Published online Jun 14, 2015. doi: 10.3748/wjg.v21.i22.6931
Peer-review started: December 3, 2014
First decision: December 26, 2014
Revised: January 19, 2015
Accepted: February 12, 2015
Article in press: February 13, 2015
Published online: June 14, 2015
Processing time: 202 Days and 16.1 Hours
AIM: To evaluate liver stiffness measurement (LSM) using non-invasive transient elastography (Fibroscan) in comparison with liver biopsy for assessment of liver fibrosis in children with biliary atresia (BA).
METHODS: Thirty-one children with BA admitted to the Department of Pediatric Surgery of Beijing Children’s Hospital from March 2012 to February 2013 were included in this study. Their preoperative LSM, liver biopsy findings, and laboratory results were studied retrospectively.
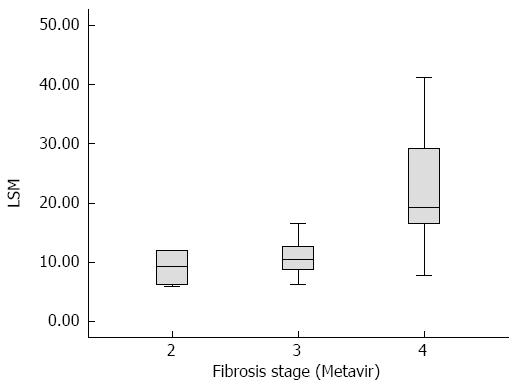
RESULTS: The grade of liver fibrosis in all 31 patients was evaluated according to the METAVIR scoring system, which showed that 4 cases were in group F2, 20 in group F3 and 7 in group F4. There were 24 non-cirrhosis cases (F2-F3) and 7 cirrhosis cases (F4). In groups F2, F3 and F4, the mean LSM was 9.10 ± 3.30 kPa, 11.02 ± 3.31 kPa and 22.86 ± 12.43 kPa, respectively. LSM was statistically different between groups F2 and F4 (P = 0.002), and between groups F3 and F4 (P = 0.000), however, there was no statistical difference between groups F2 and F3 (P = 0.593). The area under the receiver operating characteristic curve of LSM for ≥ F4 was 0.866. The cut-off value of LSM was 15.15 kPa for ≥ F4, with a sensitivity, specificity, positive predictive value and negative predictive value of 0.857, 0.917, 0.750 and 0.957, respectively.
CONCLUSION: Fibroscan can be used as a non-invasive technique to assess liver fibrosis in children with BA. The cut-off value of LSM (15.15 kPa) can distinguish cirrhotic patients from non-cirrhotic patients.
Core tip: This study was designed to evaluate liver fibrosis using non-invasive transient elastography (Fibroscan) in comparison with liver biopsy in children with biliary atresia (BA). According to the METAVIR scoring system, in groups F2, F3 and F4, the mean liver stiffness measurement (LSM) was 9.10 ± 3.30 kPa, 11.02 ± 3.31 kPa and 22.86 ± 12.43 kPa, respectively. The AUC of LSM for ≥ F4 was 0.886. The cut-off value of LSM was 15.15 kPa for ≥ F4, with high sensitivity, specificity, positive predictive value and negative predictive value. In conclusion, Fibroscan can be effectively used as a non-invasive technique to assess liver fibrosis in children with BA.
- Citation: Shen QL, Chen YJ, Wang ZM, Zhang TC, Pang WB, Shu J, Peng CH. Assessment of liver fibrosis by Fibroscan as compared to liver biopsy in biliary atresia. World J Gastroenterol 2015; 21(22): 6931-6936
- URL: https://www.wjgnet.com/1007-9327/full/v21/i22/6931.htm
- DOI: https://dx.doi.org/10.3748/wjg.v21.i22.6931
Biliary atresia (BA) is a unique pediatric liver disease characterized by progressive inflammatory obliterative cholangiopathy. If left untreated, fibrosclerosing obliteration progresses in both intrahepatic and extrahepatic bile ducts, which inevitably leads to liver cirrhosis[1]. The incidence of BA in Asia is reported to be as high as approximately 1 in 5000 live births[2], and in Western Europe is approximately 1 in 19000-15000 live births[3,4].
Assessment of liver fibrosis in BA is pivotal in treatment choice, evaluation of the results of the Kasai procedure and assessment of prognosis. Developed in France by Echosens, transient elastography (Fibroscan) is a novel, noninvasive technique which is used to assess the degree of liver fibrosis. The basic principle of Fibroscan involves a one-dimensional transient elastographic wave, which has a distinguishable traveling speed in different media, and can be translated into various degrees of fibrosis. When the liver is hard, the transient elastographic wave travels faster, resulting in a higher Liver Stiffness Measurement (LSM) value (kPa). Current studies concerning the use of Fibroscan seldom include BA patients. Recently, LSM was applied to predict liver related events in BA after Kasai hepatoportoenterostomy[5], especially oesophageal varices[6,7]. This study evaluated the cut-off value for Fibroscan to assess the degree of liver fibrosis in children with BA, and aimed to provide related evidence for the application of Fibroscan in pediatric medicine.
Thirty-one patients (10 males and 21 females) with BA admitted to the Pediatric Surgery Department of Beijing Children’s Hospital from March 2012 to February 2013 were included in this study. Regular blood tests, blood biochemistry tests and LSM were obtained three days before the Kasai procedure. Liver biopsy specimens, obtained during surgery, were well-preserved. The Japanese Society of Pediatric Surgery divided BA into three types: I, atresia of the common bile duct; II, atresia of the hepatic duct; and III, atresia of the porta hepatis. It was found that all the patients under study had type III BA. The age at operation ranged from 34 to 121 d, with a mean age of 75.58 ± 21.84 d.
Liver biopsy specimens were obtained from the front right area (5 or 6 segment of the Coinaud segment) of the liver during the Kasai procedure. All samples were preserved at -80 °C. The 10% neutral formalin-fixed and paraffin-embedded liver biopsy sections were stained with Masson trichromatic stain, which was carried out at the Department of Pathology of Beijing Friendship Hospital Affiliated to Capital Medical University. These sections were assessed using microscopy by two experienced pathologists according to the Metavir scoring system. A third pathologist was consulted when disagreement arose. According to the METAVIR scoring system[8], patients were divided into a non-cirrhosis group (F0-F3) and a cirrhosis group (F4).
Fibroscan (Echosens, France) was used to assess liver stiffness, and an experienced operator was responsible for obtaining the Fibroscan value. A probe (size S) was placed vertically on the skin surface between the right lower ribs. Ten values were then obtained avoiding major vessels. A median value calculated by the Statistics Analyze System was chosen as the final value, and the interquartile median ratio was less than 0.3.
The aspartate aminotransferase to platelet ratio index (APRI) was calculated using the following equation: APRI = [AST (/ULN)/platelet counts (109/L)] × 100, rounded to the third decimal place.
The data were analyzed using PASW software version 19.0. One-way analysis of variance was performed for the groups with different degrees of fibrosis. Spearman correlation and Logistic regression analyses were used to determine the relationship between LSM value and pathological grade of liver fibrosis. The accuracy of Fibroscan in differentiating fibrosis from cirrhosis was assessed using the area under the receiver operating characteristic (ROC) curve (AUC). An AUC between 0.7 and 0.9 represented a moderate accuracy, while that above 0.9 represented a high accuracy. The Youden index enabled selection of the optimal threshold value between liver fibrosis and liver cirrhosis, and predicted the upper limit of sensitivity and specificity of the different tests. A P value < 0.05 was considered statistically significant.
Before the Kasai procedure, all 31 patients underwent Fibroscan elastography and the success rate was 95.16% ± 6.54%. According to the METAVIR scoring system, no cases were classified in group F0 or F1, 4 cases were in group F2, 20 in group F3, and 7 in group F4. Thus, 24 cases were in the non-cirrhosis group (F0-F3) and 7 cases were in the cirrhosis group (F4). In groups F2, F3 and F4, the mean LSM was 9.10 ± 3.30 kPa, 11.02 ± 3.31 kPa and 22.86 ± 12.43 kPa, respectively. The APRI was 1.76 ± 1.12, 2.34 ± 1.93 and 2.72 ± 2.36, respectively. The blood test results are presented in Table 1.
| F2 (n = 4) | F3 (n = 20) | F4 (n = 7) | P-value | |
| LSM (kPa) | 9.10 ± 3.30 | 11.02 ± 3.31 | 22.86 ± 12.43 | 0.001 |
| AST (IU/L) | 251.25 ± 173.05 | 286.59 ± 147.11 | 328.01 ± 192.54 | 0.732 |
| ALT (IU/L) | 152.50 ± 142.69 | 180.54 ± 109.90 | 181.19 ± 92.99 | 0.893 |
| TBIL (μmol/L) | 176.98 ± 43.13 | 184.81 ± 37.06 | 194.81 ± 59.87 | 0.791 |
| DBIL (μmol/L) | 89.93 ± 27.46 | 86.21 ± 13.51 | 91.64 ± 27.04 | 0.790 |
| IBIL (μmol/L) | 87.05 ± 16.61 | 98.60 ± 37.29 | 103.17 ± 38.92 | 0.773 |
| ALP (U/L) | 471.75 ± 92.69 | 611.37 ± 310.79 | 626.44 ± 245.08 | 0.636 |
| GGT (U/L) | 631.00 ± 522.17 | 736.62 ± 587.59 | 569.89 ± 272.86 | 0.756 |
| TBA (μmol/L) | 103.73 ± 34.29 | 107.07 ± 35.01 | 144.09 ± 53.60 | 0.105 |
| APRI | 1.76 ± 1.12 | 2.34 ± 1.93 | 2.72 ± 2.36 | 0.741 |
| Age at operation (d) | 73.00 ± 18.67 | 70.70 ± 20.25 | 91.00 ± 23.49 | 0.100 |
The LSM in groups F2, F3 and F4 was analyzed by one-way analysis of variance, which revealed that the average LSM value in each group was statistically different (P = 0.001 < 0.01). Figure 1 is a box-plot of liver fibrosis stage (METAVIR) and LSM. A further comparison between F2 and F4, and between F3 and F4, revealed statistical differences (P < 0.01 for all), while group F2 compared to F3 showed no statistical difference (P = 0.593). Using Spearman rank correlation analysis, a positive correlation between LSM and liver fibrosis stage (R = 0.544, P = 0.002 < 0.01) was observed.
LSM value was determined in 24 patients in the non-cirrhosis group (F2-F3) and in 7 in the cirrhosis group (F4). After Logistic regression analysis, the coefficient of regression of the LSM value for diagnosing cirrhosis was 0.367 and the constant was -6.439 (P = 0.021 < 0.05). This regression equation model was tested by χ2 analysis (P = 0.056) and was found to be insufficient to demonstrate a significant difference, but it was sufficient to indicate that the regression equation model was suitable as the P value was very close to 0.05. This showed that with increasing LSM value, the probability of cirrhosis increased.
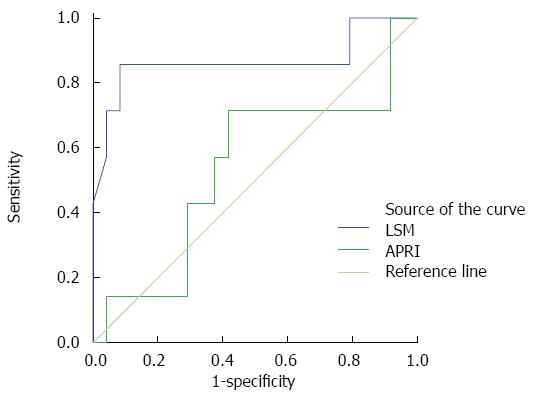
The AUC of LSM was 0.866 for the diagnosis of liver cirrhosis in group F4. The AUC of APRI was 0.536 (Figure 2). The value which corresponded to the maximum Youden index was considered the optimal threshold value. Therefore, the optimal cut-off values of LSM and APRI to diagnose cirrhosis were 15.15 kPa and 1.855, respectively. The sensitivity, specificity, positive predictive value, negative predictive value and diagnostic accuracy were 0.857, 0.917, 0.750, 0.957, and 0.903 for LSM, and 0.714, 0.583, 0.357, 0.882, and 0.645 for APRI, respectively.
As a novel noninvasive technique that allows quantification of the degree of liver fibrosis, Fibroscan has been used in adult patients with chronic hepatic diseases. It is not commonly used in children, mainly because there is no suitable probe. However, size S probe has recently been developed by Echosen, France, which can be used in children[9,10].
Some studies have suggested that if at least 10 values were obtained using Fibroscan and the success rate was higher than 65%, it was considered reliable[11,12]. In our study, size S probe (Echosen, France) was used in all 31 patients to measure liver stiffness. At least 10 values were obtained for each patient, and the success rate was 95.16% ± 6.54%.
As a noninvasive technique for assessing the degree of liver fibrosis, Fibroscan has been widely studied in adults, however, no conclusion regarding the cut-off value for different degrees of fibrosis was reached. According to the Metavir scoring system, the LSM cut-off value for F ≥ 2, F ≥ 3 and F = 4 is 6.9-7.9 kPa, 8.8-12.5 kPa and 11.9-19.5 kPa, respectively[13-16]. Based on the pathological stage of the liver specimen (Metavir scoring system) obtained during the Kasai operation, the 31 patients included in the present study were divided into different groups, and using single-factor variance analysis, a significant difference in LSM value was observed between F2 and F4, and between F3 and F4. Furthermore, ROC analysis indicated that the AUC for LSM to diagnose fibrosis or cirrhosis in groups F2, F3 and F4 was 0.259, 0.339 and 0.866, respectively. Although the AUC obtained in this study was smaller than that observed by Sporea et al[13], the overall trend was the same. That is, Fibroscan had a relatively high accuracy in differentiating cirrhosis and non-cirrhosis, although it did not provide sufficient information to determine the degree of fibrosis in non-cirrhosis patients[17]. An analysis of related risk factors for liver cirrhosis using Logistic regression indicated that LSM was the only related risk factor. This indicates that with increased LSM, the possibility of liver cirrhosis is higher. Therefore, we consider that LSM can be an assistant tool to distinguish liver cirrhosis from non-cirrhosis in children with BA.
In the analysis of the optimal cut-off value for LSM, 15.15 kPa was considered the best value to determine F4 stage. The sensitivity, specificity, positive predictive value and negative predictive value were 0.857, 0.917, 0.750 and 0.957, respectively. Compared with the positive predictive value, a higher negative predictive value indicated that BA children with an LSM value less than 15.15 kPa were less likely to have liver cirrhosis. As there are no studies on the optimal LSM cut-off value for liver cirrhosis in BA, we were unable to make comparisons. However, the cut-off value used in this study was relatively consistent with that in adult studies: 17.3 kPa for viral C cirrhosis[13], 17.5 kPa for nonalcoholic steatohepatitis[18], and 19.5 kPa for alcoholic cirrhosis[16].
When comparing Fibroscan (LSM > 15.15 kPa) with the gold standard (pathology) for the diagnosis of cirrhosis, 28 of 31 patients (90.3%) were accurately diagnosed by LSM. While the two techniques were moderately correlated, this was not the expected result. The AUC of LSM used to determine F4 was between 0.7 and 0.9, which was slightly lower than that found in other related studies[19]. The reasons for this may be as follows: (1) the sample size in this study was relatively small. The incidence of BA in mainland China has not yet been reported. This in turn added an obstacle in estimating sample capacity, therefore it is hard to estimate that 31 samples were enough or not. We are already working on a large sample size study, but it needs time; (2) different parts of the liver had different stage of fibrosis, therefore the specimen obtained may not have represented the overall stage of fibrosis. In order to minimize this error, we required that all fibroscan examinations and the liver specimens must be taken on the same location; and (3) LSM was affected by other factors[20-22], such as bilirubin, alanine aminotransferase and hemodynamics. In this study, we had done some statistical analyses about the relation between blood tests (AST, ALT, TBIL, DBIL, IBIL, ALP, GGT, and TBA), operation age and LSM, but there was no statistical significance (Table 1). Thus, we believe that the results of this study is reliable.
In this study, APRI was chosen as a representative noninvasive assessment method and was compared with Fibroscan for the accurate diagnosis of liver fibrosis. APRI was expressed as [AST(/ULN)/platelet counts (109/L)] × 100, which was developed in 2003 by Wai et al[23]. The results suggested that no significant difference in APRI was observed between groups F2, F3 and F4. The AUC to determine liver cirrhosis in children with BA was 0.536, and if the threshold value was 1.855, the sensitivity, specificity, positive predictive value and negative predictive value were 71.4%, 58.3%, 35.7% and 88.2%, respectively, which was obviously lower than the accuracy of the LSM value. These results are consistent with those of a study on the use of APRI and Fibroscan for the assessment of liver fibrosis in hepatitis B virus (HBV)-infected patients by Bonnard et al[24]. However, the results are not consistent with those obtained in a study assessing liver fibrosis and cirrhosis using APRI in children with BA by Kim et al[25]. The reasons for this may be as follows: (1) the primitive statistics in this model were too simple and were easily affected by various related factors; (2) the sample size in both studies was relatively small; and (3) errors may have occurred during blood testing, as the sample from each patient was examined separately and the agents used were not from the same batch.
In conclusion, Fibroscan has significant value in diagnosing liver cirrhosis in children with BA. A further study with a larger sample size aimed at obtaining accurate cut-off values to determine liver fibrosis and cirrhosis is required.
Assessment of liver fibrosis in biliary atresia is pivotal in treatment choice, evaluation of the results of the Kasai procedure and assessment of prognosis. Non-invasive methods for fibrosis assessment, such as Fibroscan, are being accepted more and more, replacing the invasive methods.
Many studies have been published regarding evaluation of liver fibrosis with Fibroscan in adults, but only a few have been published in biliary atresia. No study was about the cut-off value of liver stiffness measurement (LSM) to distinguish the different levels of liver fibrosis. This study was designed to evaluate liver fibrosis using non-invasive transient elastography (Fibroscan) in comparison with liver biopsy in children with biliary atresia.
This study used Fibroscan to evaluate liver fibrosis in biliary atresia in comparison with liver biopsy histopathology results. The cut-off value of LSM was 15.15 kPa for ≥ F4, with a sensitivity, specificity, positive predictive value and negative predictive value of 0.857, 0.917, 0.750 and 0.957, respectively.
Fibroscan can be effectively used as a non-invasive technique to assess liver fibrosis in children with biliary atresia. The cut-off value of LSM (15.15 kPa) can distinguish cirrhotic patients from non-cirrhotic patients.
Transient elastography (Fibroscan) is a novel, noninvasive technique which is used to assess the degree of liver fibrosis. The basic principle of Fibroscan involves a one-dimensional transient elastographic wave, which has a distinguishable traveling speed in different media, and can be translated into various degrees of fibrosis. When the liver is hard, the transient elastographic wave travels faster, resulting in a higher LSM value (kPa).
This is a retrospective analysis of transient elastography (Fibroscan) in the assessment of liver fibrosis in children with biliary atresia. This paper was concise, clear, and well-written. The results are interesting and suggest that Fibroscan can be used as a non-invasive technique to assess liver fibrosis in children with biliary atresia.
P- Reviewer: El-Zahaby SA S- Editor: Qi Y L- Editor: Wang TQ E- Editor: Zhang DN
| 1. | Hartley JL, Davenport M, Kelly DA. Biliary atresia. Lancet. 2009;374:1704-1713. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 608] [Cited by in RCA: 638] [Article Influence: 39.9] [Reference Citation Analysis (0)] |
| 2. | Hsiao CH, Chang MH, Chen HL, Lee HC, Wu TC, Lin CC, Yang YJ, Chen AC, Tiao MM, Lau BH. Universal screening for biliary atresia using an infant stool color card in Taiwan. Hepatology. 2008;47:1233-1240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 237] [Cited by in RCA: 190] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 3. | McKiernan PJ, Baker AJ, Kelly DA. The frequency and outcome of biliary atresia in the UK and Ireland. Lancet. 2000;355:25-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 298] [Cited by in RCA: 277] [Article Influence: 11.1] [Reference Citation Analysis (1)] |
| 4. | Chardot C, Carton M, Spire-Bendelac N, Le Pommelet C, Golmard JL, Auvert B. Epidemiology of biliary atresia in France: a national study 1986-96. J Hepatol. 1999;31:1006-1013. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 126] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 5. | Hahn SM, Kim S, Park KI, Han SJ, Koh H. Clinical benefit of liver stiffness measurement at 3 months after Kasai hepatoportoenterostomy to predict the liver related events in biliary atresia. PLoS One. 2013;8:e80652. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 24] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 6. | Colecchia A, Di Biase AR, Scaioli E, Predieri B, Iughetti L, Reggiani ML, Montrone L, Ceccarelli PL, Vestito A, Viola L. Non-invasive methods can predict oesophageal varices in patients with biliary atresia after a Kasai procedure. Dig Liver Dis. 2011;43:659-663. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 32] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 7. | Chang HK, Park YJ, Koh H, Kim SM, Chung KS, Oh JT, Han SJ. Hepatic fibrosis scan for liver stiffness score measurement: a useful preendoscopic screening test for the detection of varices in postoperative patients with biliary atresia. J Pediatr Gastroenterol Nutr. 2009;49:323-328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 46] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 8. | Bedossa P, Poynard T. An algorithm for the grading of activity in chronic hepatitis C. The METAVIR Cooperative Study Group. Hepatology. 1996;24:289-293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2860] [Cited by in RCA: 3082] [Article Influence: 106.3] [Reference Citation Analysis (0)] |
| 9. | Ferraioli G, Lissandrin R, Zicchetti M, Filice C. Assessment of liver stiffness with transient elastography by using S and M probes in healthy children. Eur J Pediatr. 2012;171:1415; author reply 1417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 10. | Engelmann G, Gebhardt C, Wenning D, Wühl E, Hoffmann GF, Selmi B, Grulich-Henn J, Schenk JP, Teufel U. Feasibility study and control values of transient elastography in healthy children. Eur J Pediatr. 2012;171:353-360. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 109] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 11. | Wang JH, Changchien CS, Hung CH, Eng HL, Tung WC, Kee KM, Chen CH, Hu TH, Lee CM, Lu SN. FibroScan and ultrasonography in the prediction of hepatic fibrosis in patients with chronic viral hepatitis. J Gastroenterol. 2009;44:439-446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 90] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 12. | Jang HW, Kim SU, Park JY, Ahn SH, Han KH, Chon CY, Park YN, Choi EH, Kim do Y. How many valid measurements are necessary to assess liver fibrosis using FibroScan® in patients with chronic viral hepatitis? An analysis of subjects with at least 10 valid measurements. Yonsei Med J. 2012;53:337-345. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 13. | Sporea I, Sirli R, Deleanu A, Tudora A, Popescu A, Curescu M, Bota S. Liver stiffness measurements in patients with HBV vs HCV chronic hepatitis: a comparative study. World J Gastroenterol. 2010;16:4832-4837. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 59] [Cited by in RCA: 61] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 14. | Ogawa E, Furusyo N, Toyoda K, Takeoka H, Otaguro S, Hamada M, Murata M, Sawayama Y, Hayashi J. Transient elastography for patients with chronic hepatitis B and C virus infection: Non-invasive, quantitative assessment of liver fibrosis. Hepatol Res. 2007;37:1002-1010. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 70] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 15. | Muñoz R, Ramírez E, Fernandez I, Martin A, Romero M, Romero E, Dominguez-Gil B, Hernandez A, Morales E, Andres A. Correlation between fibroscan, liver biopsy, and clinical liver function in patients with hepatitis C virus infection after renal transplantation. Transplant Proc. 2009;41:2425-2426. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 22] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 16. | Nguyen-Khac E, Chatelain D, Tramier B, Decrombecque C, Robert B, Joly JP, Brevet M, Grignon P, Lion S, Le Page L. Assessment of asymptomatic liver fibrosis in alcoholic patients using fibroscan: prospective comparison with seven non-invasive laboratory tests. Aliment Pharmacol Ther. 2008;28:1188-1198. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 156] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 17. | Poynard T, de Ledinghen V, Zarski JP, Stanciu C, Munteanu M, Vergniol J, France J, Trifan A, Le Naour G, Vaillant JC. Relative performances of FibroTest, Fibroscan, and biopsy for the assessment of the stage of liver fibrosis in patients with chronic hepatitis C: a step toward the truth in the absence of a gold standard. J Hepatol. 2012;56:541-548. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 66] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 18. | Yoneda M, Yoneda M, Mawatari H, Fujita K, Endo H, Iida H, Nozaki Y, Yonemitsu K, Higurashi T, Takahashi H. Noninvasive assessment of liver fibrosis by measurement of stiffness in patients with nonalcoholic fatty liver disease (NAFLD). Dig Liver Dis. 2008;40:371-378. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 300] [Cited by in RCA: 298] [Article Influence: 17.5] [Reference Citation Analysis (0)] |
| 19. | Alkhouri N, Sedki E, Alisi A, Lopez R, Pinzani M, Feldstein AE, Nobili V. Combined paediatric NAFLD fibrosis index and transient elastography to predict clinically significant fibrosis in children with fatty liver disease. Liver Int. 2013;33:79-85. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 82] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 20. | Yashima Y, Tsujino T, Masuzaki R, Nakai Y, Hirano K, Tateishi R, Sasahira N, Isayama H, Tada M, Yoshida H. Increased liver elasticity in patients with biliary obstruction. J Gastroenterol. 2011;46:86-91. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 21. | Trifan A, Sfarti C, Cojocariu C, Dimache M, Cretu M, Hutanasu C, Stanciu C. Increased liver stiffness in extrahepatic cholestasis caused by choledocholithiasis. Hepat Mon. 2011;11:372-375. [PubMed] |
| 22. | Harata M, Hashimoto S, Kawabe N, Nitta Y, Murao M, Nakano T, Arima Y, Shimazaki H, Ishikawa T, Okumura A. Liver stiffness in extrahepatic cholestasis correlates positively with bilirubin and negatively with alanine aminotransferase. Hepatol Res. 2011;41:423-429. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 32] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 23. | Wai CT, Greenson JK, Fontana RJ, Kalbfleisch JD, Marrero JA, Conjeevaram HS, Lok AS. A simple noninvasive index can predict both significant fibrosis and cirrhosis in patients with chronic hepatitis C. Hepatology. 2003;38:518-526. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2762] [Cited by in RCA: 3245] [Article Influence: 147.5] [Reference Citation Analysis (0)] |
| 24. | Bonnard P, Sombié R, Lescure FX, Bougouma A, Guiard-Schmid JB, Poynard T, Calès P, Housset C, Callard P, Le Pendeven C. Comparison of elastography, serum marker scores, and histology for the assessment of liver fibrosis in hepatitis B virus (HBV)-infected patients in Burkina Faso. Am J Trop Med Hyg. 2010;82:454-458. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 58] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 25. | Kim SY, Seok JY, Han SJ, Koh H. Assessment of liver fibrosis and cirrhosis by aspartate aminotransferase-to-platelet ratio index in children with biliary atresia. J Pediatr Gastroenterol Nutr. 2010;51:198-202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 74] [Article Influence: 4.9] [Reference Citation Analysis (0)] |