Published online Dec 7, 2014. doi: 10.3748/wjg.v20.i45.17196
Revised: July 9, 2014
Accepted: July 29, 2014
Published online: December 7, 2014
Processing time: 265 Days and 21.7 Hours
AIM: To determine the prevalence and time course of pancreatic exocrine insufficiency in individuals with newly diagnosed prediabetes or diabetes mellitus after acute pancreatitis.
METHODS: Relevant literature cited in three major biomedical journal databases (EMBASE, MEDLINE, and Scopus) was reviewed independently by two authors. There were no language constraints but the search was limited to human studies. Studies included were cohort studies of adult patients who were discharged after an attack of acute pancreatitis. Patients were excluded if they were under 18 years of age or had a previous diagnosis of prediabetes or diabetes mellitus, pancreatic exocrine insufficiency, or chronic pancreatitis. The main outcome measure was the prevalence of concomitant pancreatic exocrine insufficiency in patients who were diagnosed with prediabetes and diabetes mellitus after an attack of acute pancreatitis. Subgroup analysis was conducted for patients who were diagnosed with prediabetes only and those who were diagnosed with diabetes mellitus only. Subgroup analysis looking at the time course of concomitant pancreatic exocrine and endocrine insufficiency was also conducted. Pooled prevalence and corresponding 95% confidence intervals were calculated for all outcome measures and P-values < 0.05 were deemed statistically significant.
RESULTS: Eight clinical studies comprising of 234 patients met all eligibility criteria. The pooled prevalence of newly diagnosed prediabetes or diabetes in individuals after acute pancreatitis was 43% (95%CI: 30%-56%). The pooled prevalence of pancreatic exocrine insufficiency in individuals after acute pancreatitis was 29% (95%CI: 19%-39%). The prevalence of concomitant pancreatic exocrine insufficiency in individuals with newly diagnosed prediabetes or diabetes was 40% (95%CI: 25%-55%). The prevalence of concomitant pancreatic exocrine insufficiency among individuals with prediabetes alone and diabetes mellitus alone was 41% (95%CI: 12%-75%) and 39% (95%CI: 28%-51%), respectively. Further analysis showed that the prevalence of concomitant pancreatic exocrine insufficiency in individuals with prediabetes or diabetes decreases over time after an attack of acute pancreatitis.
CONCLUSION: Pancreatic exocrine insufficiency occurs in 40% of individuals with newly diagnosed prediabetes or diabetes mellitus after acute pancreatitis. Further studies are needed to investigate the pathogenesis of diabetes in this setting.
Core tip: Diabetes mellitus and pancreatic exocrine insufficiency are common after acute pancreatitis. Concomitant pancreatic exocrine insufficiency occurs in 40% of patients with prediabetes or diabetes and its prevalence decreases with time. Purposefully designed clinical studies are required to elucidate the pathogenesis of pancreatogenic diabetes.
- Citation: Das SLM, Kennedy JIC, Murphy R, Phillips ARJ, Windsor JA, Petrov MS. Relationship between the exocrine and endocrine pancreas after acute pancreatitis. World J Gastroenterol 2014; 20(45): 17196-17205
- URL: https://www.wjgnet.com/1007-9327/full/v20/i45/17196.htm
- DOI: https://dx.doi.org/10.3748/wjg.v20.i45.17196
Acute pancreatitis (AP) is the most common pancreatic disease and the incidence of AP worldwide is much higher than the combined incidence of pancreatic cancer (PC) and chronic pancreatitis (CP)[1]. It has been axiomatic that patients with AP completely recover, both clinically and morphologically. However, a recent comprehensive literature review and meta-analysis has demonstrated that patients not known to have diabetes mellitus (DM) prior to hospital admission with their first attack of AP have almost a 40% risk of being diagnosed with prediabetes or DM after discharge from hospital[2].
The pancreas is a unique dual gland that has both exocrine and endocrine functions. It consists of 95% exocrine tissue and less than 5% endocrine tissue. Given the close anatomical and physiological relationship of the exocrine and endocrine pancreas, exocrine pancreatic morphology and function have been studied in patients with DM for over 70 years[3-10]. However, to date there has been no systematic review and meta-analysis of evidence regarding the relationship between pancreatic exocrine insufficiency (PEI) and DM after AP.
The aim of this study was to conduct a systematic review and meta-analysis to determine the prevalence and time course of new onset PEI in patients with newly diagnosed prediabetes or DM after AP.
The search strategy was developed to identify all studies that investigated endocrine and exocrine function after AP. An electronic search of three major biomedical journal databases: (EMBASE, MEDLINE, and Scopus), was undertaken from inception (1980 for EMBASE, 1966 for Scopus, 1946 for MEDLINE) until 17th November 2013.
Search strategy was as follows: EMBASE and MEDLINE: “acute pancreatitis” and (“endocrine insufficiency” or “endocrine function” or “pancreatitis function” or “diabetes mellitus” or “pre diabetic state” or “type 2 diabetes mellitus” OR “type 1 diabetes mellitus” OR “adult onset diabetes mellitus” OR “maturity onset diabetes” OR “non-insulin dependent diabetes” OR “insulin dependent diabetes” OR “glucose intolerance” OR “glucose homeostasis”).
Scopus: “acute pancreatitis” AND (“endocrine insufficiency” or “endocrine function” or “pancreatitis function” or “diabetes mellitus” or “pre diabetic state” or “type 2 diabetes mellitus” or “type 1 diabetes mellitus” or “adult onset diabetes mellitus” OR “maturity onset diabetes” or “non-insulin dependent diabetes” or “insulin dependent diabetes” or “glucose intolerance” or “glucose homeostasis”) and [LIMIT-TO (Exact Keyword, “Human”)].
There were no language constraints but the search was limited to human studies. A full text review was performed looking for papers that investigated both exocrine and endocrine function. Bibliographies of articles chosen for full text review were also screened for relevant articles. Corresponding authors were contacted, if necessary.
Articles were screened for eligibility by two authors independently (JICK, SLMD) and any disagreement over study inclusion or exclusion was resolved by the senior author (MSP).
Studies were included if they met the following criteria for inclusion: (1) study design: cohort study; (2) population: individuals after an attack of AP; and (3) outcomes studied: both pancreatic endocrine and exocrine function.
Studies were excluded for the following reasons: studies did not use the standard definition of AP[11] (the presence of two of the following three features: abdominal pain characteristic of AP, serum amylase and/or lipase three times the upper limit of normal and/or characteristic findings of AP on CT scan), studied population was limited to patients with chronic, autoimmune or hereditary pancreatitis or gestational diabetes. In addition to this, patients who had previous history of prediabetes/DM or PEI, or history suggesting CP were excluded from analysis.
Prediabetes was defined by specific fasting blood glucose and/or 2 h post-oral glucose tolerance test criteria as reported by the study authors. DM was defined as reported by the authors of primary studies by specific fasting blood glucose testing and/or 2 h post-oral glucose tolerance test criteria and/or treatment with insulin, oral hypoglycaemia agents or specific dietary management. If raw data had been available in primary studies or through correspondence with authors, prediabetes or DM was defined using the 1999 WHO (World Health Organization) criteria to ensure consistency in reporting. PEI was defined by the primary authors and this included the use of both specific exocrine testing (secretin-caerulein 2 h infusion, serum pancreoauryl testing, faecal elastase, and faecal fat testing) and patient-reported need for enzyme supplementation and/or steatorrhoea. The “time course” was defined as the prevalence of concomitant PEI at specific mean follow up time points as stated by the authors of primary studies.
The quality of studies was assessed by the Newcastle-Ottawa scale[12]. It allocates a maximum of nine points based on three aspects study design: patient selection, comparability of study groups, and exposure and outcome of study participants. Studies were considered to be of high quality if 5 or more points are scored, and of low quality if less than 5 points are scored[13].
Data were extracted from the relevant articles using a standardised data extraction sheet under the following variables: country, year of publication, number of participants, study design, study period, criteria to define AP, exclusion criteria, aetiology, severity of AP, surgical intervention, if applicable, average age, gender composition, endocrine insufficiency and test used, PEI and test used, and prevalence of concomitant insufficiency.
Statistical Analysis was performed via Microsoft Excel (Windows) and StatsDirect for Windows V.3.0.97.0 (Stats Direct Ltd.). Studies were combined to determine the pooled prevalence and corresponding 95% confidence intervals. A random effects model was used to provide the most conservative estimate. Statistical significance was defined as P-values < 0.05. Statistical heterogeneity between the studies was assessed using the I2 metric with cut-offs of 50% and 75% to define low, moderate and high heterogeneity, respectively. The Harbord test was used to assess publication bias. Corresponding 95% confidence intervals were indicated in both cases. Subgroup analyses were performed with patients who developed either prediabetes alone or DM alone after AP. Subgroup analysis looking at the time course of PEI in patients with prediabetes or DM after AP was also performed.
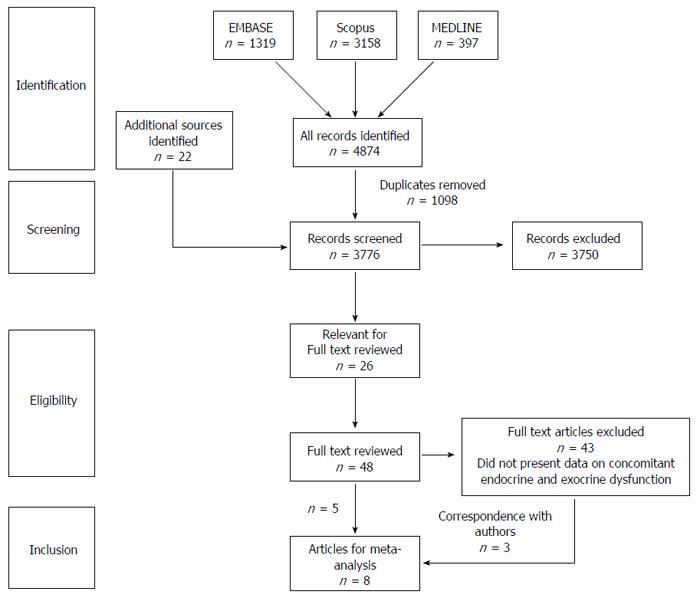
The search strategy identified 48 papers which investigated both pancreatic exocrine and endocrine function. Forty-three of these were subsequently excluded as they did not report on both exocrine and endocrine insufficiency concomitantly (Figure 1). However, a further 3 studies were able to be included after correspondence with the respective authors. Therefore, a total of 8 cohort studies comprising of 234 patients were included. Baseline characteristics of the patients are shown in Table 1.
| Ref. | No. of patients | Sex | Age | Aetiology | Time to follow up (mo) | |||
| Male | Female | Biliary | Alcohol | Other | ||||
| Andersson et al[14] | 41 | 16 | 25 | 61 (48-68)1 | 20 | 10 | 11 | 42 (36-53)1 |
| Angelini et al[15] | 27 | 24 | 3 | Not Stated | 10 | 11 | 6 | 12-60 |
| Bavare et al[16] | 18 | 18 | 0 | 36 (25-47)1 | 4 | 10 | 4 | 12-18 |
| Endlicher et al[17] | 9 | 5 | 4 | 44 (19-69)1 | 4 | 2 | 3 | 30 (15-52)2 |
| Gupta et al[18] | 30 | 24 | 6 | 37.5 (14-65)2 | 12 | 10 | 8 | 31.3 (7-118)2 |
| Hochman et al[19] | 25 | 16 | 9 | 58.8 (37-86)2 | 11 | 4 | 10 | 24-36 |
| Tsiotos et al[20] | 44 | 33 | 11 | 58 (20-93)2 | 17 | 5 | 22 | 60 (3-132)2 |
| Uomo et al[21] | 40 | 17 | 23 | 48.4 ± 18.23 | 28 | 0 | 12 | 179 (156-203)1 |
| Total | 234 | 153 | 81 | 106 | 52 | 76 | ||
The Newcastle-Ottawa scale was applied to each of the studies. All studies gained > 5 points indicating that they are of high methodological quality (Table 2).
Andersson et al[14] studied 41 patients, 14 severe and 27 mild, approximately 42 mo after their AP event in Lund, Sweden. In non-diabetic patients, endocrine function was determined by an oral glucose tolerance test and the 1999 WHO (World Health Organization) criteria were used to diagnose prediabetes or DM. In addition to this, exocrine function was determined by faecal fat excretion testing (abnormal values > 200 mg/dL) as well as through patient questionnaire (steatorrhea and/or the need for enzyme supplementation) (Table 3).
| Ref. | No. of patients | No. with prediabetes or diabetes (%) | Total1 | No. with pancreatitic exocrine insufficiency (%) | Total1 | Concomitant insufficiency (%) | Total1 |
| Andersson et al[14] | 41 | 24 (58.5) | 41 | 9 (22.0) | 41 | 6 (14.6) | 41 |
| Angelini et al[15] | 27 | 9 (47.4) | 19 | 1 (7.1) | 14 | 1 (7.1) | 14 |
| Bavare et al[16] | 18 | 13 (72.2) | 18 | 5 (27.8) | 18 | 5 (27.8) | 18 |
| Endlicher et al[17] | 9 | 3 (50.0) | 6 | 7 (87.5) | 8 | 3 (60.0) | 5 |
| Gupta et al[18] | 30 | 12 (40.0) | 30 | 12 (40.0) | 30 | 8 (26.7) | 30 |
| Hochman et al[19] | 25 | 8 (32.0) | 25 | 5 (20.0) | 25 | 2 (8.0) | 25 |
| Tsiotos et al[20] | 44 | 16 (36.4) | 44 | 11 (25.0) | 44 | 5 (11.4) | 44 |
| Uomo et al[21] | 40 | 6 (15.8) | 38 | 9 (22.5) | 40 | 3 (7.9) | 38 |
| Total | 234 | 91 (41.2) | 221 | 59 (26.8) | 220 | 33 (15.3) | 215 |
Angelini et al[15] undertook long term follow up of 27 patients treated with necrosectomy in Verona, Italy, for acute necrotising pancreatitis at 12-60 mo post discharge. Patients underwent an oral glucose tolerance test and a secretin-caelurin 2 h infusion to determine endocrine and exocrine function, respectively. Abnormal values for the glucose tolerance test were not stated and for the secretin-caerulein test a personal control group was used to determine a lowest value for “normal” (Table 3).
Bavare et al[16] undertook long term follow up of 18 patients treated with necrosectomy in Mumbai, India, that survived for at least a month post-surgery. Patients underwent fasting blood glucose testing to determine endocrine pancreatic function and a measurement of faecal fat excretion to determine their pancreatic exocrine function. The values for abnormality were > 200 mg/dL and > 7 g/24 h, respectively (Table 3).
Endlicher et al[17] enrolled 9 patients admitted to their institution in Regensburg, Germany, who had infected pancreatic necrosis and catheter drainage as part of their initial treatment. Patients underwent a 75 g oral glucose tolerance test (abnormal: > 200 mg/dL at 120 min) and a serum pancreoauryl test to assess pancreatic endocrine and exocrine function, respectively. Cut-offs for the pancreoauryl test was 4.5 and 2.5 μg/mL to define normal, moderate and severe insufficiency, respectively (Table 3).
Gupta et al[18] studied 30 patients with severe pancreatitis in Chandigarh, India. Endocrine function was determined by fasting and postprandial blood glucose levels. Patients who had normal values underwent an oral glucose tolerance test. Abnormal values for this test were not discussed. Exocrine function was determined with a faecal fat and D-xylose excretion test. Abnormality was defined as > 7 g/24 h and < 20%, respectively (Table 3).
Hochman et al[19] used a self-reporting questionnaire to assess exocrine and endocrine function of patients with severe acute pancreatitis in three teaching hospitals affiliated with the University of Alberta, Canada. The reported diagnosis of DM and requirement for enzyme supplementation were used to determine endocrine and exocrine function, respectively (Table 3).
Tsiotos et al[20] conducted a prospective follow up study of 72 patients who had undergone necrosectomy in Minnesota, United States. Of the 54 patients who survived surgery and the initial postoperative period, 44 were able to be contacted on average 5 years post discharge. Patients were subjected to a fasting blood glucose test and a faecal fat excretion test to determine both their pancreatic endocrine and exocrine function, respectively. Abnormalities were defined as > 200 mg/dL and > 7 g/24 h, respectively (Table 3).
Uomo et al[21] studied 40 non-surgical patients in Naples, Italy, with acute necrotizing pancreatitis at 6 monthly intervals for the first 24 mo and then at 12 monthly intervals. Patients were followed for a median of 15 years. At each follow up visit an oral glucose tolerance test was performed if the individual had a fasting blood glucose level between 120-140 mg/dL and a spot test for faecal elastase (abnormal: < 200 μg/g stool) (Table 3).
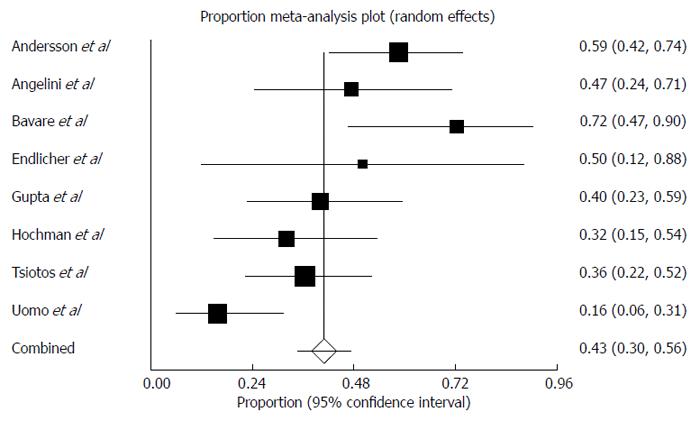
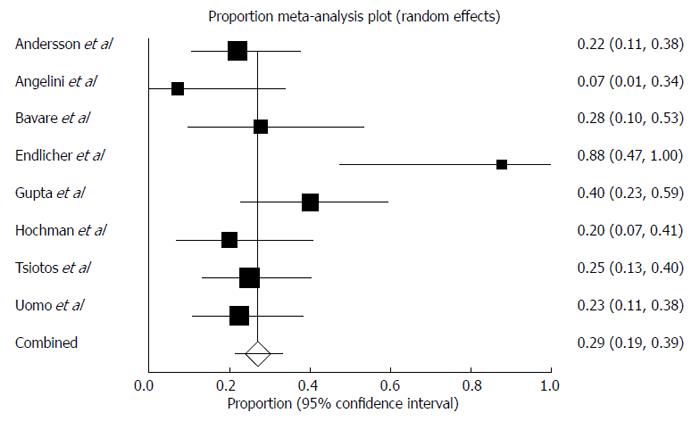
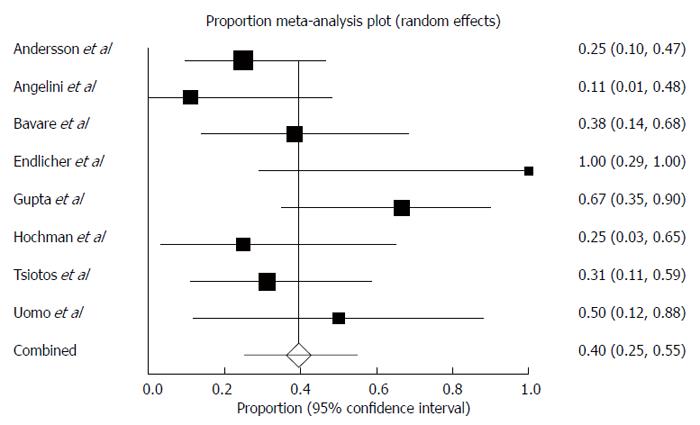
The pooled prevalence of newly diagnosed prediabetes/DM in patients with AP was 43% (Figure 2) (range of follow-up: 12-179 mo) and there was moderate statistical heterogeneity between studies (I2 = 72%). The pooled prevalence of newly diagnosed PEI in individuals after AP during this period of time was 29% (Figure 3) and there was moderate statistical heterogeneity between studies (I2 = 64%). There was no significant difference between the prevalence of newly diagnosed PEI after AP and the prevalence of newly diagnosed prediabetes/DM after AP (P = 0.93). In those individuals who developed prediabetes/DM, the prevalence of newly diagnosed concomitant PEI was 40% (Figure 4) and there was moderate statistical heterogeneity between studies (I2 = 54%).
The pooled prevalence of newly diagnosed prediabetes only in individuals after AP was 31% (22%-40%) and there was no statistical heterogeneity between studies (I2 = 0%). Among those individuals, the prevalence of newly diagnosed concomitant PEI was 41% (12%-75%) and there was moderate statistical heterogeneity between studies (I2 = 70%).
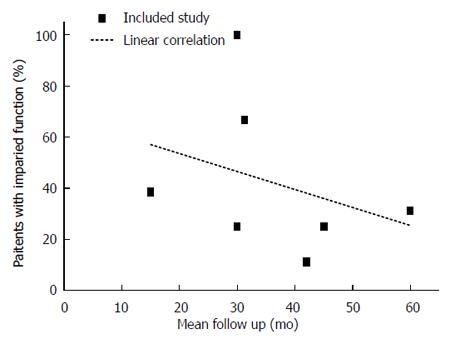
The pooled prevalence of newly diagnosed DM only in individuals after AP was 29% (18%-40%) and there was moderate statistical heterogeneity between studies (I2 = 70%). Among those individuals, the prevalence of newly diagnosed concomitant PEI was 39% (28%-51%) and there was no statistical heterogeneity between studies (I2 = 0%). There was no publication bias with any of the meta-analyses performed (data not shown). A time course analysis of the prevalence of PEI in patients with prediabetes/DM is presented in Figure 5.
This is the first systematic review and meta-analysis of the available clinical evidence related to pancreatic exocrine and endocrine function, both in isolation and combined, among individuals who have been discharged after an attack of AP. The prevalence of newly diagnosed PEI appears to be lower than the prevalence of newly diagnosed prediabetes or DM after AP, though there was no significant difference between the two. The prevalence of concomitant PEI in patients with newly diagnosed prediabetes or DM was 40% and it was very similar between individuals with prediabetes and those with DM. Also it appears that the prevalence of concomitant PEI in individuals with prediabetes or DM decreases with time, suggesting that pancreatic exocrine function after AP recovers with time as it is known that pancreatic endocrine function deteriorates with time[2].
Pancreatogenic DM is a recognised condition and is classified as a form of secondary (type 3c) DM by the American Diabetes Association and by the World Health Organization[22-24]. Pancreatic diseases underlying type 3c DM include acute, recurrent, and CP of any etiology, haemochromatosis, cystic fibrosis, fibrocalculous pancreatopathy, pancreatic trauma, pancreatectomy, pancreatic agenesis, and PC[24-26]. The prevalence and clinical importance of type 3c DM has been underestimated and underappreciated so far[27]. In a German study, it was estimated that only half of the cases of type 3c DM were classified correctly[23] and, on top of that, AP has recently been identified as a significant cause for DM[2]. It is likely that a larger proportion of patients with DM after AP are misclassified and further clinical characterization of such patients is needed to determine whether they have type 1, 2, or 3c DM. To classify pancreatogenic DM correctly, a robust definition is needed and the first attempt at diagnostic criteria has recently been published[27]. It has been proposed to diagnose pancreatogenic DM based on the presence of PEI (according to the fecal elastase-1 test or direct function tests) and pathological pancreatic imaging (endoscopic ultrasound, MRI, CT) together with an absence of type 1 DM autoimmune markers and markers of significant insulin resistance typical of type 2 diabetes (acanthosis nigricans, high fasting C-peptide or insulin).
Findings from the present study are highly relevant to this ongoing discussion on the diagnostic criteria for pancreatogenic DM. In particular, because concomitant PEI is only present in 40 percent of individuals with DM after AP, the reliance on this as one of the diagnostic criteria for type 3c DM may result in the misclassification of the majority of patients with DM after AP (assuming that individuals with DM after AP develop type 3c DM). But it is also worth acknowledging the possibility that AP is itself a new risk factor of type 2 DM. These diagnostic issues highlight the importance of investigating the pathogenesis of DM after AP. It is known that the hormonal profile is different for different types of DM, including insulin levels, glucagon level, pancreatic polypeptide, glucose dependent insulinotropic polypeptide, and glucagon-like peptide 1. The acknowledged difficulties in managing type 3c DM, often described as “brittle diabetes”, may derive from the combination of persistent hyperglycemia and exaggerated sensitivity to insulin[28] due to destruction of both alpha and beta cells reducing glucagon and insulin levels, respectively. There are no published data on whether DM after AP has features and the hormonal profile of type 1, 2, or 3c DM, highlighting a priority area for further research.
A structural relationship between the endocrine and exocrine pancreas was first described in 1882[29], when vascular connections between the endocrine islets and exocrine lobules were found in the Indian ink-injected rabbit pancreas. There is now strong evidence of a “portal” connection between the islet cells and the acini and it appears to be present in all mammals, including humans[30-33]. There is also some evidence to support the notion of a functional relationship between the endocrine and exocrine pancreas[34]. It has been observed that the pancreatic volume is reduced in both type 1 DM[35] and type 2 DM[36] patients compared with controls using ultrasonography[37] and CT scanning[38]. Autopsy studies have also demonstrated that type 1 DM is associated with pancreatic atrophy, increased fibrosis, and fatty infiltration[36]. Another subtle observation supporting a relationship between the islets and acinar cells is the focal severe acinar cell atrophy around insulin-deficient islets in patients with newly diagnosed type 1 DM, which is not found to be associated with normal insulin-containing islets[39].
The relationship between the exocrine and endocrine pancreas after AP has not been studied in the clinical setting but several animal studies in AP have demonstrated that there is a very intimate relationship between them[40]. These studies support the notion that insulin and probably other islet cell products have a key role in the regulation of the exocrine pancreas in animal models. Also, there is strong evidence that islet cells play the role of the gatekeeper of the pancreas by controlling the function and integrity of the pancreas and playing a significant role in metabolism[41,42]. For example, studies on arginine-induced pancreatitis in rats with DM have demonstrated that peri-insular acini remained intact during an acute attack of pancreatitis[43-45]. This phenomenon suggests that the close proximity of the islets of Langerhans may protect the acinar cells and might even accelerate the regenerative process. This may go some way to explain the finding of the present study that the majority of patients with DM do not have PEI. Further studies are needed to investigate the intricate relationship between the exocrine and endocrine pancreas after AP.
A particular strength of this systematic review has been the pooling of over 200 individuals from prospective high quality clinical studies. They have been discharged after an attack of AP and had the measurement of both exocrine and endocrine function. Individuals with known DM and PEI prior to their attack of AP were excluded from the study to enable reporting of the most conservative estimates of outcomes. The statistical analysis was robust with the use of random effects model to provide the most conservative estimates. There was also no language restriction in our search strategy, which reduced the language bias. Last, contact was made with the authors of primary studies if insufficient information was available from the published data. By doing so it was possible to include 3 more studies.
The present study has several limitations that need to be acknowledged. First, there was moderate statistical heterogeneity in some of our analyses. A possible explanation for this is the variety of pancreatic exocrine function assessment tools used across the studies. This included secretin-caerulein infusion testing, fecal elastase, fecal fat or D-xylose excretion testing, pancreoauryl testing, and self-reported questionnaires. It is also important to note that none of the primary studies used the secretin-pancreozymin test, which is regarded as the “gold standard” test for PEI[46,47]. Future studies will need to use more standardized methods of pancreatic exocrine function assessment to facilitate comparison of data. Second, only 5 out of 8 included studies focused on the first attack of AP and only 1 study reported on whether patients had repeated attacks of AP during follow up. Due to insufficient data it was not possible to account for the number of attacks of AP. Also, we were unable to control for other factors such as obesity and family history of DM. Similarly, we were unable to control for pre-existing patient factors that might contribute to PEI after AP. For example, some studies have shown that a hard pancreatic texture induced by obstructive pancreatitis can result in a higher incidence of PEI[48]. Last, the present study included patients that were treated conservatively as well as those who underwent surgery. Management of AP may affect pancreatic function[49] but, due to insufficient data, it was not possible to adjust for this possible confounder. In particular, it has been shown that distal pancreatectomy is associated with a higher rate of DM than other surgical procedures (pancreaticoduodenectomy or pancreatic drainage) in patients with CP[50]. This is consistent with the distribution of islets of Langerhans with higher population density in the body and tail of the pancreas. It is of some comfort though that the prevalence of new onset DM in the present study is similar to that reported in a recent systematic review of AP patients who received mainly conservative treatment[2].
In conclusion, this systematic literature review confirms that changes of pancreatic exocrine and endocrine functions are common after AP. The majority (60%) of patients with DM after AP do not have a concomitant PEI. This review also suggests that pancreatic exocrine function after an attack of AP recovers with time, at least in patients with prediabetes or DM. The relationship between the exocrine and endocrine pancreas is complex and the elucidation of the pathogenesis and subtype of DM that arises after AP is an important area for further research.
The authors would like to thank Dr Bodil Andersson (Lund University hospital, Sweden), Dr Rajesh Gupta (Postgraduate Institute of Medical Education and Research, India), and Dr David Hochman (University of Alberta Hospital, Canada) for providing additional information on their studies.
Pancreatogenic diabetes is classified as a form of secondary diabetes by the American Diabetes Association and by the World Health Organization. Accumulating evidence demonstrates that pancreatogenic diabetes accounts for at least 10% of diabetes in the western population. Further, this estimate is based on the studies focused on new onset diabetes after chronic pancreatitis and pancreatic cancer, but not acute pancreatitis. Given that acute pancreatitis is the most frequent disease of the pancreas and taking into account the recently emerged evidence of high incidence of diabetes after acute pancreatitis, it is very likely that the relative contribution of pancreatogenic diabetes has actually been underestimated in the literature.
Studies have shown that pancreatic exocrine insufficiency is common after chronic pancreatitis. Whether this holds true for patients with diabetes after acute pancreatitis remains unknown as, to date, no study has systematically assessed the relationship between the pancreatic exocrine and endocrine functions in patients after acute pancreatitis.
Concomitant pancreatic exocrine insufficiency occurs in 40% of patients with prediabetes or diabetes and its prevalence decreases with time.
Pancreatic exocrine insufficiency should not be used as a criterion for pancreatogenic diabetes after acute pancreatitis. Further studies are needed to investigate the pathogenesis of pancreatogenic diabetes.
Pancreatic exocrine insufficiency is a deficiency of the exocrine pancreatic enzymes, resulting in the inability to digest food properly.
This is a well-constructed meta-analysis and appropriate subject matter for review. Certainly the impairment of pancreatic endocrine function that develops as a result of primary pancreatic disease such as acute/chronic pancreatitis or after pancreatic surgery has been studied extensively. There are less rigorous studies on the development of pancreatic exocrine dysfunction after these disease processes, possibly due to the wide variety of pancreatic exocrine function assessment tools used (compared to the more rigorous definitions of diabetes) or the fact that, truthfully, many physicians tend to treat with enzyme supplementation based on patient self-report of symptoms rather than true diagnostic testing. There are even fewer reports still of combined endocrine/exocrine insufficiency after pancreatitis and therefore, despite the moderate statistical heterogeneity in some of the analyses presented, this manuscript still sheds some light on the prevalence of exocrine insufficiency in patients with newly diagnosed diabetes and pre-diabetes. The discussion is well written with practical clinical relevance to the working diagnostic criteria for pancreatogenic diabetes as well as mechanistic insight into the functional relationship between the endocrine and exocrine pancreas. I appreciate that the authors were thoughtful in their acknowledgement of the limitations in this meta-analysis, particularly with regards to the different pancreatic exocrine function assessment tools used and that four of their studies included patients that underwent necrosectomy, which could account for a higher prevalence of exocrine insufficiency - these are both important points that were addressed.
P- Reviewer: Ojeda AG, Phillip V, Visser BC, Xia SH, Zerem E S- Editor: Qi Y L- Editor: A E- Editor: Zhang DN
| 1. | Alsamarrai A, Das SL, Windsor JA, Petrov MS. Factors that affect risk for pancreatic disease in the general population: a systematic review and meta-analysis of prospective cohort studies. Clin Gastroenterol Hepatol. 2014;12:1635-1644.e5; quiz e103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 98] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 2. | Das SL, Singh PP, Phillips AR, Murphy R, Windsor JA, Petrov MS. Newly diagnosed diabetes mellitus after acute pancreatitis: a systematic review and meta-analysis. Gut. 2014;63:818-831. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 211] [Cited by in RCA: 276] [Article Influence: 25.1] [Reference Citation Analysis (0)] |
| 3. | Ewald N, Raspe A, Kaufmann C, Bretzel RG, Kloer HU, Hardt PD. Determinants of exocrine pancreatic function as measured by fecal elastase-1 concentrations (FEC) in patients with diabetes mellitus. Eur J Med Res. 2009;14:118-122. [PubMed] |
| 4. | Hardt PD, Krauss A, Bretz L, Porsch-Ozcürümez M, Schnell-Kretschmer H, Mäser E, Bretzel RG, Zekhorn T, Klör HU. Pancreatic exocrine function in patients with type 1 and type 2 diabetes mellitus. Acta Diabetol. 2000;37:105-110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 107] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 5. | Hardt PD, Killinger A, Nalop J, Schnell-Kretschmer H, Zekorn T, Klör HU. Chronic pancreatitis and diabetes mellitus. A retrospective analysis of 156 ERCP investigations in patients with insulin-dependent and non-insulin-dependent diabetes mellitus. Pancreatology. 2002;2:30-33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 40] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 6. | Hardt PD, Ewald N. Exocrine pancreatic insufficiency in diabetes mellitus: a complication of diabetic neuropathy or a different type of diabetes? Exp Diabetes Res. 2011;2011:761950. [PubMed] |
| 7. | Icks A, Haastert B, Giani G, Rathmann W. Low fecal elastase-1 in type I diabetes mellitus. Z Gastroenterol. 2001;39:823-830. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 52] [Article Influence: 2.2] [Reference Citation Analysis (1)] |
| 8. | Löhr M, Klöppel G. Residual insulin positivity and pancreatic atrophy in relation to duration of chronic type 1 (insulin-dependent) diabetes mellitus and microangiopathy. Diabetologia. 1987;30:757-762. [PubMed] |
| 9. | Pollard HM, Miller L, Brewer WA. The external secretion of the pancreas and diabetes mellitus. Am J Digest Dis. 1943;10:20-23. [RCA] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 33] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 10. | Rathmann W, Haastert B, Icks A, Giani G, Hennings S, Mitchell J, Curran S, Wareham NJ. Low faecal elastase 1 concentrations in type 2 diabetes mellitus. Scand J Gastroenterol. 2001;36:1056-1061. [PubMed] |
| 11. | Dellinger EP, Forsmark CE, Layer P, Lévy P, Maraví-Poma E, Petrov MS, Shimosegawa T, Siriwardena AK, Uomo G, Whitcomb DC. Determinant-based classification of acute pancreatitis severity: an international multidisciplinary consultation. Ann Surg. 2012;256:875-880. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 421] [Cited by in RCA: 342] [Article Influence: 26.3] [Reference Citation Analysis (1)] |
| 12. | Wells G, Shea B, O’Connell D, Peterson J, Welch V, Losos M, Tugwell P. The Newcastle-Ottawa Scale (NOS) for assessing the quality of non-randomized studies in meta-analysis. 2014. Available from: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp. |
| 13. | Halpin SJ, Ford AC. Prevalence of symptoms meeting criteria for irritable bowel syndrome in inflammatory bowel disease: systematic review and meta-analysis. Am J Gastroenterol. 2012;107:1474-1482. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 483] [Cited by in RCA: 456] [Article Influence: 35.1] [Reference Citation Analysis (0)] |
| 14. | Andersson B, Pendse ML, Andersson R. Pancreatic function, quality of life and costs at long-term follow-up after acute pancreatitis. World J Gastroenterol. 2010;16:4944-4951. [PubMed] |
| 15. | Angelini G, Pederzoli P, Caliari S, Fratton S, Brocco G, Marzoli G, Bovo P, Cavallini G, Scuro LA. Long-term outcome of acute necrohemorrhagic pancreatitis. A 4-year follow-up. Digestion. 1984;30:131-137. [PubMed] |
| 16. | Bavare C, Prabhu R, Supe A. Early morphological and functional changes in pancreas following necrosectomy for acute severe necrotizing pancreatitis. Indian J Gastroenterol. 2004;23:203-205. [PubMed] |
| 17. | Endlicher E, Völk M, Feuerbach S, Schölmerich J, Schäffler A, Messmann H. Long-term follow-up of patients with necrotizing pancreatitis treated by percutaneous necrosectomy. Hepatogastroenterology. 2003;50:2225-2228. [PubMed] |
| 18. | Gupta R, Wig JD, Bhasin DK, Singh P, Suri S, Kang M, Rana SS, Rana S. Severe acute pancreatitis: the life after. J Gastrointest Surg. 2009;13:1328-1336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 39] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 19. | Hochman D, Louie B, Bailey R. Determination of patient quality of life following severe acute pancreatitis. Can J Surg. 2006;49:101-106. [PubMed] |
| 20. | Tsiotos GG, Luque-de León E, Sarr MG. Long-term outcome of necrotizing pancreatitis treated by necrosectomy. Br J Surg. 1998;85:1650-1653. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 104] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 21. | Uomo G, Gallucci F, Madrid E, Miraglia S, Manes G, Rabitti PG. Pancreatic functional impairment following acute necrotizing pancreatitis: long-term outcome of a non-surgically treated series. Dig Liver Dis. 2010;42:149-152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 32] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 22. | Kuzuya T. Early diagnosis, early treatment and the new diagnostic criteria of diabetes mellitus. Br J Nutr. 2000;84 Suppl 2:S177-S181. [PubMed] |
| 23. | Ewald N, Kaufmann C, Raspe A, Kloer HU, Bretzel RG, Hardt PD. Prevalence of diabetes mellitus secondary to pancreatic diseases (type 3c). Diabetes Metab Res Rev. 2012;28:338-342. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 185] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 24. | Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Report of the expert committee on the diagnosis and classification of diabetes mellitus. Diabetes Care. 2003;26 Suppl 1:S5-20. [PubMed] |
| 25. | American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care. 2014;37 Suppl 1:S81-S90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2986] [Cited by in RCA: 3449] [Article Influence: 313.5] [Reference Citation Analysis (16)] |
| 26. | Tiengo A, Del Prato S, Briani G, Trevisan R, De Kreutzenberg S. [Acute and chronic complications of diabetes secondary to pancreatopathies]. Journ Annu Diabetol Hotel Dieu. 1995;179-189. [PubMed] |
| 27. | Ewald N, Bretzel RG. Diabetes mellitus secondary to pancreatic diseases (Type 3c)--are we neglecting an important disease? Eur J Intern Med. 2013;24:203-206. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 113] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 28. | Cui Y, Andersen DK. Pancreatogenic diabetes: special considerations for management. Pancreatology. 2011;11:279-294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 175] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 29. | Kuhne W, Lea A. Beobachtung uber die Absonderung des Pankreas. Unters Physiol Inst Univ Heidelberg. 1882;2:448-487. |
| 31. | Murakami T, Fujita T, Taguchi T, Nonaka Y, Orita K. The blood vascular bed of the human pancreas, with special reference to the insulo-acinar portal system. Scanning electron microscopy of corrosion casts. Arch Histol Cytol. 1992;55:381-395. [PubMed] |
| 32. | Murakami T, Hitomi S, Ohtsuka A, Taguchi T, Fujita T. Pancreatic insulo-acinar portal systems in humans, rats, and some other mammals: scanning electron microscopy of vascular casts. Microsc Res Tech. 1997;37:478-488. [PubMed] |
| 33. | Thiel A. [Studies on vascular system of pancreatic lobule in various mammals with special reference to the capillary glomeruli of Islands of Langerhans]. Z Zellforsch Mikrosk Anat. 1954;39:339-372. [PubMed] |
| 34. | Phillips AR, Abu-Zidan FM, Bonham MJ, Cooper GJ, Windsor JA. Amylin and severe acute pancreatitis. Pancreas. 2000;20:105-106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 35. | Rahier J. The diabetic pancreas: pathologist’s view. The pathology fo the endocrine pancreas in diabetes. New York: Springer-Verlag 1988; 17-40. [DOI] [Full Text] |
| 36. | Rahier J, Goebbels RM, Henquin JC. Cellular composition of the human diabetic pancreas. Diabetologia. 1983;24:366-371. [PubMed] |
| 37. | Fonseca V, Berger LA, Beckett AG, Dandona P. Size of pancreas in diabetes mellitus: a study based on ultrasound. Br Med J (Clin Res Ed). 1985;291:1240-1241. [PubMed] |
| 38. | Gilbeau JP, Poncelet V, Libon E, Derue G, Heller FR. The density, contour, and thickness of the pancreas in diabetics: CT findings in 57 patients. AJR Am J Roentgenol. 1992;159:527-531. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 71] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 39. | Foulis AK, Stewart JA. The pancreas in recent-onset type 1 (insulin-dependent) diabetes mellitus: insulin content of islets, insulitis and associated changes in the exocrine acinar tissue. Diabetologia. 1984;26:456-461. [PubMed] |
| 40. | Czakó L, Hegyi P, Rakonczay Z, Wittmann T, Otsuki M. Interactions between the endocrine and exocrine pancreas and their clinical relevance. Pancreatology. 2009;9:351-359. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 82] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 41. | Pour PM, Standop J, Batra SK. Are islet cells the gatekeepers of the pancreas? Pancreatology. 2002;2:440-448. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 42. | Petrov MS. Abdominal fat: a key player in metabolic acute pancreatitis. Am J Gastroenterol. 2013;108:140-142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 39] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 43. | Hegyi P, Takács T, Jármay K, Nagy I, Czakó L, Lonovics J. Spontaneous and cholecystokinin-octapeptide-promoted regeneration of the pancreas following L-arginine-induced pancreatitis in rat. Int J Pancreatol. 1997;22:193-200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 16] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 44. | Hegyi P, Takács T, Tiszlavicz L, Czakó L, Lonovics J. Recovery of exocrine pancreas six months following pancreatitis induction with L-arginine in streptozotocin-diabetic rats. J Physiol Paris. 2000;94:51-55. [PubMed] |
| 45. | Takács T, Hegyi P, Jármay K, Czakó L, Góg C, Rakonczay Z, Németh J, Lonovics J. Cholecystokinin fails to promote pancreatic regeneration in diabetic rats following the induction of experimental pancreatitis. Pharmacol Res. 2001;44:363-372. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 46. | Lankisch PG, Schmidt I, König H, Lehnick D, Knollmann R, Löhr M, Liebe S. Faecal elastase 1: not helpful in diagnosing chronic pancreatitis associated with mild to moderate exocrine pancreatic insufficiency. Gut. 1998;42:551-554. [PubMed] |
| 47. | Lankisch PG, Andrén-Sandberg A. Standards for the diagnosis of chronic pancreatitis and for the evaluation of treatment. Int J Pancreatol. 1993;14:205-212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 20] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 48. | Nakamura H, Murakami Y, Uemura K, Hayashidani Y, Sudo T, Ohge H, Sueda T. Predictive factors for exocrine pancreatic insufficiency after pancreatoduodenectomy with pancreaticogastrostomy. J Gastrointest Surg. 2009;13:1321-1327. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 59] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 49. | Petrov MS, Zagainov VE. Influence of enteral versus parenteral nutrition on blood glucose control in acute pancreatitis: a systematic review. Clin Nutr. 2007;26:514-523. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 53] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 50. | Okuno G, Oki A, Kawakami F, Doi K, Baba S. Prevalence and clinical features of diabetes mellitus secondary to chronic pancreatitis in Japan; a study by questionnaire. Diabetes Res Clin Pract. 1990;10:65-71. [PubMed] |