugs. The approval of sorafenib, a molecular targeted drug that inhibits RAF kinase and several other angiogenesis-related receptor tyrosine kinases, opens a door for systematic treatment of HCC. The pathogenesis of HCC involves hyperactivation of several signal pathways and aberrant expression of some key molecules, suggesting combination treatment may yield major improvements in the management of this disease. The emerging sorafenib-based combination treatments are reviewed in the present article.
- Citation: Gao JJ, Shi ZY, Xia JF, Inagaki Y, Tang W. Sorafenib-based combined molecule targeting in treatment of hepatocellular carcinoma. World J Gastroenterol 2015; 21(42): 12059-12070
- URL: https://www.wjgnet.com/1007-9327/full/v21/i42/12059.htm
- DOI: https://dx.doi.org/10.3748/wjg.v21.i42.12059
Liver cancer is the second most common cause of death from cancer worldwide, estimated to be responsible for nearly 746000 deaths in 2012 according to the statistics published by World Health Organization[1]. Among the diverse, histologically distinct primary hepatic neoplasms, hepatocellular carcinoma (HCC) is the most common type of liver cancer, accounting for 83% of all cases[2-4]. The prevalence of HCC is especially severe in China and Japan due to high rates of hepatitis B virus (HBV) and hepatitis C virus (HCV) infection in the population[5-8]. Curative treatments including liver transplantation and hepatic resection are only suitable for fewer than 20% of HCC patients because most cases have progressed to an advanced stage with intra- or extra-hepatic metastasis when the disease is diagnosed[9-13]. As a palliative treatment, chemotherapy is a highly needed means for the patients with unresectable or metastatic HCC[14-17].
HCC is generally recognized as a chemo-resistant tumor that is not sensitive to most currently used cytotoxic drugs[18-20]. Recent discoveries in the molecular mechanisms of HCC pathogenesis have created many opportunities for developing targeted therapies[21-24]. Sorafenib, which targets RAF kinase (c-RAF and b-RAF) and several other angiogenesis-related receptor tyrosine kinases (RTKs) including vascular endothelial growth factor receptor (VEGFR) and platelet-derived growth factor receptor (PDGFR), is the first approved molecular targeted agent that has been applied to systemic chemotherapy in HCC patients with metastatic disease or transcatheter arterial chemoembolization (TACE)-refractory disease[25,26]. Although clinical studies demonstrated some benefits of sorafenib on the time to progression (TTP) and overall survival (OS), its efficacy against HCC remains moderate[25]. The median TTP was demonstrated to be 5.5 mo for sorafenib and 2.8 mo for placebo, and the median OS was 10.7 mo for sorafenib and 7.9 mo for placebo[25]. Results of subsequent clinical trials on single agents including linifanib, brivanib, and sunitinib are disappointing since they are not superior to sorafenib or placebo in terms of efficacy or safety[27]. At this stage, there have not yet been any verified drugs except for sorafenib in systematic treatment of HCC.
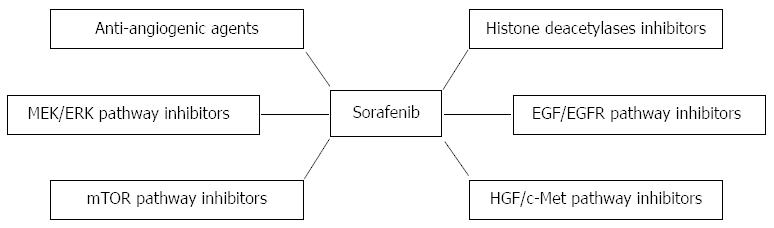
Combination of sorafenib and other molecular targeted drugs for treatment of HCC has drawn wide attention in recent years based on the following understanding: (1) HCC is a highly heterogeneous disease in terms of etiology and clinical behavior, which signifies that cancer-driven genes may be varied in tumors of different patients[28]; (2) Cell clones in one tumor are heterogeneous with respect to both morphology and function, which means that the tumor is made up of diverse cell populations that show different drug sensitivity[29,30]. The drug-resistant clones may survive under the evolutionary selection pressure and then drive the cancer progression; and (3) Hepatocarcinogenesis is a stepwise process during which multiple genes are altered[31]. Genetic changes and their biological consequences in cancerous cells may include tumor suppressor genes, oncogenes, reactivation of developmental pathways, growth factors and their receptors, and angiogenesis[32]. The complexity of pathogenesis of HCC suggests that combination of drugs that target multiple key molecules implicated in tumor initiation and progression is a promising strategy to conquer this pressing disease. In this review, we summarizes recent advances of sorafenib-based combined treatments for HCC (Figure 1 and Table 1).
| Combined drugs | Treatment modality | Current stage | Outcome | Ref. |
| Antiangiogenic agents | ||||
| Dalantercept | Simultaneous combination with sorafenib | Phase Ib (ongoing) | Awaited | NCT02024087 |
| Axitinib | Sequential treatment after sorafenib failing | Phase II (finished) | Warranting further clinical study | [49] |
| Sequential treatment after sorafenib failing | Phase II (ongoing) | Awaited | EUCTR2011-002029-24-IT | |
| Apatinib | Sequential treatment after sorafenib failing | Phase III (ongoing) | Awaited | NCT02329860 |
| MEK/ERK inhibitors | ||||
| AZD6244 | Simultaneous combination with sorafenib | Phase II (terminated) | Unknown | NCT01029418 |
| Refametinib | Simultaneous combination with sorafenib | Phase II (finished) | Warranting further clinical study | [64] |
| mTOR inhibitors | ||||
| Sirolimus | Simultaneous combination with sorafenib | Preclinical study | Synergistic effect | [73] |
| Everolimus | Simultaneous combination with sorafenib | Phase II (ongoing) | Awaited | NCT01005199 |
| Temsirolimus | Simultaneous combination with sorafenib | Phase II (ongoing) | Awaited | NCT01687673 |
| PI-103 | Simultaneous combination with sorafenib | Preclinical study | Synergistic effect | [86] |
| PKI-587 | Simultaneous combination with sorafenib | Preclinical study | Synergistic effect | [87] |
| HDACs inhibitors | ||||
| Panobinostat | Simultaneous combination with sorafenib | Phase I (terminated) | Precluding phase II studies | NCT00873002 |
| Vorinostat | Simultaneous combination with sorafenib | Phase I (suspended) | Unknown | NCT01075113 |
| Resminostat | Simultaneous combination with sorafenib | Phase II (finished) | Warranting further clinical study | [100] |
| Simultaneous combination with sorafenib | Phase I/II (ongoing) | Awaited | NCT02400788 | |
| MPT0E028 | Simultaneous combination with sorafenib | Preclinical study | Synergistic effects | [97] |
| EGFR inhibitors | ||||
| Erlotinib | Simultaneous combination with sorafenib | Phase III (finished) | Not recommended | [109] |
| c-Met inhibitors | ||||
| MSC2156119J | Sequential treatment after sorafenib failing | Phase I/II (ongoing) | Awaited | NCT02115373 |
| Tivantinib | Sequential treatment after sorafenib failing | Phase III (ongoing) | Awaited | NCT02029157 |
| Simultaneous combination with sorafenib | Phase I (finished) | Warranting further clinical study | [118] | |
| DE605 | Simultaneous combination with sorafenib | Preclinical study | Synergistic effects | [117] |
HCC is a hypervascular tumor and angiogenesis plays an important role in the progression of the disease[33]. Studies revealed that CD34, a sensitive angiogenesis-related endothelial marker, was not detected in healthy liver and cirrhotic liver, but was obviously expressed in HCC, suggesting that angiogenesis is probably a driving force in HCC development[34,35]. Regarding the molecular mechanisms of angiogenesis, VEGF is the best known angiogenic factor that promotes endothelial cell (EC) proliferation and migration[36-38]. Besides, various VEGF-independent drivers have also been recognized, including multiple interactions among diverse growth factors and receptors involving EC, delta-like ligand 4 (DLL4), angiopoietin (Ang)-Tie, placental growth factor (PIGF), tumoral cells (SDF1/CXCR4), pericytes [platelet-derived growth factor (PDGF) and transforming growth factor β (TGF-β)], extracellular matrix (ECM) components (integrins and cadherins), inflammatory cells (tumor-associated macrophages and Tie-2-expressing monocytes), and bone marrow-derived cells[39,40]. The antiangiogenic effect of sorafenib is mainly ascribed to its ability of inhibiting VEGFR-2, PDGFR, and these RTKs-mediated RAF/MEK/ERK pathway[41]. Although sorafenib exhibits reliable antiangiogenic effects, the complexity of angiogenesis suggests that it cannot block the formation of tumor microvessels completely[42]. The redundancy of angiogenic mechanisms may contribute to drug resistance through activation of alternative proangiogenic pathways. In this sense, combination of sorafenib and other antiangiogenic agents with different targets may improve the efficacy of sorafenib monotherapy and minimize the arising of drug resistance.
Activin receptor-like kinase-1 (ALK1) is a type I, endothelial cell-specific member of the TGF-β superfamily of receptors with high affinity for bone morphogenetic protein-9 (BMP9) and BMP10. Multiple lines of evidence demonstrated that BMP9/BMP10/ALK1 pathway is implicated in blood vessel formation and organization[43,44]. Dalantercept, a soluble form of ALK1 that prevents activation of endogenous ALK1 by BMP9 or BMP10, inhibits maturation of vascular endothelial cells, disrupts vascular development, and displays potent antitumor activity in preclinical models[45,46]. Distinguished with sorafenib, dalantercept targets this alternative angiogenic pathway and blocks common downstream events in the angiogenic process like the later vascular maturation stage. Thus combining dalantercept and sorafenib may achieve enhanced inhibition of tumor angiogenesis. It is worth noting that the side-effects of dalantercept mainly include lower extremity peripheral edema (grades 1-2) and congestive cardiac failure (grades 1-3), which do not appear to overlap with the toxicity profile of sorafenib[47]. Currently, an open-label, multi-center phase 1b study (NCT02024087) is ongoing to evaluate the safety, tolerability, pharmacokinetics, pharmacodynamics, preliminary activity, and the recommended phase 2 dose of dalantercept and sorafenib when used in combination in advanced HCC. This clinical study will also examine the biomarkers associated with tumor response including BMP9/10 and ALK1 in tumor biopsies and serum.
In parallel with the endeavors exploring the efficacy of simultaneous combination of sorafenib and antiangiogenic agents, studies have also been performed to investigate whether antiangiogenic drugs are available as the second-line therapy to control disease progression when sorafenib treatment fails. Axitinib is a multiple tyrosine kinase inhibitor targeting VEGFR1, VEGFR2, VEGFR3, PDGFR, and c-Kit[48]. It has been approved by the United States Food and Drug Administration for treatment of advanced kidney cancer. Preclinical studies demonstrated that the drug also exhibited potent activity against liver cancer[49]. A phase II trial (NCT01334112) of second-line axitinib following prior antiangiogenic therapy (sorafenib or bevacizumab) in advanced HCC showed that axitinib induced partial response in 1 of 26 patients and stable disease in 10 of 26 patients with a 16-wk cutoff, equivalent to a response rate of 3.8% and tumor control rate of 42.3%, which met the primary end point of the study[50]. The common adverse events in the axitinib group were hypertension, fatigue, dysphonia, and hypothyroidism[50]. Overall, axitinib showed encouraging clinical activity in patients pretreated with antiangiogenic agents in this study. Currently, there is also an ongoing multi-center, second-line study (EUCTR2011-002029-24-IT) of axitinib in patients with advanced HCC that progressed with sorafenib. The primary endpoint of the study is to assess the rate of patients without progression in an evaluation period of 4 mo and the results are still awaited.
Another antiangiogenic agent that is currently being evaluated as the second-line option for advanced HCC that acquires resistance to sorafenib is apatinib, which is a tyrosine kinase inhibitor selectively targeting VEGFR2[51]. In vitro studies demonstrated that it effectively inhibited proliferation, migration, and tube formation of human umbilical vein endothelial cells as well as blocked the budding of rat aortic ring[52]. In vivo studies showed that apatinib was capable of inhibiting the growth of several established human tumor xenografts[52]. A phase I study of apatinib showed encouraging antitumor activity and a manageable toxicity profile[53]. An interesting feature of this compound is that it can circumvent the multidrug resistance of cancer cells to certain conventional drugs by inhibiting the functions of multidrug resistance-associated proteins such as ABCB1 and ABCG2[54,55]. This characteristic of apatinib indicates that it might be useful in combining other drugs for cancer treatment. Thus far, a phase II trial (NCT01192971) of apatinib in patients with advanced HCC has been completed but the results have not been published. A phase III trial (NCT02329860) sponsored by the same company, i.e., Jiangsu HengRui Medicine Co., Ltd. (Jiangsu, China), was started on December 2014, aiming to evaluate the efficacy and safety of apatinib in patients with advanced HCC who have progressed on targeted therapy such as sorafenib and/or chemotherapy.
Except for the antiangiogenic effect, sorafenib can directly suppress the proliferation of HCC cells, which is ascribed to its ability of inhibiting RAF kinase and thus blocking the RAF/MEK/ERK signal pathway[56]. It is known that RAS/RAF/MEK/ERK represents a dominant signaling pathway promoting cell proliferation and survival[56,57]. The binding of different growth factors such as epidermal growth factor (EGF), hepatocyte growth factor (HGF), and insulin-like growth factor (IGF) to their receptors induces activation of RAS which in turn activates RAF, MEK, and ERK[58]. The activated ERK translocates into the nucleus and then activates transcription factors that regulate the expression of genes involved in cell proliferation and survival[58]. The RAS/RAF/MEK/ERK pathway is in an activated status in the majority of advanced HCC cases due to increased signaling induced by upstream growth factors such as EGF, HGF, and IGF[58,59]. Thus, inhibiting this signal pathway may significantly suppress the growth of HCC, which has been evidenced by many studies thus far. Although sorafenib is capable of interfering the signal transduction of this pathway via inhibiting the functions of RAF, its efficiency may be compromised by the complementary and/or feed-back mechanisms, which may partially restore the levels of phosphorylated ERK[60,61]. In order to overcome this issue, studies were performed to assess the efficacy and safety of combining sorafenib and MEK/ERK inhibitors in treatment of HCC. Thus far, some favorable results have been achieved in preclinical and clinical studies.
Huynh et al[60] reported that sorafenib significantly inhibited the growth of HCC xenografts but also caused elevation of insulin-like growth factor receptor 1 (IGF-1R) and phospho-c-RAF Ser 338 (activated form of c-RAF). The underlying mechanisms were revealed to be related with inhibition of c-RAF Ser 259 by sorafenib. It was reported that the phosphorylation of c-RAF at Ser259 prevented c-RAF activation, and that the dephosphorylation of c-RAF at Ser259 was an essential part of the c-RAF activation process[62]. It is possible that inhibition of phospho-c-RAF Ser 259 by sorafenib facilitates the phosphorylation of c-Raf at Ser338 which in turn promotes the phosphorylation of MEK and ERK. This finding warrants evaluation of the efficacy of combining sorafenib and MEK inhibitors in HCC treatment. As expected, inhibition of MEK by a small molecular compound AZD6244 (also named ARRY-142886) obviously enhanced the antitumor effect of sorafenib in both orthotopic and ectopic models of HCC[60]. A phase II trial (NCT01029418) was started in December 2009 to evaluate the efficacy of combination of AZD6244 and sorafenib in treatment of advanced HCC. Unfortunately, this trial was terminated without any definite conclusions due to funding issue in April 2015. Further studies are needed to verify the strategy of combining sorafenib and AZD6244 in treatment of HCC.
Another feed-back model involved in the RAF/MEK/ERK pathway also demonstrated the rationale of simultaneous inhibition of RAF and MEK in treatment of HCC. It has been described that the activated ERK could inhibit RAF, which is a negative feed-back regulation of RAF/MEK/ERK signal transduction[63]. Inhibition of MEK/ERK signaling by MEK inhibitors relieves ERK-dependent feedback inhibition of RAF and then compensatorily induces MEK phosphorylation[63,64]. Thus, MEK inhibitors combining sorafenib that inhibits RAF may block the feed-back loop and efficiently inhibit the RAF/MEK/ERK signal transduction. Interestingly, this model of action for MEK feed-back regulation is true for refametinib (BAY 86-9766)[61], an orally available small molecule that binds to an allosteric region adjacent to the ATP-binding pocket of MEK and inhibits both MEK 1 and MEK 2 with high potency and selectivity. In preclinical studies, refametinib exhibited potent antiproliferative activity in HCC cell lines and was strongly synergistic with sorafenib in suppressing the growth of HCC xenografts[61]. At the signaling pathway level, the combination of refametinib and sorafenib led to inhibition of the upregulatory feedback loop toward MEK phosphorylation observed after refametinib monotreatment[61]. In a completed single-arm phase II trial (NCT01204177), disease control rate (DCR), TTP, and OS were higher compared with previous sorafenib monotherapy studies (44.8% vs 35.3% for DCR; 4.1 mo vs 2.8 mo for TTP; 9.7 mo vs 6.5 mo for OS), especially in relation to Asian patients[65]. With regard to the toxicity, both refametinib and sorafenib were tolerated; however, most patients required dose modifications, mainly due to frequent grade 3 adverse events. Overall, combining refametinib and sorafenib provides a potential option for patients with unresectable HCC, which warrants a phase III trial for further evaluation.
Similar to the RAF/MEK/ERK pathway, PI3K/AKT/mTOR is also a major intracellular signaling pathway that regulates multiple cellular functions, including cell growth and proliferation, motility, survival, apoptosis, autophagy, and angiogenesis[66-68]. It has been described that the PI3K/AKT/mTOR signaling pathway plays a pivotal role in HCC and is activated in 30%-50% of HCC cases[69]. Many inhibitors targeting this pathway are currently being evaluated for HCC treatment in preclinical and clinical studies. Studies demonstrated that the RAF/MEK/ERK pathway could be activated as a consequence of mTOR inhibition, which might attenuate the antitumor effects of mTOR inhibitors[70,71]. On the other hand, inhibition of RAF/MEK/ERK signaling by sorafenib could induce increased mTOR phosphorylation, especially in sorafenib-resistant HCC cell lines[71,72]. The fact that blockage of RAF/MEK/ERK or PI3K/AKT/mTOR, separately, can result in activation of the other pathway underscores the potential of a combined therapeutic approach with agents targeting these two pathways. Another favorable factor of this combination strategy is that some mTOR inhibitors such as sirolimus and everolimus can be used as immunosuppressants to prevent organ rejection response after liver transplantation[73]. Thus, combination of mTOR inhibitors and sorafenib confers both anticancer and immunosuppressive properties, which might be suitable for treatment of recurrent HCC after liver transplantation.
Consistent with the theoretical assumption, addition of mTOR inhibitors such as sirolimus, everolimus, and temsirolimus to sorafenib augments antitumor effects in HCC preclinical studies in vitro and in vivo[74-76]. The issue of this combination strategy is that patients with advanced HCC showed lower tolerance to mTOR inhibitor or sorafenib compared to monotherapy. In a phase I study of the combination of everolimus and standard dose of sorafenib (400 mg twice daily) in advanced HCC, the maximum-tolerated dose (MTD) of everolimus was recommended to be 2.5 mg daily which is one third or fourth of the MTD established in everolimus monotherapy for advanced HCC[77]. Everolimus escalation beyond 5 mg daily in the combination settings was showed to be accompanied by dose limited toxicities such as grade 3 aspartate aminotransferase (AST) elevation, hyperbilirubinemia, and grade 3/4 thrombocytopenia. However, based on the effective dose of everolimus, a phase II trial (NCT01005199) is currently ongoing to assess the efficacy of sorafenib (400 mg twice daily) plus everolimus (5 mg daily) in treating patients with localized, unresectable, or metastatic liver cancer. Similar with everolimus, temsirolimus combined with sorafenib also results in reduced MTD of both drugs. A phase I dose-finding trial demonstrated that the MTD and recommended phase 2 dose of the combination strategy are temsirolimus 10 mg weekly plus sorafenib 200 mg twice daily[78], which are both lower than each single-agent MTD as well as the combination MTD identified in melanoma patients without hepatic dysfunction, which is temsirolimus 25 mg weekly plus sorafenib 400 mg in the mornings and 200 mg in the evenings[79]. A phase II trial (NCT01687673) is currently ongoing to evaluate the efficacy of temsirolimus (10 mg weekly) plus sorafenib (200 mg twice daily) in treatment of advanced HCC.
The efficacy of combination therapy with mTOR inhibitors and sorafenib for recurrent HCC after liver transplantation was initially described in several case reports, implying the usefulness of this combination regimen[80,81]. Due to concerns of the toxicity of mTOR inhibitors and sorafenib when used in combination, two clinical retrospective analyses were performed to assess the efficacy and safety of this combination modality for recurrent HCC after liver transplantation[82,83]. A single-center retrospective study analyzed the data of 7 patients receiving everolimus and sorafenib treatment and showed that the regimen is challenging regarding side effects and requires close patient monitoring to adapt everolimus dosage to sorafenib exposure and toxicity[83]. The other clinical study analyzed the data of 31 patient who were concomitantly administered everolimus or sirolimus plus sorafenib and confirmed the effectiveness of this combination strategy described in previous case reports[82]. A significant but manageable toxicity profile of mTOR inhibitor plus sorafenib was also observed in this study, which cautions that a careful assessment of potential vascular or hemorrhagic complications should be performed for all patients. The study suggests that combined treatment with mTOR inhibitor and sorafenib might be a therapeutic option for post-transplant HCC recurrence not amenable to curative treatments, which warrants further randomized and controlled clinical studies in the future.
The above mentioned mTOR inhibitors including sirolimus, everolimus, and temsirolimus specifically target mTORC1 (mechanistic target of rapamycin complex 1), which is one of the two components of mTOR[84]. Because they do not directly inhibit the other component of mTOR, mTORC2, that is also vital to tumor maintenance and progression, the action mode of these drugs does not fully exploit the antitumor potential of mTOR targeting in cancer[85]. Additionally, mTORC1 can also negatively regulate PI3K, implicating potential feedback activation of PI3K by sirolimus, everolimus, or temsirolimus[86]. An agent that is able to inhibit both mTORC1 and mTORC2, and also block upstream of mTOR, will result in a stronger inhibition of the PI3K/AKT/mTOR pathway. PI-103 and PKI-587 are representatives of this class of drugs, which showed potent inhibitory effects on various human cancers including HCC in pre-clinical studies[87,88]. Gedaly et al[71,88] demonstrated that PI-103 plus sorafenib or PKI-587 plus sorafenib could synergistically inhibit EGF-stimulated HCC cells proliferation compared with monotherapy. Further studies revealed that activation of PI3K/AKT/mTOR by EGF was blocked by PI-103 or PKI-587 which, however, stimulated MEK and ERK phosphorylation; activation of RAS/RAF/MEK/ERK by EGF was suppressed by sorafenib which, on the other hand, induced AKT and mTOR phosphorylation[71,88]. Either PI-103 or PKI-587 combining with sorafenib could inhibit all the tested kinases in the RAS/RAF/MEK/ERK and PI3K/AKT/mTOR pathways, which accounts for the superior effects of combination regimen to single drug in suppressing the proliferation of HCC cells. These studies reinforce the rationale of simultaneous inhibition of HCC pivotal pathways Ras/Raf/MEK/ERK and PI3K/AKT/mTOR.
Histone deacetylases (HDACs), along with histone acetyltransferases, reciprocally regulate the acetylation status of the positively charged NH2-terminal histone tails of nucleosomes[89]. Acetylation of histones by HDACs reduces the affinity of histones for DNA, resulting in an open DNA structure that facilitates gene expression[89,90]. HDAC inhibitors are currently considered to be among the most promising anticancer agents in drug development and some of them such as panobinostat and vorinostat have been approved for marketing in treatment of multiple myeloma and cutaneous T cell lymphoma[91,92]. It has been demonstrated that overexpression of HDACs is associated with poor prognosis and survival rates of HCC patients[93]. The efficacy and safety of HDAC inhibitors including resminostat and belinostat in treatment of advanced HCC have been validated in phase II clinical trials[94,95], indicating the potential value of HDAC inhibitors for HCC therapy.
The strategy of combining HDAC inhibitors and sorafenib for enhanced antitumor efficacy has been tested in HCC. Preclinical studies showed that addition of HDAC inhibitors including panobinostat, vorinostat, and MPT0E028 to sorafenib achieved additive or synergistic effects against HCC both in vitro and in vivo, providing the rationale for clinical studies with this combination regimen[96-98]. Panobinostat, a non-selective HDAC inhibitor (pan-HDAC inhibitor), was once sporadically reported to enhance the antitumor effect of sorafenib in patients with advanced HCC[99,100]. However, the subsequent phase I trial (NCT00873002) was terminated due to dose limiting toxicity, which precluded phase II study of this combination in treatment of advanced HCC. Vorinostat, an inhibitor of HDACs 1 and 3, is currently under phase I evaluation (NCT01075113) when combination of sorafenib in the setting of HCC, which is now suspended for interim analysis of dose limiting toxicity. Among HDAC inhibitors that are evaluated as combined drugs with sorafenib, resminostat that inhibits the activities of HDACs 1, 2, and 3 was testified to be a promising option. A recent multi-center, phase II Shelter study (NCT00943449) carried out in 8 German and 6 Italian centers validated the efficacy and safety of resminostat plus sorafenib for advanced HCC patients. Results demonstrated that combination of resminostat and sorafenib yielded a striking 70% progression free survival (PFS) rate at 12 wk[101]. Moreover, the treatment improved the OS of patients from 3 mo to 8 mo. Encouraged by these findings, a phase I/II study (NCT02400788) was implemented in Japan and South Korea in March 2015 to assess the availability of resminostat plus sorafenib in Asian patients with advanced HCC. While much work remains to be done, the currently achieved results open a door to better treatment prospects for this devastating disease.
The mechanisms underlying the synergistic effects of combination of HDAC inhibitors and sorafenib against HCC have been revealed from the following several aspects. First, sorafenib inhibits the RAF/MEK/ERK pathway and causes increased expression of Wnt-pathway regulator CDH1, a pivotal player of hepatocarcinogenesis known to be implicated in growth arrest[102]. Because HDACs can directly suppress CDH1 expression, combination of HDAC inhibitors and sorafenib results in a further rise of CDH1 expression, which might partially explain the observed potent antitumor effects of the combined therapy[96]. Second, HDAC inhibitors such as MPT0E028 is capable of activating ERK and its downstream molecules via induction of FGFR3-mediated signaling, suggesting that HDAC inhibitors may render HCC cells more dependent on ERK signaling[98]. Abrogation of RAF/MEK/ERK signaling by sorafenib may sensitize tumor cells to HDAC inhibitor-induced cell apoptosis[98]. Third, HDAC inhibitors can upregulate signal transduction pathways related to angiogenesis by modulating the expression of growth factors like VEGF or of downstream kinases like mitogen activated protein kinases (MAPKs), which are also the targets of sorafenib[103,104]. The dual blockade of tumor cell proliferation and tumor angiogenesis could thus represent a molecule basis for the synergistic effects of the combination therapy.
The EGFR pathway was demonstrated to be implicated in the pathogenesis of several cancers including HCC[58]. As an orally active inhibitor of EGFR tyrosine kinase, erlotinib has been approved to treat patients with advanced non-small cell lung and pancreatic cancers. In terms of HCC, two single-arm phase II trials showed that erlotinib exhibits modest antitumor activity but promising OS benefit[105,106]. It was reported that EGFR activation may interfere with HCC response to sorafenib, indicating that EGFR inhibition may enhance the antitumor effects of sorafenib[107]. Although preclinical studies showed no improvement of sorafenib efficacy upon combination with erlotinib in an orthotopic rat model of HCC[108], the combination showed promising antitumor efficacy (including HCC) in a phase I trial[109]. Encouraged by the results of this clinical trial, phase II trials were skipped and a phase III study (NCT0901901) was directly performed to compare the efficacy and safety of first-line sorafenib/erlotinib with sorafenib/placebo in patients with advanced HCC. Recently published results of this study demonstrated that median OS and median TTP were similar in the sorafenib plus erlotinib and sorafenib plus placebo groups[110]. With regard to the safety profile, the rates of treatment-emergent serious adverse events (AEs) and drug-related serious AEs were comparable in the two arms. The lack of synergistic or additive effect of the combination of erlotinib and sorafenib suggests that EGFR signaling may not be pivotal in advanced HCC. Thus, application of EGFR inhibitors plus sorafenib is not recommended to treat advanced HCC.
Like EGFR, the mesenchymal-epithelial transition factor (c-Met) is also a typical member of RTKs. HGF and des-γ-carboxy prothrombin (DCP) are revealed to be high-affinity ligands for c-Met[10,111,112]. Aberrant activation of the c-Met signaling pathway plays a critical role in cancer progression and metastasis by promoting cell proliferation, survival, and motility, suggesting that c-Met is a promising target for cancer therapy[113]. Overexpression of c-Met alone has also been demonstrated to be sufficient for developing HCC in c-Met-transgenic mice[114]. In addition, c-Met overexpression is observed in 20%-66% of human HCC samples and is closely related with patients’ prognosis[10]. It is worth noting that c-Met expression in HCC cells was upregulated after sorafenib treatment, which might be related with tumor response to the drug[115]. Studies by the current authors and other researchers showed that inhibition of c-Met kinase led to an obvious antitumor effect in HCC, indicating that targeting c-Met is also a promising strategy for HCC treatment[26,59]. Indeed, a randomized, placebo-controlled phase II trial showed that tivantinib, a selective inhibitor of c-Met, almost doubled median TTP and median OS in patients with high c-Met-expressing HCC who failed sorafenib treatment[116]. A confirmatory phase III, randomized, placebo-controlled trial (NCT02029157) is currently ongoing to evaluate the efficacy and safety of tivantinib in HCC patients who had high c-Met expression in their tumors and developed progressive disease under sorafenib therapy. Besides tivantinib, MSC2156119J, another c-Met inhibitor that shows potent inhibitory effects on the proliferation and metastasis of HCC cells in vivo[117], is currently under phase I/II study (NCT02115373) to assess its efficacy, safety, and pharmacokinetics in subjects with c-Met positive advanced HCC who have failed sorafenib treatment. Besides a second-line treatment option, c-Met inhibitors have also been tested for its efficacy upon combination with sorafenib. A recent report shows that sorafenib and DE605, a novel c-Met inhibitor, synergistically suppress the growth of HCC both in vitro and in vivo[118]. A phase I study of tivantinib in combination with sorafenib in patients with advanced solid tumors including HCC showed that the combination of tivantinib (360 mg, bid) plus sorafenib (240 mg, bid) was well tolerated and might have therapeutic potential in this setting[119]. Further studies are warranted to evaluate the efficacy and safety of this combination in the future. The above studies suggested that c-Met inhibitors may be combined as one of the options in sorafenib-based combination therapy.
Sorafenib remains the standard care for first-line treatment of advanced HCC. Unfortunately, the benefits of sorafenib may not be sustained, which requires alternative effective treatment regimens. The molecular pathogenesis of HCC is very complex, involving different pathways and molecular aberrations such as RAS/RAF/MEK/ERK, PI3K/AKT/mTOR, VEGF, c-Met, and HDACs, which warrants multiple-targeted therapeutic approaches. Simultaneous or sequential abrogation of these critical pathways or the functions of key molecules may yield major improvements in the management of HCC.
Several issues revealed in the past studies require attention when the combination regimen is translated from bench to bedside. First, the failure of combination of sorafenib and erlotinib in the phase III trial may be ascribed to the absence of phase II trials that assess the efficacy and safety of this combination in advanced HCC although promising results of phase II trials of erlotinib monotherapy are available. Future phase III trials should select agents and regimens with proven favorable safety profiles in HCC-specific phase I trials and documented efficacy based on well-designed and preferably randomized phase II trials. Second, clinical evidence suggests that combination regimen may be more often associated with dose-limiting effects, whereas single drugs can be used at maximal dosing level without causing intolerable toxic effects. Concerns over the toxicity of amplified drug toxicity in combination treatment trials have been the current bottleneck to translating positive preclinical experiments into clinical trials in HCC. This has been the case with the combination of sorafenib and mTOR inhibitors. Thus, combination of drugs that have none or less overlapped toxicity profiles and drug-drug interactions may minimize the risk of amplified adverse effects. Third, achievement of favorable therapeutic effects with combination regimens may also require patient screening to identify a responsive subset, which is critical for successful development of molecule targeted drugs and helpful for the subsequent individualized treatment. Identification of biomarkers that can predict or monitor tumor response to the treatment can facilitate this process and should be highlighted in the future. The heterogeneity of HCC determines the diversity of treatments. The emerging various kinds of tested sorafenib-based combination regimens may meet this requirement and help better manage this pressing disease.
P- Reviewer: Grassi G, Sangiovanni A, Welling TH S- Editor: Ma YJ L- Editor: Wang TQ E- Editor: Liu XM
| 1. | WHO. GLOBOCAN 2012: Estimated Cancer Incidence, Mortality and Prevalence Worldwide in 2012. Available from: http://globocan.iarc.fr/Pages/fact_sheets_cancer.aspx. Accessed December 30, 2013. |
| 2. | American Cancer Society. Cancer facts and figs 2005. Available from: http://wwwcancerorg/docroot/home/indexasp. Accessed October 21, 2013. |
| 3. | Schlageter M, Terracciano LM, D’Angelo S, Sorrentino P. Histopathology of hepatocellular carcinoma. World J Gastroenterol. 2014;20:15955-15964. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 138] [Cited by in RCA: 167] [Article Influence: 15.2] [Reference Citation Analysis (4)] |
| 4. | Chen X, Liu HP, Li M, Qiao L. Advances in non-surgical management of primary liver cancer. World J Gastroenterol. 2014;20:16630-16638. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 43] [Cited by in RCA: 49] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 5. | Song P, Feng X, Zhang K, Song T, Ma K, Kokudo N, Dong J, Yao L, Tang W. Screening for and surveillance of high-risk patients with HBV-related chronic liver disease: promoting the early detection of hepatocellular carcinoma in China. Biosci Trends. 2013;7:1-6. [PubMed] |
| 6. | Song P, Feng X, Inagaki Y, Song T, Zhang K, Wang Z, Zheng S, Ma K, Li Q, Kong D, Wu Q, Zhang T, Zhao X, Hasegawa K, Sugawara Y, Kokudo N, Tang W; Japan-China Joint Team for Medical Research and Cooperation on HCC. Clinical utility of simultaneous measurement of alpha-fetoprotein and des-γ-carboxy prothrombin for diagnosis of patients with hepatocellular carcinoma in China: A multi-center case-controlled study of 1,153 subjects. Biosci Trends. 2014;8:266-273. [PubMed] |
| 7. | Zhou DS, Xu L, Luo YL, He FY, Huang JT, Zhang YJ, Chen MS. Inflammation scores predict survival for hepatitis B virus-related hepatocellular carcinoma patients after transarterial chemoembolization. World J Gastroenterol. 2015;21:5582-5590. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 19] [Cited by in RCA: 21] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 8. | Kudo M. Surveillance, diagnosis, treatment, and outcome of liver cancer in Japan. Liver Cancer. 2015;4:39-50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 146] [Article Influence: 14.6] [Reference Citation Analysis (0)] |
| 9. | Johnson PJ. Non-surgical treatment of hepatocellular carcinoma. HPB (Oxford). 2005;7:50-55. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 30] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 10. | Gao J, Feng X, Inagaki Y, Song P, Kokudo N, Hasegawa K, Sugawara Y, Tang W. Des-γ-carboxy prothrombin and c-Met were concurrently and extensively expressed in hepatocellular carcinoma and associated with tumor recurrence. Biosci Trends. 2012;6:153-159. [PubMed] |
| 11. | Zhang W, Zhao G, Wei K, Zhang Q, Ma W, Song T, Wu Q, Zhang T, Kong D, Li Q. Adjuvant sorafenib reduced mortality and prolonged overall survival and post-recurrence survival in hepatocellular carcinoma patients after curative resection: a single-center experience. Biosci Trends. 2014;8:333-338. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 28] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 12. | Guo R, Feng X, Xiao S, Yan J, Xia F, Ma K, Li X. Short- and long-term outcomes of hepatectomy with or without radiofrequency-assist for the treatment of hepatocellular carcinomas: a retrospective comparative cohort study. Biosci Trends. 2015;9:65-72. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 13. | Hsu C, Chen BB, Chen CH, Ho MC, Cheng JC, Kokudo N, Murakami T, Yeo W, Seong J, Jia JD. Consensus Development from the 5th Asia-Pacific Primary Liver Cancer Expert Meeting (APPLE 2014). Liver Cancer. 2015;4:96-105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 14. | Song P. Standardizing management of hepatocellular carcinoma in China: devising evidence-based clinical practice guidelines. Biosci Trends. 2013;7:250-252. [PubMed] |
| 15. | Wang Z, Zhang G, Wu J, Jia M. Adjuvant therapy for hepatocellular carcinoma: current situation and prospect. Drug Discov Ther. 2013;7:137-143. [PubMed] |
| 16. | Zhong Y, Liu B, Deng M, Xu R. Adjuvant systemic drug therapy and recurrence of hepatocellular carcinoma following curative resection. Drug Discov Ther. 2013;7:164-166. [PubMed] |
| 17. | Slotta JE, Kollmar O, Ellenrieder V, Ghadimi BM, Homayounfar K. Hepatocellular carcinoma: Surgeon’s view on latest findings and future perspectives. World J Hepatol. 2015;7:1168-1183. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 42] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 18. | Yamaguchi T, Nakajima N, Nakamura I, Mashiba H, Kawashiro T, Ebara K, Ichimura E, Nishimura C, Okamoto K, Ichikawa Y. Preclinical anticancer effects and toxicologic assessment of hepatic artery infusion of fine-powder cisplatin with lipiodol in vivo. Drug Discov Ther. 2013;7:201-208. [PubMed] |
| 19. | Bodzin AS, Busuttil RW. Hepatocellular carcinoma: Advances in diagnosis, management, and long term outcome. World J Hepatol. 2015;7:1157-1167. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 68] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 20. | Ni JY, Xu LF, Wang WD, Sun HL, Chen YT. Conventional transarterial chemoembolization vs microsphere embolization in hepatocellular carcinoma: a meta-analysis. World J Gastroenterol. 2014;20:17206-17217. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 26] [Cited by in RCA: 33] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 21. | Chen J, Gao J. Advances in the study of molecularly targeted agents to treat hepatocellular carcinoma. Drug Discov Ther. 2014;8:154-164. [PubMed] |
| 22. | Au V, Tsang FH, Man K, Fan ST, Poon RT, Lee NP. Expression of ankyrin repeat and SOCS box containing 4 (ASB4) confers migration and invasion properties of hepatocellular carcinoma cells. Biosci Trends. 2014;8:101-110. [PubMed] |
| 23. | Hu L, Xue F, Shao M, Deng A, Wei G. Aberrant expression of Notch3 predicts poor survival for hepatocellular carcinomas. Biosci Trends. 2013;7:152-156. [PubMed] |
| 24. | Xia JF, Gao JJ, Inagaki Y, Kokudo N, Nakata M, Tang W. Flavonoids as potential anti-hepatocellular carcinoma agents: recent approaches using HepG2 cell line. Drug Discov Ther. 2013;7:1-8. [PubMed] |
| 25. | Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, de Oliveira AC, Santoro A, Raoul JL, Forner A. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359:378-390. [PubMed] |
| 26. | Qin Y, Lu Y, Wang R, Li W, Qu X. SL1122-37, a novel derivative of sorafenib, has greater effects than sorafenib on the inhibition of human hepatocellular carcinoma (HCC) growth and prevention of angiogenesis. Biosci Trends. 2013;7:237-244. [PubMed] |
| 27. | Shen YC, Lin ZZ, Hsu CH, Hsu C, Shao YY, Cheng AL. Clinical trials in hepatocellular carcinoma: an update. Liver Cancer. 2013;2:345-364. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 59] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 28. | Cabibbo G, Maida M, Genco C, Parisi P, Peralta M, Antonucci M, Brancatelli G, Cammà C, Craxì A, Di Marco V. Natural history of untreatable hepatocellular carcinoma: A retrospective cohort study. World J Hepatol. 2012;4:256-261. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 62] [Cited by in RCA: 70] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 29. | Di Costanzo GG, Tortora R. Intermediate hepatocellular carcinoma: How to choose the best treatment modality? World J Hepatol. 2015;7:1184-1191. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 17] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 30. | Yim HJ, Suh SJ, Um SH. Current management of hepatocellular carcinoma: an Eastern perspective. World J Gastroenterol. 2015;21:3826-3842. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 42] [Cited by in RCA: 47] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 31. | Ikura Y. Transitions of histopathologic criteria for diagnosis of nonalcoholic fatty liver disease during the last three decades. World J Hepatol. 2014;6:894-900. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 11] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 32. | Zekri AR, Bahnasy AA, Shoeab FE, Mohamed WS, El-Dahshan DH, Ali FT, Sabry GM, Dasgupta N, Daoud SS. Methylation of multiple genes in hepatitis C virus associated hepatocellular carcinoma. J Adv Res. 2014;5:27-40. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 31] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 33. | Pecchi A, Besutti G, De Santis M, Del Giovane C, Nosseir S, Tarantino G, Di Benedetto F, Torricelli P. Post-transplantation hepatocellular carcinoma recurrence: Patterns and relation between vascularity and differentiation degree. World J Hepatol. 2015;7:276-284. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 23] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 34. | Chen L, Yang Z, Wang G, Wang C. Expression of angiopoietin-2 gene and its receptor Tie2 in hepatocellular carcinoma. J Tongji Med Univ. 2001;21:228-230, 235. [PubMed] |
| 35. | Scaggiante B, Kazemi M, Pozzato G, Dapas B, Farra R, Grassi M, Zanconati F, Grassi G. Novel hepatocellular carcinoma molecules with prognostic and therapeutic potentials. World J Gastroenterol. 2014;20:1268-1288. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 58] [Cited by in RCA: 61] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 36. | Gong C, Xu C, Ji L, Wang Z. A novel semi-synthetic andrographolide analogue A5 inhibits tumor angiogenesis via blocking the VEGFR2-p38/ERK1/2 signal pathway. Biosci Trends. 2013;7:230-236. [PubMed] |
| 37. | Cui Z, Zhou H, He C, Wang W, Yang Y, Tan Q. Upregulation of Bcl-2 enhances secretion of growth factors by adipose-derived stem cells deprived of oxygen and glucose. Biosci Trends. 2015;9:122-128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 38. | Gu X, Miao X, Xu J, Cao J. An in vitro study of pcDNA 3.0-hVEGF165 gene transfection in endothelial progenitor cells derived from peripheral blood of rabbits. Biosci Trends. 2012;6:270-275. [PubMed] |
| 39. | Goh PP, Sze DM, Roufogalis BD. Molecular and cellular regulators of cancer angiogenesis. Curr Cancer Drug Targets. 2007;7:743-758. [PubMed] |
| 40. | Ferrara N. Pathways mediating VEGF-independent tumor angiogenesis. Cytokine Growth Factor Rev. 2010;21:21-26. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 216] [Cited by in RCA: 222] [Article Influence: 13.9] [Reference Citation Analysis (0)] |
| 41. | Tejeda-Maldonado J, García-Juárez I, Aguirre-Valadez J, González-Aguirre A, Vilatobá-Chapa M, Armengol-Alonso A, Escobar-Penagos F, Torre A, Sánchez-Ávila JF, Carrillo-Pérez DL. Diagnosis and treatment of hepatocellular carcinoma: An update. World J Hepatol. 2015;7:362-376. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 84] [Cited by in RCA: 94] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 42. | Sun H, Zhu MS, Wu WR, Shi XD, Xu LB. Role of anti-angiogenesis therapy in the management of hepatocellular carcinoma: The jury is still out. World J Hepatol. 2014;6:830-835. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 19] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 43. | Albiñana V, Sanz-Rodríguez F, Recio-Poveda L, Bernabéu C, Botella LM. Immunosuppressor FK506 increases endoglin and activin receptor-like kinase 1 expression and modulates transforming growth factor-β1 signaling in endothelial cells. Mol Pharmacol. 2011;79:833-843. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 47] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 44. | Oh SP, Seki T, Goss KA, Imamura T, Yi Y, Donahoe PK, Li L, Miyazono K, ten Dijke P, Kim S. Activin receptor-like kinase 1 modulates transforming growth factor-beta 1 signaling in the regulation of angiogenesis. Proc Natl Acad Sci USA. 2000;97:2626-2631. [PubMed] |
| 45. | Cunha SI, Pardali E, Thorikay M, Anderberg C, Hawinkels L, Goumans MJ, Seehra J, Heldin CH, ten Dijke P, Pietras K. Genetic and pharmacological targeting of activin receptor-like kinase 1 impairs tumor growth and angiogenesis. J Exp Med. 2010;207:85-100. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 134] [Cited by in RCA: 138] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 46. | Mitchell D, Pobre EG, Mulivor AW, Grinberg AV, Castonguay R, Monnell TE, Solban N, Ucran JA, Pearsall RS, Underwood KW. ALK1-Fc inhibits multiple mediators of angiogenesis and suppresses tumor growth. Mol Cancer Ther. 2010;9:379-388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 121] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 47. | Bendell JC, Gordon MS, Hurwitz HI, Jones SF, Mendelson DS, Blobe GC, Agarwal N, Condon CH, Wilson D, Pearsall AE. Safety, pharmacokinetics, pharmacodynamics, and antitumor activity of dalantercept, an activin receptor-like kinase-1 ligand trap, in patients with advanced cancer. Clin Cancer Res. 2014;20:480-489. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 50] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 48. | Minguet J, Smith KH, Bramlage CP, Bramlage P. Targeted therapies for treatment of renal cell carcinoma: recent advances and future perspectives. Cancer Chemother Pharmacol. 2015;76:219-233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 25] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 49. | Chen L, Liu H, Liu J, Zhu Y, Xu L, He H, Zhang H, Wang S, Wu Q, Liu W. Klotho endows hepatoma cells with resistance to anoikis via VEGFR2/PAK1 activation in hepatocellular carcinoma. PLoS One. 2013;8:e58413. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 34] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 50. | McNamara MG, Le LW, Horgan AM, Aspinall A, Burak KW, Dhani N, Chen E, Sinaei M, Lo G, Kim TK, Rogalla P, Bathe OF, Knox JJ. A phase II trial of second-line axitinib following prior antiangiogenic therapy in advanced hepatocellular carcinoma. Cancer. 2015;121:1620-1627. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 47] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 51. | Scott AJ, Messersmith WA, Jimeno A. Apatinib: a promising oral antiangiogenic agent in the treatment of multiple solid tumors. Drugs Today (Barc). 2015;51:223-229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 118] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 52. | Tian S, Quan H, Xie C, Guo H, Lü F, Xu Y, Li J, Lou L. YN968D1 is a novel and selective inhibitor of vascular endothelial growth factor receptor-2 tyrosine kinase with potent activity in vitro and in vivo. Cancer Sci. 2011;102:1374-1380. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 362] [Cited by in RCA: 428] [Article Influence: 30.6] [Reference Citation Analysis (0)] |
| 53. | Li J, Zhao X, Chen L, Guo H, Lv F, Jia K, Yv K, Wang F, Li C, Qian J. Safety and pharmacokinetics of novel selective vascular endothelial growth factor receptor-2 inhibitor YN968D1 in patients with advanced malignancies. BMC Cancer. 2010;10:529. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 166] [Cited by in RCA: 209] [Article Influence: 13.9] [Reference Citation Analysis (0)] |
| 54. | Mi YJ, Liang YJ, Huang HB, Zhao HY, Wu CP, Wang F, Tao LY, Zhang CZ, Dai CL, Tiwari AK. Apatinib (YN968D1) reverses multidrug resistance by inhibiting the efflux function of multiple ATP-binding cassette transporters. Cancer Res. 2010;70:7981-7991. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 267] [Cited by in RCA: 286] [Article Influence: 19.1] [Reference Citation Analysis (0)] |
| 55. | Tong XZ, Wang F, Liang S, Zhang X, He JH, Chen XG, Liang YJ, Mi YJ, To KK, Fu LW. Apatinib (YN968D1) enhances the efficacy of conventional chemotherapeutical drugs in side population cells and ABCB1-overexpressing leukemia cells. Biochem Pharmacol. 2012;83:586-597. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 106] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 56. | Woo HY, Heo J. Sorafenib in liver cancer. Expert Opin Pharmacother. 2012;13:1059-1067. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 47] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 57. | Wang H, Zhu QW, Ye P, Li ZB, Li Y, Cao ZL, Shen L. Pioglitazone attenuates myocardial ischemia-reperfusion injury via up-regulation of ERK and COX-2. Biosci Trends. 2012;6:325-332. [PubMed] |
| 58. | Berasain C, Nicou A, Garcia-Irigoyen O, Latasa MU, Urtasun R, Elizalde M, Salis F, Perugorría MJ, Prieto J, Recio JA. Epidermal growth factor receptor signaling in hepatocellular carcinoma: inflammatory activation and a new intracellular regulatory mechanism. Dig Dis. 2012;30:524-531. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 36] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 59. | Gao J, Inagaki Y, Song P, Qu X, Kokudo N, Tang W. Targeting c-Met as a promising strategy for the treatment of hepatocellular carcinoma. Pharmacol Res. 2012;65:23-30. [PubMed] |
| 60. | Huynh H, Ngo VC, Koong HN, Poon D, Choo SP, Toh HC, Thng CH, Chow P, Ong HS, Chung A. AZD6244 enhances the anti-tumor activity of sorafenib in ectopic and orthotopic models of human hepatocellular carcinoma (HCC). J Hepatol. 2010;52:79-87. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 78] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 61. | Schmieder R, Puehler F, Neuhaus R, Kissel M, Adjei AA, Miner JN, Mumberg D, Ziegelbauer K, Scholz A. Allosteric MEK1/2 inhibitor refametinib (BAY 86-9766) in combination with sorafenib exhibits antitumor activity in preclinical murine and rat models of hepatocellular carcinoma. Neoplasia. 2013;15:1161-1171. [PubMed] |
| 62. | Dhillon AS, Meikle S, Yazici Z, Eulitz M, Kolch W. Regulation of Raf-1 activation and signalling by dephosphorylation. EMBO J. 2002;21:64-71. [PubMed] |
| 63. | Ishii N, Harada N, Joseph EW, Ohara K, Miura T, Sakamoto H, Matsuda Y, Tomii Y, Tachibana-Kondo Y, Iikura H. Enhanced inhibition of ERK signaling by a novel allosteric MEK inhibitor, CH5126766, that suppresses feedback reactivation of RAF activity. Cancer Res. 2013;73:4050-4060. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 128] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 64. | Wang Y, Van Becelaere K, Jiang P, Przybranowski S, Omer C, Sebolt-Leopold J. A role for K-ras in conferring resistance to the MEK inhibitor, CI-1040. Neoplasia. 2005;7:336-347. [PubMed] |
| 65. | Lim HY, Heo J, Choi HJ, Lin CY, Yoon JH, Hsu C, Rau KM, Poon RT, Yeo W, Park JW. A phase II study of the efficacy and safety of the combination therapy of the MEK inhibitor refametinib (BAY 86-9766) plus sorafenib for Asian patients with unresectable hepatocellular carcinoma. Clin Cancer Res. 2014;20:5976-5985. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 80] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 66. | Meric-Bernstam F, Gonzalez-Angulo AM. Targeting the mTOR signaling network for cancer therapy. J Clin Oncol. 2009;27:2278-2287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 494] [Cited by in RCA: 503] [Article Influence: 31.4] [Reference Citation Analysis (0)] |
| 67. | Ge P, Cui Y, Liu F, Luan J, Zhou X, Han J. L-carnitine affects osteoblast differentiation in NIH3T3 fibroblasts by the IGF-1/PI3K/Akt signalling pathway. Biosci Trends. 2015;9:42-48. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 20] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 68. | Chen J, Wang W, Wang H, Liu X, Guo X. Combination treatment of ligustrazine piperazine derivate DLJ14 and adriamycin inhibits progression of resistant breast cancer through inhibition of the EGFR/PI3K/Akt survival pathway and induction of apoptosis. Drug Discov Ther. 2014;8:33-41. [PubMed] |
| 69. | Villanueva A, Chiang DY, Newell P, Peix J, Thung S, Alsinet C, Tovar V, Roayaie S, Minguez B, Sole M. Pivotal role of mTOR signaling in hepatocellular carcinoma. Gastroenterology. 2008;135:1972-1983, 1983.e1-e11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 564] [Cited by in RCA: 591] [Article Influence: 34.8] [Reference Citation Analysis (0)] |
| 70. | Carracedo A, Ma L, Teruya-Feldstein J, Rojo F, Salmena L, Alimonti A, Egia A, Sasaki AT, Thomas G, Kozma SC. Inhibition of mTORC1 leads to MAPK pathway activation through a PI3K-dependent feedback loop in human cancer. J Clin Invest. 2008;118:3065-3074. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 165] [Cited by in RCA: 765] [Article Influence: 45.0] [Reference Citation Analysis (0)] |
| 71. | Gedaly R, Angulo P, Hundley J, Daily MF, Chen C, Koch A, Evers BM. PI-103 and sorafenib inhibit hepatocellular carcinoma cell proliferation by blocking Ras/Raf/MAPK and PI3K/AKT/mTOR pathways. Anticancer Res. 2010;30:4951-4958. [PubMed] |
| 72. | Masuda M, Chen WY, Miyanaga A, Nakamura Y, Kawasaki K, Sakuma T, Ono M, Chen CL, Honda K, Yamada T. Alternative mammalian target of rapamycin (mTOR) signal activation in sorafenib-resistant hepatocellular carcinoma cells revealed by array-based pathway profiling. Mol Cell Proteomics. 2014;13:1429-1438. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 58] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 73. | Liang W, Wang D, Ling X, Kao AA, Kong Y, Shang Y, Guo Z, He X. Sirolimus-based immunosuppression in liver transplantation for hepatocellular carcinoma: a meta-analysis. Liver Transpl. 2012;18:62-69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 125] [Cited by in RCA: 130] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 74. | Huynh H, Ngo VC, Koong HN, Poon D, Choo SP, Thng CH, Chow P, Ong HS, Chung A, Soo KC. Sorafenib and rapamycin induce growth suppression in mouse models of hepatocellular carcinoma. J Cell Mol Med. 2009;13:2673-2683. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 103] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 75. | Newell P, Toffanin S, Villanueva A, Chiang DY, Minguez B, Cabellos L, Savic R, Hoshida Y, Lim KH, Melgar-Lesmes P. Ras pathway activation in hepatocellular carcinoma and anti-tumoral effect of combined sorafenib and rapamycin in vivo. J Hepatol. 2009;51:725-733. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 178] [Cited by in RCA: 177] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 76. | Piguet AC, Saar B, Hlushchuk R, St-Pierre MV, McSheehy PM, Radojevic V, Afthinos M, Terracciano L, Djonov V, Dufour JF. Everolimus augments the effects of sorafenib in a syngeneic orthotopic model of hepatocellular carcinoma. Mol Cancer Ther. 2011;10:1007-1017. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 68] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 77. | Finn RS, Poon RT, Yau T, Klümpen HJ, Chen LT, Kang YK, Kim TY, Gomez-Martin C, Rodriguez-Lope C, Kunz T. Phase I study investigating everolimus combined with sorafenib in patients with advanced hepatocellular carcinoma. J Hepatol. 2013;59:1271-1277. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 62] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 78. | Kelley RK, Nimeiri HS, Munster PN, Vergo MT, Huang Y, Li CM, Hwang J, Mulcahy MF, Yeh BM, Kuhn P. Temsirolimus combined with sorafenib in hepatocellular carcinoma: a phase I dose-finding trial with pharmacokinetic and biomarker correlates. Ann Oncol. 2013;24:1900-1907. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 46] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 79. | Davies MA, Fox PS, Papadopoulos NE, Bedikian AY, Hwu WJ, Lazar AJ, Prieto VG, Culotta KS, Madden TL, Xu Q. Phase I study of the combination of sorafenib and temsirolimus in patients with metastatic melanoma. Clin Cancer Res. 2012;18:1120-1128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 38] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 80. | Kim R, Aucejo F. Radiologic complete response with sirolimus and sorafenib in a hepatocellular carcinoma patient who relapsed after orthotopic liver transplantation. J Gastrointest Cancer. 2011;42:50-53. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 81. | Wang Y, Speeg KV, Washburn WK, Halff G. Sirolimus plus sorafenib in treating HCC recurrence after liver transplantation: a case report. World J Gastroenterol. 2010;16:5518-5522. [PubMed] |
| 82. | Gomez-Martin C, Bustamante J, Castroagudin JF, Salcedo M, Garralda E, Testillano M, Herrero I, Matilla A, Sangro B. Efficacy and safety of sorafenib in combination with mammalian target of rapamycin inhibitors for recurrent hepatocellular carcinoma after liver transplantation. Liver Transpl. 2012;18:45-52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 126] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 83. | De Simone P, Crocetti L, Pezzati D, Bargellini I, Ghinolfi D, Carrai P, Leonardi G, Della Pina C, Cioni D, Pollina L. Efficacy and safety of combination therapy with everolimus and sorafenib for recurrence of hepatocellular carcinoma after liver transplantation. Transplant Proc. 2014;46:241-244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 35] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 84. | Choi J, Chen J, Schreiber SL, Clardy J. Structure of the FKBP12-rapamycin complex interacting with the binding domain of human FRAP. Science. 1996;273:239-242. [PubMed] |
| 85. | Yu K, Shi C, Toral-Barza L, Lucas J, Shor B, Kim JE, Zhang WG, Mahoney R, Gaydos C, Tardio L. Beyond rapalog therapy: preclinical pharmacology and antitumor activity of WYE-125132, an ATP-competitive and specific inhibitor of mTORC1 and mTORC2. Cancer Res. 2010;70:621-631. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 161] [Cited by in RCA: 164] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 86. | Carracedo A, Pandolfi PP. The PTEN-PI3K pathway: of feedbacks and cross-talks. Oncogene. 2008;27:5527-5541. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 624] [Cited by in RCA: 694] [Article Influence: 40.8] [Reference Citation Analysis (0)] |
| 87. | Yang H, Rudge DG, Koos JD, Vaidialingam B, Yang HJ, Pavletich NP. mTOR kinase structure, mechanism and regulation. Nature. 2013;497:217-223. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 642] [Cited by in RCA: 802] [Article Influence: 66.8] [Reference Citation Analysis (0)] |
| 88. | Gedaly R, Angulo P, Hundley J, Daily MF, Chen C, Evers BM. PKI-587 and sorafenib targeting PI3K/AKT/mTOR and Ras/Raf/MAPK pathways synergistically inhibit HCC cell proliferation. J Surg Res. 2012;176:542-548. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 74] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 89. | Bolden JE, Peart MJ, Johnstone RW. Anticancer activities of histone deacetylase inhibitors. Nat Rev Drug Discov. 2006;5:769-784. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2201] [Cited by in RCA: 2301] [Article Influence: 121.1] [Reference Citation Analysis (0)] |
| 90. | Yang PH, Zhang L, Zhang YJ, Zhang J, Xu WF. HDAC6: physiological function and its selective inhibitors for cancer treatment. Drug Discov Ther. 2013;7:233-242. [PubMed] |
| 91. | Shi B, Xu W. The development and potential clinical utility of biomarkers for HDAC inhibitors. Drug Discov Ther. 2013;7:129-136. [PubMed] |
| 92. | Li X, Xu W. HDAC1/3 dual selective inhibitors - new therapeutic agents for the potential treatment of cancer. Drug Discov Ther. 2014;8:225-228. [PubMed] |
| 93. | Rikimaru T, Taketomi A, Yamashita Y, Shirabe K, Hamatsu T, Shimada M, Maehara Y. Clinical significance of histone deacetylase 1 expression in patients with hepatocellular carcinoma. Oncology. 2007;72:69-74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 138] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 94. | Yeo W, Chung HC, Chan SL, Wang LZ, Lim R, Picus J, Boyer M, Mo FK, Koh J, Rha SY. Epigenetic therapy using belinostat for patients with unresectable hepatocellular carcinoma: a multicenter phase I/II study with biomarker and pharmacokinetic analysis of tumors from patients in the Mayo Phase II Consortium and the Cancer Therapeutics Research Group. J Clin Oncol. 2012;30:3361-3367. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 160] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 95. | Zhao J, Lawless MW. Resminostat: Opening the door on epigenetic treatments for liver cancer. Hepatology. 2015;Epub ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 96. | Lachenmayer A, Toffanin S, Cabellos L, Alsinet C, Hoshida Y, Villanueva A, Minguez B, Tsai HW, Ward SC, Thung S. Combination therapy for hepatocellular carcinoma: additive preclinical efficacy of the HDAC inhibitor panobinostat with sorafenib. J Hepatol. 2012;56:1343-1350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 169] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 97. | Hsu FT, Liu YC, Chiang IT, Liu RS, Wang HE, Lin WJ, Hwang JJ. Sorafenib increases efficacy of vorinostat against human hepatocellular carcinoma through transduction inhibition of vorinostat-induced ERK/NF-κB signaling. Int J Oncol. 2014;45:177-188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 49] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 98. | Chen CH, Chen MC, Wang JC, Tsai AC, Chen CS, Liou JP, Pan SL, Teng CM. Synergistic interaction between the HDAC inhibitor, MPT0E028, and sorafenib in liver cancer cells in vitro and in vivo. Clin Cancer Res. 2014;20:1274-1287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 38] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 99. | Knieling F, Waldner MJ, Goertz RS, Strobel D. Quantification of dynamic contrast-enhanced ultrasound in HCC: prediction of response to a new combination therapy of sorafenib and panobinostat in advanced hepatocellular carcinoma. BMJ Case Rep. 2012;2012:pii bcr2012007576. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 100. | Gahr S, Wissniowski T, Zopf S, Strobel D, Pustowka A, Ocker M. Combination of the deacetylase inhibitor panobinostat and the multi-kinase inhibitor sorafenib for the treatment of metastatic hepatocellular carcinoma - review of the underlying molecular mechanisms and first case report. J Cancer. 2012;3:158-165. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 20] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 101. | Bitzer M, Giannini EG, Horger M, Lauer UM, Ganten TM, Woerns MA. International Liver Cancer Association, Sixth Annual Conference. Berlin: Germany 2012; . |
| 102. | Nagai T, Arao T, Furuta K, Sakai K, Kudo K, Kaneda H, Tamura D, Aomatsu K, Kimura H, Fujita Y. Sorafenib inhibits the hepatocyte growth factor-mediated epithelial mesenchymal transition in hepatocellular carcinoma. Mol Cancer Ther. 2011;10:169-177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 98] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 103. | Lemoine M, Derenzini E, Buglio D, Medeiros LJ, Davis RE, Zhang J, Ji Y, Younes A. The pan-deacetylase inhibitor panobinostat induces cell death and synergizes with everolimus in Hodgkin lymphoma cell lines. Blood. 2012;119:4017-4025. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 66] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 104. | LaBonte MJ, Wilson PM, Fazzone W, Russell J, Louie SG, El-Khoueiry A, Lenz HJ, Ladner RD. The dual EGFR/HER2 inhibitor lapatinib synergistically enhances the antitumor activity of the histone deacetylase inhibitor panobinostat in colorectal cancer models. Cancer Res. 2011;71:3635-3648. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 76] [Cited by in RCA: 77] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 105. | Philip PA, Mahoney MR, Allmer C, Thomas J, Pitot HC, Kim G, Donehower RC, Fitch T, Picus J, Erlichman C. Phase II study of Erlotinib (OSI-774) in patients with advanced hepatocellular cancer. J Clin Oncol. 2005;23:6657-6663. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 341] [Cited by in RCA: 344] [Article Influence: 17.2] [Reference Citation Analysis (0)] |
| 106. | Thomas MB, Chadha R, Glover K, Wang X, Morris J, Brown T, Rashid A, Dancey J, Abbruzzese JL. Phase 2 study of erlotinib in patients with unresectable hepatocellular carcinoma. Cancer. 2007;110:1059-1067. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 229] [Cited by in RCA: 234] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 107. | Blivet-Van Eggelpoël MJ, Chettouh H, Fartoux L, Aoudjehane L, Barbu V, Rey C, Priam S, Housset C, Rosmorduc O, Desbois-Mouthon C. Epidermal growth factor receptor and HER-3 restrict cell response to sorafenib in hepatocellular carcinoma cells. J Hepatol. 2012;57:108-115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 139] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 108. | Sieghart W, Pinter M, Dauser B, Rohr-Udilova N, Piguet AC, Prager G, Hayden H, Dienes HP, Dufour JF, Peck-Radosavljevic M. Erlotinib and sorafenib in an orthotopic rat model of hepatocellular carcinoma. J Hepatol. 2012;57:592-599. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 29] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 109. | Duran I, Hotté SJ, Hirte H, Chen EX, MacLean M, Turner S, Duan L, Pond GR, Lathia C, Walsh S. Phase I targeted combination trial of sorafenib and erlotinib in patients with advanced solid tumors. Clin Cancer Res. 2007;13:4849-4857. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 73] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 110. | Zhu AX, Rosmorduc O, Evans TR, Ross PJ, Santoro A, Carrilho FJ, Bruix J, Qin S, Thuluvath PJ, Llovet JM. SEARCH: a phase III, randomized, double-blind, placebo-controlled trial of sorafenib plus erlotinib in patients with advanced hepatocellular carcinoma. J Clin Oncol. 2015;33:559-566. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 396] [Cited by in RCA: 429] [Article Influence: 42.9] [Reference Citation Analysis (0)] |
| 111. | Zhang K, Song P, Gao J, Li G, Zhao X, Zhang S. Perspectives on a combined test of multi serum biomarkers in China: towards screening for and diagnosing hepatocellular carcinoma at an earlier stage. Drug Discov Ther. 2014;8:102-109. [PubMed] |
| 112. | Wang Z, Song P, Xia J, Inagaki Y, Tang W, Kokudo N. Can gamma-glutamyl transferase levels contribute to a better prognosis for patients with hepatocellular carcinoma? Drug Discov Ther. 2014;8:134-138. [PubMed] |
| 113. | Birchmeier C, Birchmeier W, Gherardi E, Vande Woude GF. Met, metastasis, motility and more. Nat Rev Mol Cell Biol. 2003;4:915-925. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1992] [Cited by in RCA: 2088] [Article Influence: 99.4] [Reference Citation Analysis (0)] |
| 114. | Wang R, Ferrell LD, Faouzi S, Maher JJ, Bishop JM. Activation of the Met receptor by cell attachment induces and sustains hepatocellular carcinomas in transgenic mice. J Cell Biol. 2001;153:1023-1034. [PubMed] |
| 115. | Gherardi E, Birchmeier W, Birchmeier C, Vande Woude G. Targeting MET in cancer: rationale and progress. Nat Rev Cancer. 2012;12:89-103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1057] [Cited by in RCA: 1153] [Article Influence: 88.7] [Reference Citation Analysis (0)] |
| 116. | Santoro A, Rimassa L, Borbath I, Daniele B, Salvagni S, Van Laethem JL, Van Vlierberghe H, Trojan J, Kolligs FT, Weiss A. Tivantinib for second-line treatment of advanced hepatocellular carcinoma: a randomised, placebo-controlled phase 2 study. Lancet Oncol. 2013;14:55-63. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 432] [Cited by in RCA: 463] [Article Influence: 38.6] [Reference Citation Analysis (0)] |
| 117. | Bladt F, Friese-Hamim M, Ihling C, Wilm C, Blaukat A. The c-Met Inhibitor MSC2156119J Effectively Inhibits Tumor Growth in Liver Cancer Models. Cancers (Basel). 2014;6:1736-1752. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 53] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 118. | Jiang X, Feng K, Zhang Y, Li Z, Zhou F, Dou H, Wang T. Sorafenib and DE605, a novel c-Met inhibitor, synergistically suppress hepatocellular carcinoma. Oncotarget. 2015;6:12340-12356. [PubMed] |
| 119. | Martell RE, Puzanov I, Ma WW, Santoro A, Dy GK, Goff LW, Fetterly GJ, Michael SA, Means-Powell JA, Chai F. Safety and efficacy of MET inhibitor tivantinib (ARQ 197) combined with sorafenib in patients (pts) with hepatocellular carcinoma (HCC) from a phase I study. 2012 ASCO Annual Meeting. Available from: http://meetinglibrary.asco.org/content/94768-114. |