Published online Nov 28, 2014. doi: 10.3748/wjg.v20.i44.16674
Revised: April 18, 2014
Accepted: May 25, 2014
Published online: November 28, 2014
Processing time: 287 Days and 19.4 Hours
AIM: To investigate the effects of Glytan on splanchnic hemodynamics and its reduction of portal pressure in portal hypertensive rats.
METHODS: Glytan (Ganluotong in Chinese), is composed of salvianolic acid B and diammonium glycyrrhizinate. Portal hypertension (PHT) was induced in the rats by common bile duct ligation (BDL). Hemodynamic studies were performed using the colored microsphere method. Radioimmunoassay (RIA) was used to determine endothelin (ET)-1 levels in the mesenteric circulation. Western blotting methods were used to investigate the effect of Glytan on ET A receptor (ETAR), ET B receptor (ETBR), endothelial NO synthase (eNOS), G-protein-coupled receptor kinase (GRK)2, and β-arrestin 2 expression in the mesentery. The mRNA of ETAR and ETBR was determined using real-time polymerase chain reaction.
RESULTS: Treatment with Glytan reduced portal pressure (PP) and portal territory blood flow (PTBF) and increased both mean arterial pressure (MAP) and splanchnic vascular resistance (SVR). Especially at 4 wk, PP decreased by about 40%, while MAP increased by 13%, SVR increased by 12%, and PTBF decreased by about 21%. The effect of blood flow reduction was greatest in the mesentery (about 33%) at 4 wk. The mesenteric circulation ET-1 levels of BDL rats were lower and negatively correlated with PP at 4 wk. Glytan can increase mesenteric ET-1 content and inhibit ETBR, eNOS, GRK2, and β-arrestin 2 expression in the mesentery. Moreover, Glytan showed no effect on the expression of ETAR protein and mRNA.
CONCLUSION: The decreased PP and PTBF observed after Glytan treatment were related to increased mesenteric vasoconstriction and increased receptor sensitivity to vasoconstrictor.
Core tip: The traditional Chinese medicine Glytan is composed of salvianolic acid B and diammonium glycyrrhizinate. Previous studies have shown that Glytan is a new preparation for portal hypertension. The present study indicated that decreases in portal pressure and portal territory blood flow observed after Glytan treatment in portal hypertensive rats were related to increased mesenteric endothelin-1 content and reduced endothelin B receptor, endothelial NO synthase, G-protein-coupled receptor kinase 2, and β-arrestin 2 expression, which may promote mesenteric vasoconstriction and increase receptor sensitivity to vasoconstrictors. These results suggest the therapeutic potential of Glytan in portal hypertension induced by liver cirrhosis.
-
Citation: Du QH, Han L, Jiang JJ, Xu Y, Li WH, Li PT, Wang XY, Jia X. Glytan decreases portal pressure
via mesentery vasoconstriction in portal hypertensive rats. World J Gastroenterol 2014; 20(44): 16674-16682 - URL: https://www.wjgnet.com/1007-9327/full/v20/i44/16674.htm
- DOI: https://dx.doi.org/10.3748/wjg.v20.i44.16674
Since portal hypertension (PHT) was first proposed, finding a perfect medicine for PHT to reduce the risk of bleeding from esophageal varices has been the focus in this field. Non-selective β-receptor blockers have been considered the only drugs suitable for long-term administration. However, only 30%-40% of patients achieve a good therapeutic outcome with non-selective β-receptor blockers, because of their contraindications or side effects[1].
Glytan, which is based on traditional Chinese medicine theory, is a new preparation for PHT. Glytan is composed of salvianolic acid B (SA-B) and diammonium glycyrrhizinate (DG). SA-B is one of the water-soluble compounds derived from Salvia miltiorrhiza Bunge (Danshen in Chinese), which is widely used for chronic liver diseases. DG is extracted and purified from liquorices (Gancao in Chinese). The liquorices exert an important function in the treatment of hepatitis because of their anti-inflammatory effects. Our previous work found that Glytan can reduce portal pressure (PP), improve liver function, and inhibit pseudolobule formation in rats with liver cirrhosis[2]. Notably, the compatibility of SA-B and DG inhibits the steroid-like effects of DG[2]. Based on its efficacy in decreasing PP, Glytan has been approved to begin stage II clinical trials for PHT treatment in China.
In accordance with Ohm’s law, PP depends on intrahepatic resistance and portal inflow. In cases of cirrhosis, both intrahepatic resistance and splanchnic blood flow are increased. The initiating factor is an increase in intrahepatic vascular resistance, whereas the increase in splanchnic blood flow is a secondary phenomenon that maintains or worsens the increased PP and gives rise to hyperdynamic circulation[3].
Hyperdynamic circulation is characteristic of progressive splanchnic vasodilatation[4]. In previous work from our laboratory, we observed that treatment with Glytan resulted in reduced mesenteric vasodilation. Thus, one aim of the present study was to verify whether this phenomenon was related to a decrease in PP.
A previous study demonstrated that endothelin (ET)-1 and ET B receptor (ETBR) may be one of the mechanisms of splanchnic vasodilatation[5]. In addition, the effects of ET-1 receptors are also affected by their responsiveness. ET receptors could be desensitized by phosphorylation through G-protein-coupled receptor kinases (GRKs) and binding to β-arrestin-2[6]. Thus far, seven types of GRKs have been cloned. GRK2 is the most likely of the GRKs to initiate the desensitization of human ETAR and ETBR[7]. ET signaling in arterial smooth muscle is tightly regulated by GRK2[8]. ETAR-bound β-arrestin 2 demonstrates a higher affinity than β-arrestin 1 and does not interact with visual arrestin[9]. Therefore, we investigated the effect of Glytan on ET-1 and its receptors to investigate its mechanism of decreasing PP.
Male Sprague-Dawley rats (about 250 g; Vital River Laboratory Animal Technology Co. Ltd., Beijing, China) underwent sham surgery or common bile duct ligation (BDL). The common bile ducts were exposed after median laparotomy and ligated twice. In each animal, the segment between the two ligations was resected, and the abdomen of the animal was sutured closed. Sham-operated rats served as controls. In these rats, the common bile duct was exposed, but no ligation or resection was performed. Seven animals were used in each group. All experimental procedures were conducted in accordance with the guidelines for the use of experimental animals and were approved by the Institutional Review Committee on Animal Care and Use at the Experimental Animal Centre of Beijing University of Chinese Medicine [Certificate of Conformity: SCXK (jing) 2012-0001].
The rats were divided into the sham, BDL and Glytan groups. Each group was examined at 2 and 4 wk. Glytan was provided by Beijing Huaxin Wanbang Medicine Technology Limited Company. The proportion of SA-B to DG in Glytan was 1:1. Before use, Glytan was diluted with distilled water. After 1 wk BDL, the Glytan group was treated with Glytan (25 mg/kg per day) by gavage. The dose of Glytan depended on the pharmacodynamic experimental results[2]. Rats in the sham and BDL groups were administered the same amount of distilled water by gavage.
After 2 or 4 wk following BDL, hemodynamic studies were performed under chloral hydrate anaesthesia (3.5 g/kg intraperitoneal injection). The left femoral artery and vein were cannulated with PE-50 catheters for the measurement of mean arterial pressure (MAP) and blood withdrawal. A PE-50 catheter was inserted into the portal vein to measure PP. Another PE-50 catheter was advanced via the right carotid artery into the left ventricle under pulse curve control. This catheter was used for microsphere application. The catheter in the femoral artery and portal vein was connected to a pressure transducer for blood pressure measurements.
A reference sample was obtained for 1 min at a rate of 0.65 mL/min using a continuous withdrawal pump; 300000 yellow microspheres (15 μm in diameter; Triton Technologies, San Diego, CA, United States) were suspended in 0.3 mL saline containing 0.05% Tween. Microspheres were injected into the left ventricle 10 s after the withdrawal pump had been started. Upon completion of the hemodynamic measurements, the animals were sacrificed and the stomach, intestine, colon, pancreas, mesentery, and spleen were resected. The tissues were weighed and minced with scissors. The tissue and blood samples were processed and the microspheres were recovered by sedimentation. The tissue samples and the blood reference were digested by the addition of alkaline digestion reagent containing 1 mol/L KOH. The tubes from the tissue and blood preparation steps were placed in a temperature-controlled laboratory oven, set to a maximum of 50 °C, and the tissue and blood samples were allowed to digest overnight. The digested tissue and blood samples were centrifuged. The color from the filtered microspheres was dissolved in 0.2 mL dimethylformamide (DMF), and the absorption was measured by spectrophotometry.
The hemodynamic parameters for portal circulation were calculated as follows. Portal territory blood flow (PTBF) was defined as the sum of the blood flow through the stomach, intestine, colon, pancreas, spleen, and mesentery. Splanchnic perfusion pressure was defined as the difference between MAP and PP. Splanchnic vascular resistance (SVR) was calculated from the ratio between splanchnic perfusion pressure and PTBF.
Blood was drawn from the superior mesenteric artery (SMA) for ET-1 analysis. The level of ET-1 was measured in the mesenteric circulation using a commercial RIA kit (PLA Institute of RIA, Beijing, China) according to the manufacturer’s protocol.
The mesentery was isolated and stored in liquid nitrogen for RNA detection by real-time PCR. Total RNA was isolated according to the TRIzol reagent instructions (Invitrogen, Carlsbad, CA, United States). The primers of ETAR and ETBR were designed by Primer Premier 5.0 with amplification products of 205, 212, and 183 bp. For ETAR, the upstream primer was 5′-GGT TCC CTC TTC ACT TAA GC-3′ and the downstream primer was 5′-GTG ACA ACA GCA ACA GAG G-3′. For ETBR, the upstream primer was 5-GAG CAA TCC TCA GAG GTG T-3′ and the downstream primer was 5′-GAC TGT TTT TCC TCA AAC GTT-3′. For β-actin, the upstream primer was 5′-AAC GAG CGG TTC CGA TGC CCT GAG-3′ and the downstream primer was 5′- TGT CGC CTT CAC CGT TCC AGTT-3′. β-Actin, an internal housekeeping gene, was used to normalize the differences in RNA isolation and RNA degradation. The abundance of mRNA was determined by real-time PCR. Real-time PCR was performed using the ABI 7700 sequence detector (Applied Biosystems, Foster City, CA, United States). PCR was performed in a volume of 25 μL SYBR Mix (Hangzhou Bioer Technology Co. Ltd.) containing 2 μL cDNA. The results are expressed as the number of cycles required to exceed a threshold (Ct value) at which the fluorescence signal exceeded a defined threshold. The difference in Ct values of the target gene and the endogenous control are expressed as negative ∆Ct values. Therefore, higher 2-∆Ct values denote higher mRNA levels.
For western blot analysis, samples of rat mesentery were homogenized in radioimmunoprecipitation assay (RIPA) lysis buffer containing 50 mmol/L Tris (pH 7.4), 150 mmol/L NaCl, 1% Triton X-100, 1% sodium deoxycholate, 0.1% SDS, 5 mmol/L EDTA, 1 mmol/L sodium orthovanadate, 20 mmol/L pepstatin A, 20 mmol/L leupeptin, and 1 mmol/L phenylmethanesulfonyl fluoride. The protein content of the cleared homogenates was assessed with a bicinchoninic acid (BCA) assay kit (Applygen, Beijing, China). After boiling with SDS sample buffer (Applygen), 50 μg protein per lane from each sample was subjected to SDS-PAGE (10% gels for GRK2 and 12.5% gels for ETAR, ETBR and β-arrestin 2). After blotting on polyvinylidene difluoride membrane (Millipore, Bedford, MA, United States), the membranes were probed with primary antibodies diluted in TBS containing blocking protein and 0.1% Tween, and left to incubate overnight at 4 °C. The following primary antibodies in the indicated dilutions were used: mouse anti-GRK2, 1:500 (Abcam, Cambridge, MA, United States); and rabbit anti-ETAR/ETBR/endothelial NO synthase (eNOS) and mouse anti-β-arrestin 2, 1:200 (Santa Cruz Biotechnology, Santa Cruz, CA, United States). Thereafter, the membranes were washed and incubated with the appropriate peroxidase-coupled secondary antibodies diluted 1:5000 in TBS containing blocking protein and 0.1% Tween for 45 min (goat anti-rabbit or goat anti-mouse; Jackson, West Grove, PA, United States). Detection was performed with enhanced chemiluminescence (Applygen).
All data are presented as the mean ± SD; statistical comparisons were performed using one-way analysis of variance. The results of the molecular assays represent the means of samples from at least five rats in each group. P < 0.05 was considered statistically significant.
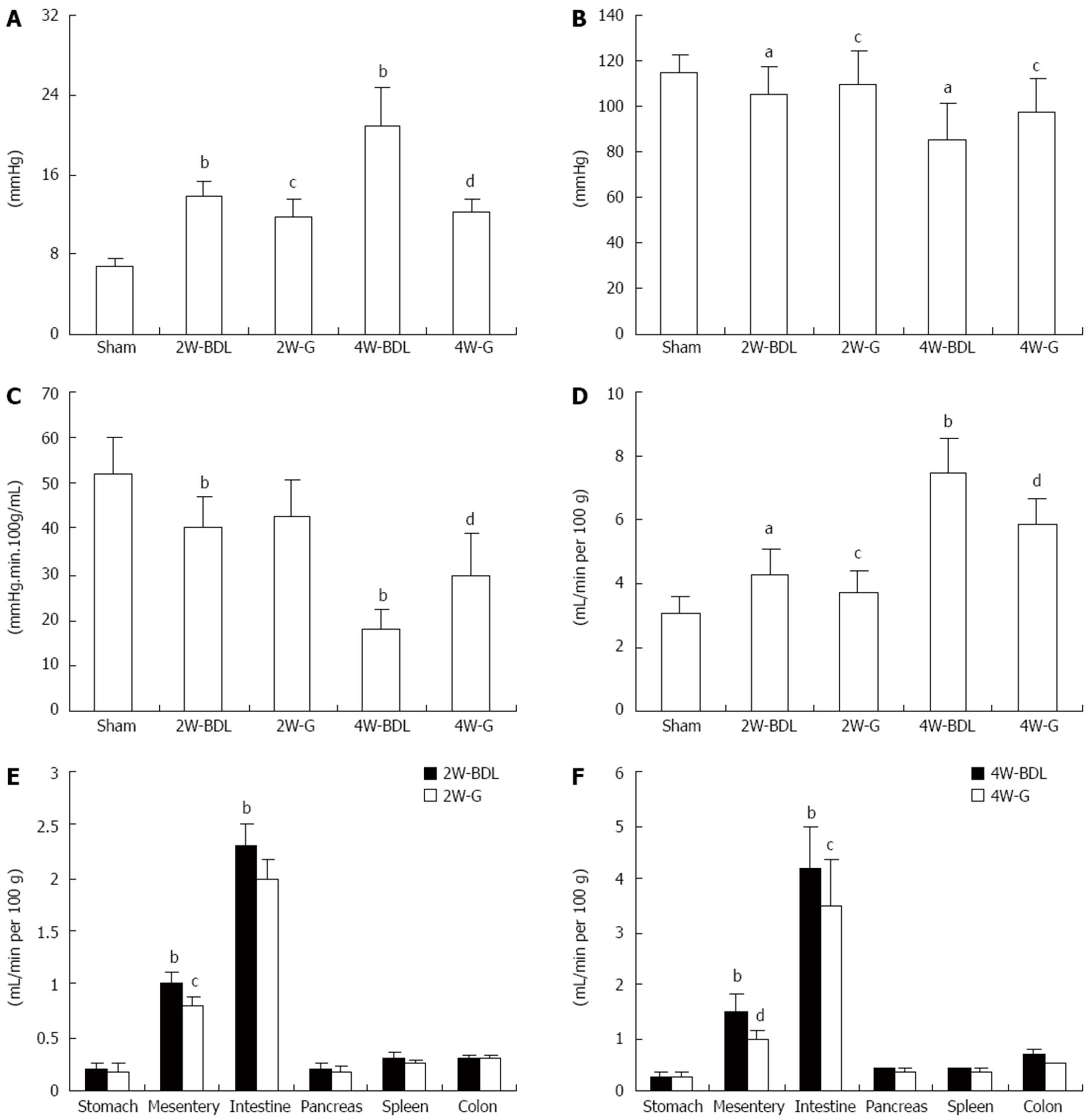
To assess whether Glytan can decrease PP and to determine its effects on splanchnic hemodynamics, we tested PP, MAP and various splanchnic hemodynamic parameters. The BDL rats showed increased PP and decreased MAP at 2 and 4 wk (Figure 1A and B). With the progression of PHT, characteristic splanchnic hemodynamic abnormalities appeared, including decreased SVR and increased PTBF (Figure 1C and D). Treatment with Glytan reduced PP and PTBF, while it increased MAP and SVR (Figure 1). Especially at 4 wk, PP was decreased by about 40%, while MAP increased by 13%, SVR increased by 12%, and PTBF decreased by about 21%. To determine which organs demonstrated decreased blood flow, we tested the blood flow of various organs, including the stomach, intestine, colon, pancreas, spleen, and mesentery. The blood flow of the intestine and mesentery decreased significantly at 4 wk, by 16% and 33%, respectively (Figure 1F). We investigated the mechanism behind the mesenteric-blood-flow-reducing properties of Glytan.
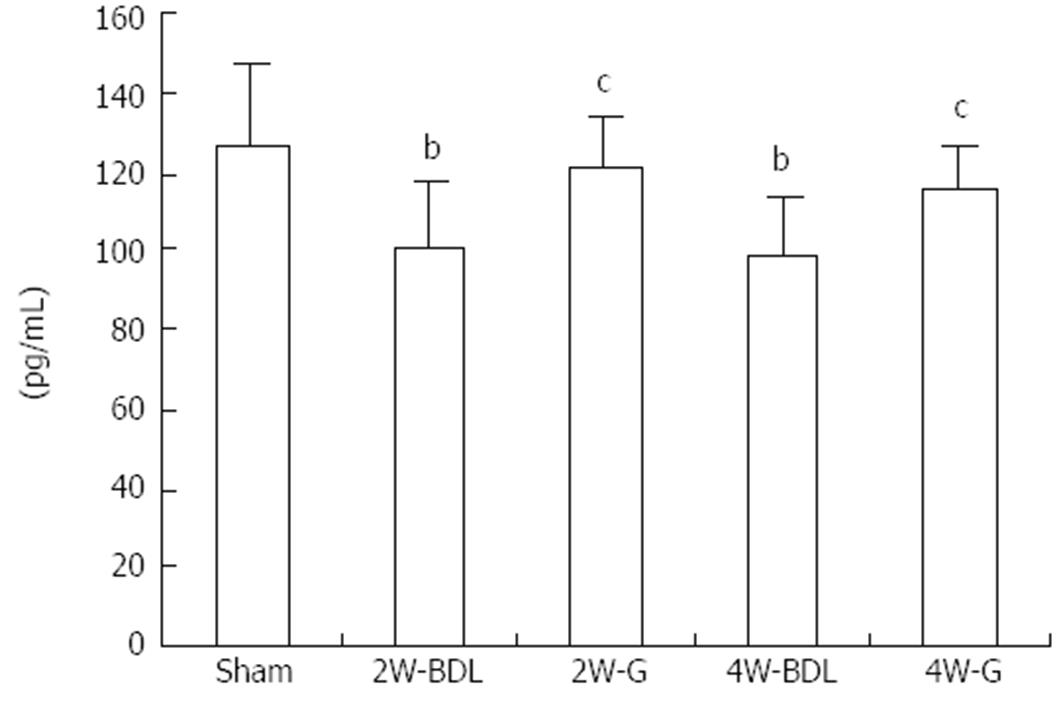
The concentration of ET-1 in the mesenteric circulation was measured by an RIA method (Figure 2). At 2 and 4 wk, ET-1 levels in the mesenteric circulation of BDL rats were significantly lower than those in sham group rats. After treatment with Glytan, ET-1 content was obviously increased at 4 wk.
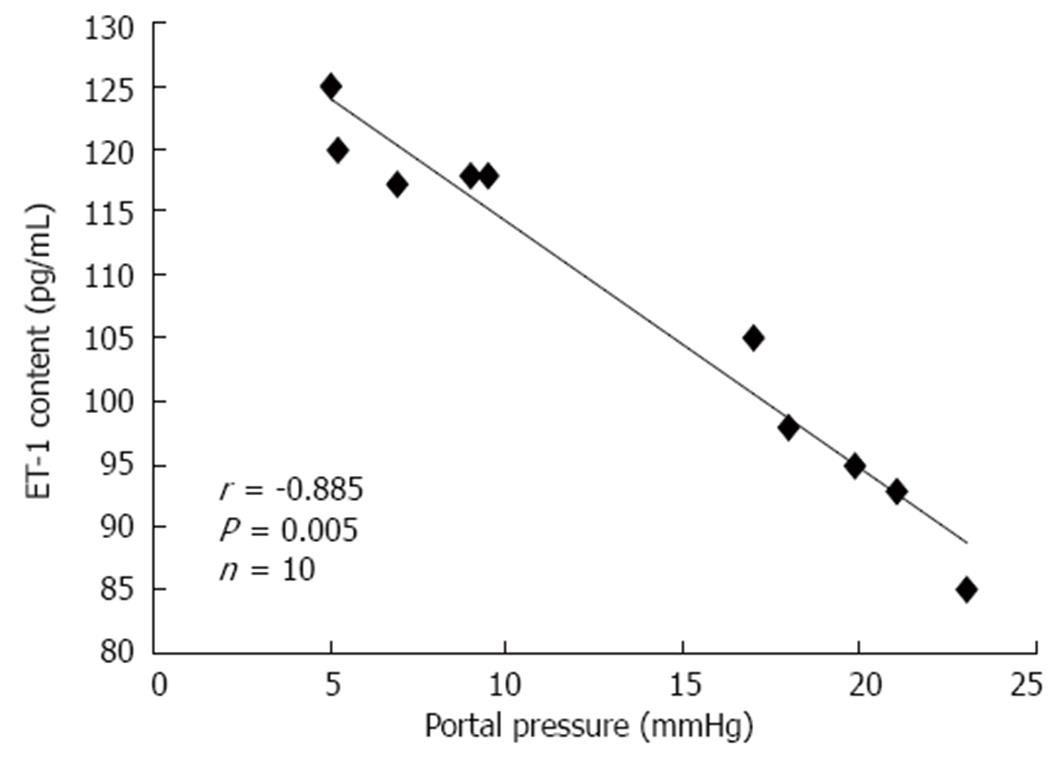
There was a significant negative correlation between PP and ET-1 levels in the mesenteric circulation at 4 wk (Figure 3). This correlation was not significant at 2 wk.
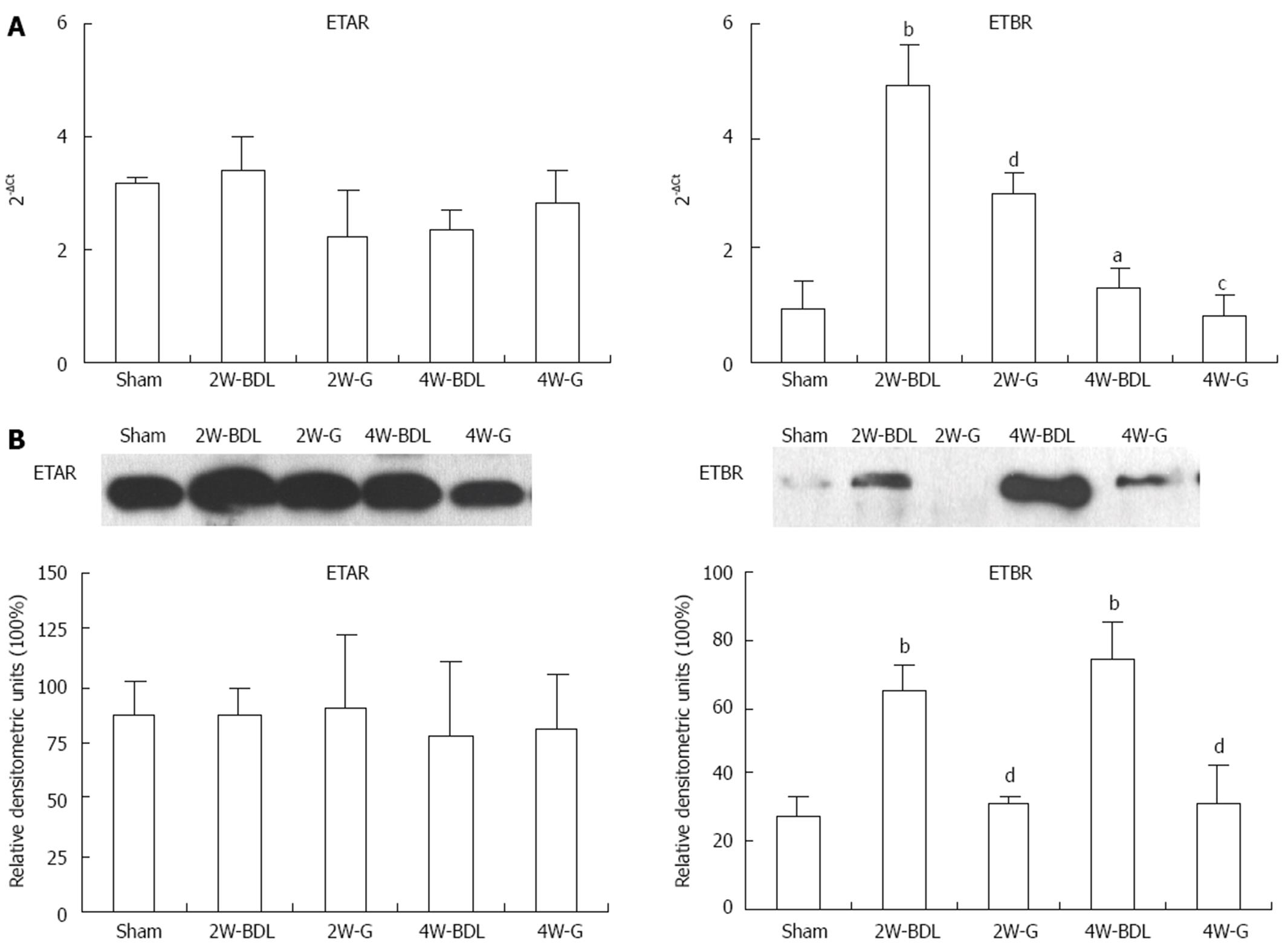
The effect of ET-1 on vascular tone depends on the receptor type. The protein and mRNA trends of ETBR in BDL rats differed (Figure 4A and B). ETBR mRNA was higher at 2 wk than that at 4 wk, while ETBR protein expression increased constantly over the two time points. Treatment with Glytan resulted in post-transcriptional and mRNA downregulation of ETBR expression in the mesentery (Figure 4). However, treatment with Glytan had no effect on ETAR expression at protein and mRNA levels (Figure 4).
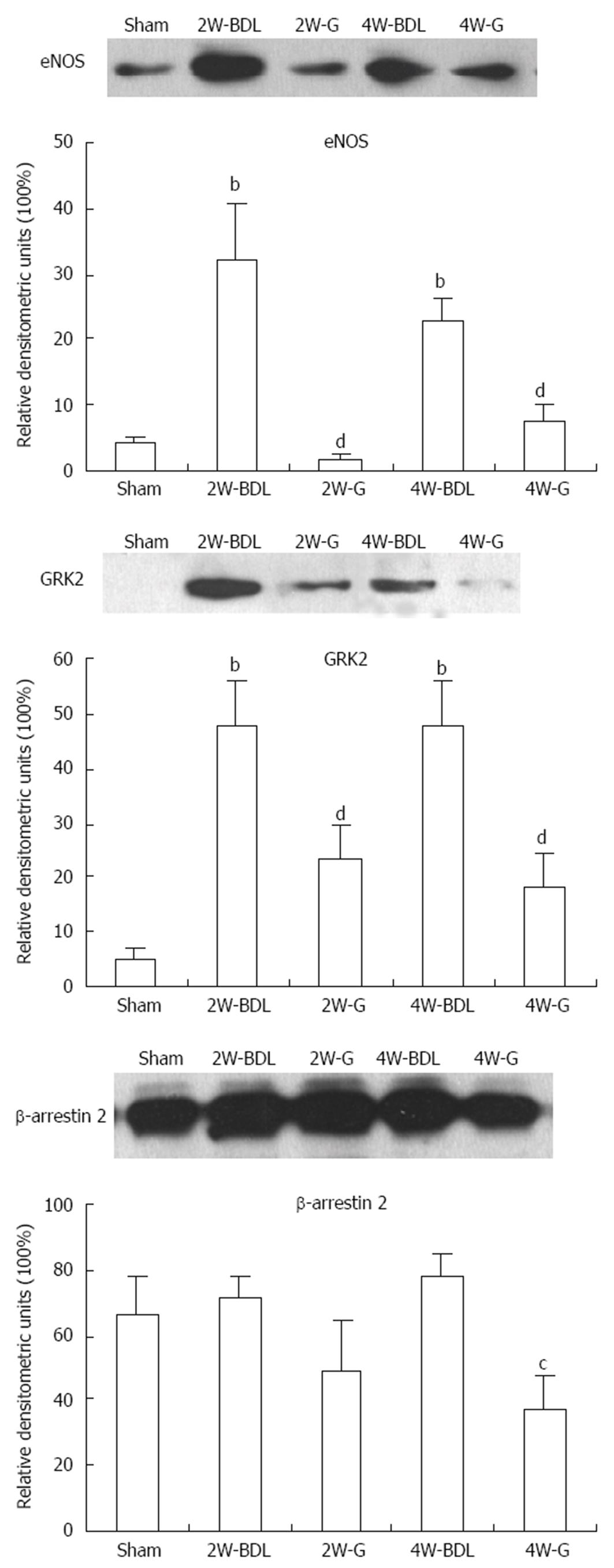
Expression of eNOS always implies the degree of vasodilation. β-arrestin 2 and GRK2 both affect ET receptor responsiveness. Treatment with Glytan significantly reduced eNOS and GRK2 expression at 2 and 4 wk. Glytan downregulated β-arrestin 2 expression at 4 wk (Figure 5).
We examined the effect of the Chinese medicine Glytan on splanchnic hemodynamics and the mechanisms of its effects. The results indicate that Glytan reduced PP and PTBF, while it increased MAP and SVR. The mesentery was the target organ of Glytan. On the one hand, Glytan increased ET-1 levels to promote vasoconstriction. On the other hand, Glytan inhibited mesenteric eNOS, ETBR and GRK2 expression to inhibit vasodilation.
In this study, the proportion of SA-B and DG was 1:1, which was proven by a compatibility proportion test[10]. Glytan was administered after 1 wk BDL. At 1 wk, the increased PP was not elevated to a level meeting the diagnostic criteria of PHT; therefore, this was a preventive study. Treatment with Glytan reduced PP and PTBF and increased SVR. Especially at 4 wk, PP was decreased by about 40% and PTBF was decreased by about 21%. Through analysis of blood flow of the splanchnic organs, we verified that the mesentery was the target organ of Glytan. Glytan reduced mesenteric blood flow by 33% at 4 wk, which is consistent with the decreased vasodilation of the mesenteric vascular bed observed in the present study. Next, we attempted to explain why Glytan reduced mesenteric blood flow and PP from the standpoint of ET-1 and its receptors.
ET-1 is one of the strongest vasoconstrictors. Changes in ET-1 levels in the splanchnic circulation are not consistent. Nagasue et al[11] found that ET-1 levels were increased in the mesenteric venous plasma. Vashist et al[12] found decreased SVR in PHT and that central hypovolemia stimulated sympathetic nerves, increasing vasoconstriction. However, Coll et al[13] found that gene and protein expression decreased in the SMA of rats with hepatic fibrosis and portal vein ligation. In our study, the ET-1 levels in the mesenteric circulation of BDL rats were significantly lower than those in sham group rats at 2 and 4 wk. ET-1 levels were negatively correlated with PP at 4 wk. Therefore, increased ET-1 levels resulting from Glytan administration may be a reason for the decrease observed in PP.
Excessive synthesis of NO in splanchnic tissue contributes to vasodilation in hyperdynamic circulation[14]. eNOS is the main source of NO[15]. Thus, inhibition of eNOS expression or activity in the visceral vascular system can effectively inhibit vascular dilatation. In addition, ET-1 binding to ETBR expressed in vascular endothelial cells activates the Gα and Gβγ subunits. Gβγ activates the PI3-Akt-eNOS pathway and promotes NO synthesis[16]. Therefore, either directly inhibiting eNOS expression or indirectly inhibiting ETBR expression can both reduce NO synthesis, in theory. Our results showed that eNOS expression was significantly increased in the mesenteric tissue of BDL rats. Increased eNOS not only resulted in mesenteric vasodilation but also induced a lower reactivity of the vasculature to vasoconstrictors[17-19]. Treatment with Glytan decreased eNOS significantly, which may be related to PP reductions.
The effect of ETBR in the mesentery is more complex. ET-1 binding to ETBR induces NO synthesis and thus vasodilation. ET-1 binding to ETAR causes vasoconstriction[20]. ETBR also acts in the clearance of ET-1[21]. With an increase in PP, ETBR expression increases in the mesenteric vasculature of BDL rats. It shows simultaneous stimulation of ETAR and ETBR in the rat renal microcirculation, and ETBR plays a major role in vasodilation[22]. In our study, increased ETBR in the mesentery of BDL rats may have promoted vasodilation to accommodate increased blood flow. The fact that Glytan decreases ETBR expression could mean two things: (1) it may inhibit vasodilation directly or indirectly through an ETBR-eNOS pathway; and (2) it may promote vasoconstriction through decreased clearance of ET-1. Because of increased levels of ET-1, ETAR can bind to more ET-1 to induce vasoconstriction.
Although NO is an important factor in splanchnic vascular dilation, NOS-deficient mice still demonstrate progressive splanchnic vasodilation. This indicates that NO is not the only factor related to hyperdynamic circulation[23,24]. Our study also found that eNOS expression was not increased with increases in PP. Therefore, a NO-independent pathway, such as hyporesponsiveness of the splanchnic vascular system to vasoconstrictors, cannot be ignored. In rats with portal vein ligation, the mesenteric artery and thoracic aorta show hyporesponsiveness to an adrenergic receptor[25]. In the aortas of rats with liver fibrosis and the hepatic arteries of human patients with liver cirrhosis, the numbers of angiotensin receptors are unchanged. However, AT1-R is desensitized by GRK2 and β-arrestin 2[26]. It is known that GRKs and arrestins are key participants in the canonical pathways leading to phosphorylation-dependent or independent G-protein-coupled receptor desensitization and endocytosis[27,28]. The difference between the phosphorylation-dependent and -independent pathways is that the former depends on arrestins, while the latter does not. Our previous results showed that increased PP resulted in upregulation of mesenteric GRK2 expression but not of β-arrestin 2[5]. In this study, treatment with Glytan resulted in decreased GRK2 and β-arrestin 2 expression in BDL rats. These results imply that the sensitivity of ETAR or other vasoconstrictor receptors to their agonists may increase. The exact sensitivity change in ET receptors by GRK2 and β-arrestin 2 after Glytan treatment needs to be verified through an immunoprecipitation test. In addition, Liu et al[29,30] have found that GRK2 expression increases in sinusoidal endothelial cells from portal hypertensive rats, while the knockout of GRK2 restores both Akt phosphorylation and NO production and normalizes PP. Therefore, in the liver, increased GRK2 promotes vasoconstriction. In the splanchnic organs, it remains unclear whether increased GRK2 has the same effect as in the liver. GRK2 promotes both vasoconstriction and vasodilation, therefore, the final effect of GRK2 is a balance of both functions.
In summary, treatment with Glytan reduced PP and PTBF, especially at 4 wk. The mesentery was the target organ of Glytan. Increased ET-1 levels and decreased ETBR and eNOS expression by Glytan may promote mesenteric vasoconstriction. Decreased GRK2 and β-arrestin 2 expression by Glytan may increase the sensitivity of vasoconstrictor receptors to their agonists and promote vasoconstriction. These findings suggest the therapeutic potential of Glytan for PHT.
Portal hypertension (PHT) is one of the most significant complications associated with liver cirrhosis, which can give rise to many other severe and often lethal conditions, such as bleeding esophageal varices, ascites, hepatic encephalopathy and hepatorenal syndrome. Non-selective β-receptor blockers have been considered the only drugs suitable for long-term administration. However, only 30%-40% of patients achieve a good therapeutic outcome with non-selective β-receptor blockers, because of their contraindications or side effects. Finding a perfect medicine for PHT to reduce the risk of bleeding from esophageal varices has been the focus in this field. Glytan is composed of salvianolic acid B (SA-B) and diammonium glycyrrhizinate (DG). SA-B is one of the water-soluble compounds derived from Salvia miltiorrhiza Bunge, which is widely used for chronic liver diseases. DG is extracted and purified from liquorices. The liquorices exert an important function in the treatment of hepatitis because of their anti-inflammatory effects. Our previous work found that Glytan can reduce portal pressure (PP), improve liver function, and inhibit pseudolobule formation in rats with liver cirrhosis. Notably, treatment with Glytan resulted in reduced mesenteric vasodilation. According to Ohm’s law, PP depends on intrahepatic resistance and portal inflow. An increase in splanchnic blood flow worsens and maintains PHT. Thus, the aim of this study was to verify whether reduced mesenteric vasodilation is related to a decrease in PP and to investigate the mechanisms by which Glytan decreases PP in portal hypertensive rats.
Bleeding from esophageal varices is the most serious complication of PHT. The most important goal of treating PHT is to reduce the risk of bleeding. It has been found that the incidence of varices at 5 years increased from 25% to 50% in patients with hepatic vein pressure gradient (HVPG) > 10 mmHg. Either non-selective β-receptor blockers or endoscopic band ligation can be used for primary prophylaxis in patients with cirrhosis and medium or large varices with high risk of bleeding. Both propranolol and carvedilol are non-selective β-receptor blockers. Carvedilol possesses both non-selective β1/2-antagonist and α1-receptor antagonist activity. Although carvedilol is more tolerated and highly effective than propranolol, it may cause arterial hypotension and worsen renal function. Traditional Chinese medicine is characteristic of multi-targets, which is consistent with the complicated pathophysiological features of PHT. In recent years, researchers have realized that it is feasible to develop drugs for PHT according to the progress of traditional Chinese medicine in the field of anti-fibrosis. Traditional Chinese medicine is the next battlefield for researchers to develop drugs for PHT.
Based on its efficacy in decreasing PP, Glytan is the first herbal compound that has been approved to begin stage II clinical trials in China. This study is believed to be the first to show that the mesentery is a target organ of Glytan. The decreased PP and portal territory blood flow (PTBF) observed after Glytan treatment are related to increased mesenteric vasoconstriction and increased endothelin (ET) receptor sensitivity to vasoconstrictors. This is also the first study to demonstrate the effect of Glytan on hyperdynamic circulation.
Based on the effect of Glytan on splanchnic hemodynamics and mesenteric ET receptors, this study suggests the therapeutic potential of Glytan on liver cirrhosis-induced PHT.
PHT is the main complication of cirrhosis and is defined as an HVPG > 5 mmHg. Clinically significant PHT is defined as HVPG ≥ 10 mmHg. Hyperdynamic circulation is a state of portal hypertension that is characterized by splanchnic and peripheral vasodilation, increased plasma volume, and increased cardiac output. Glytan is a new preparation for PHT, which is based on traditional Chinese medicine theory. Glytan is composed of SA-B and DG.
The manuscript discusses a very interesting topic. In addition, the manuscript is well written. It presents the results in a clear and well explained manner both in text and in figures.
P- Reviewer: Ranieri G S- Editor: Nan J L- Editor: Kerr C E- Editor: Zhang DN
| 1. | Villanueva C, Aracil C, Colomo A, Hernández-Gea V, López-Balaguer JM, Alvarez-Urturi C, Torras X, Balanzó J, Guarner C. Acute hemodynamic response to beta-blockers and prediction of long-term outcome in primary prophylaxis of variceal bleeding. Gastroenterology. 2009;137:119-128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 140] [Article Influence: 8.8] [Reference Citation Analysis (1)] |
| 2. | Du QH, Han L, Jiang JJ, Li PT, Wang XY, Tang HL. Preliminary study of Glytan on treating portal hypertension rats induced by biliary liver fibrosis. Zhonghua Zhongyiyao Zazhi. 2009;24:446-451. |
| 3. | Iwakiri Y. Endothelial dysfunction in the regulation of cirrhosis and portal hypertension. Liver Int. 2012;32:199-213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 132] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 4. | Iwakiri Y, Groszmann RJ. The hyperdynamic circulation of chronic liver diseases: from the patient to the molecule. Hepatology. 2006;43:S121-S131. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 418] [Cited by in RCA: 404] [Article Influence: 21.3] [Reference Citation Analysis (14)] |
| 5. | Du QH, Han L, Jiang JJ, Li PT, Wang XY, Jia X. Increased endothelin receptor B and G protein coupled kinase-2 in the mesentery of portal hypertensive rats. World J Gastroenterol. 2013;19:2065-2072. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 6] [Cited by in RCA: 5] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 6. | Gurevich EV, Tesmer JJ, Mushegian A, Gurevich VV. G protein-coupled receptor kinases: more than just kinases and not only for GPCRs. Pharmacol Ther. 2012;133:40-69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 16] [Reference Citation Analysis (0)] |
| 7. | Freedman NJ, Ament AS, Oppermann M, Stoffel RH, Exum ST, Lefkowitz RJ. Phosphorylation and desensitization of human endothelin A and B receptors. Evidence for G protein-coupled receptor kinase specificity. J Biol Chem. 1997;272:17734-17743. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 157] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 8. | Morris GE, Nelson CP, Standen NB, Challiss RA, Willets JM. Endothelin signalling in arterial smooth muscle is tightly regulated by G protein-coupled receptor kinase 2. Cardiovasc Res. 2010;85:424-433. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 55] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 9. | Oakley RH, Laporte SA, Holt JA, Caron MG, Barak LS. Differential affinities of visual arrestin, beta arrestin1, and beta arrestin2 for G protein-coupled receptors delineate two major classes of receptors. J Biol Chem. 2000;275:17201-17210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 667] [Cited by in RCA: 696] [Article Influence: 27.8] [Reference Citation Analysis (0)] |
| 10. | Liu SF, Cai DY, Li PT, Xiang PR. Study on Components and Compatibility of Medicines of You hua gan shen Compound in Resisting Hepatic Fibrosis. Zhonghua Zhongyiyao Zazhi. 2005;20:373-375. |
| 11. | Nagasue N, Dhar DK, Yamanoi A, Emi Y, Udagawa J, Yamamoto A, Tachibana M, Kubota H, Kohno H, Harada T. Production and release of endothelin-1 from the gut and spleen in portal hypertension due to cirrhosis. Hepatology. 2000;31:1107-1114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 46] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 12. | Vashist Y, Semela D, Dufour JF. Hyperdynamic circulation in liver cirrhosis: desensitization of vasoconstrictive receptors by G protein-coupled receptor kinases. Med Hypotheses. 2004;62:82-85. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 7] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 13. | Coll M, Martell M, Raurell I, Ezkurdia N, Cuenca S, Hernández-Losa J, Esteban R, Guardia J, Bosch J, Genescà J. Atrophy of mesenteric sympathetic innervation may contribute to splanchnic vasodilation in rat portal hypertension. Liver Int. 2010;30:593-602. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 18] [Reference Citation Analysis (0)] |
| 14. | Michielsen PP, Boeckxstaens GE, Sys SU, Herman AG, Pelckmans PA. Role of nitric oxide in hyporeactivity to noradrenaline of isolated aortic rings in portal hypertensive rats. Eur J Pharmacol. 1995;273:167-174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 21] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 15. | Niederberger M, Ginés P, Martin PY, Tsai P, Morris K, McMurtry I, Schrier RW. Comparison of vascular nitric oxide production and systemic hemodynamics in cirrhosis versus prehepatic portal hypertension in rats. Hepatology. 1996;24:947-951. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 82] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 16. | Liu S, Premont RT, Kontos CD, Huang J, Rockey DC. Endothelin-1 activates endothelial cell nitric-oxide synthase via heterotrimeric G-protein betagamma subunit signaling to protein jinase B/Akt. J Biol Chem. 2003;278:49929-49935. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 116] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 17. | Verbeke L, Farre R, Trebicka J, Komuta M, Roskams T, Klein S, Elst IV, Windmolders P, Vanuytsel T, Nevens F. Obeticholic acid, a farnesoid X receptor agonist, improves portal hypertension by two distinct pathways in cirrhotic rats. Hepatology. 2014;59:2286-2298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 191] [Cited by in RCA: 213] [Article Influence: 19.4] [Reference Citation Analysis (0)] |
| 18. | Langer DA, Shah VH. Nitric oxide and portal hypertension: interface of vasoreactivity and angiogenesis. J Hepatol. 2006;44:209-216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 48] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 19. | Wiest R, Groszmann RJ. The paradox of nitric oxide in cirrhosis and portal hypertension: too much, not enough. Hepatology. 2002;35:478-491. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 338] [Cited by in RCA: 308] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 20. | Clozel M, Gray GA, Breu V, Löffler BM, Osterwalder R. The endothelin ETB receptor mediates both vasodilation and vasoconstriction in vivo. Biochem Biophys Res Commun. 1992;186:867-873. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 318] [Cited by in RCA: 305] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 21. | Böhm F, Pernow J, Lindström J, Ahlborg G. ETA receptors mediate vasoconstriction, whereas ETB receptors clear endothelin-1 in the splanchnic and renal circulation of healthy men. Clin Sci (Lond). 2003;104:143-151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 20] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 22. | Just A, Olson AJ, Arendshorst WJ. Dual constrictor and dilator actions of ET(B) receptors in the rat renal microcirculation: interactions with ET(A) receptors. Am J Physiol Renal Physiol. 2004;286:F660-F668. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 61] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 23. | Iwakiri Y, Cadelina G, Sessa WC, Groszmann RJ. Mice with targeted deletion of eNOS develop hyperdynamic circulation associated with portal hypertension. Am J Physiol Gastrointest Liver Physiol. 2002;283:G1074-G1081. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 55] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 24. | Koshy A, De Gottardi A, Ledermann M, Saegesser H, Shaw SG, Zimmermann A, Reichen J. Endothelial nitric oxide synthase is not essential for the development of fibrosis and portal hypertension in bile duct ligated mice. Liver Int. 2005;25:1044-1052. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 25. | Bosch J, Abraldes JG, Fernández M, García-Pagán JC. Hepatic endothelial dysfunction and abnormal angiogenesis: new targets in the treatment of portal hypertension. J Hepatol. 2010;53:558-567. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 154] [Cited by in RCA: 152] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 26. | Hennenberg M, Trebicka J, Biecker E, Schepke M, Sauerbruch T, Heller J. Vascular dysfunction in human and rat cirrhosis: role of receptor-desensitizing and calcium-sensitizing proteins. Hepatology. 2007;45:495-506. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 61] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 27. | Reiter E, Lefkowitz RJ. GRKs and beta-arrestins: roles in receptor silencing, trafficking and signaling. Trends Endocrinol Metab. 2006;17:159-165. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 502] [Cited by in RCA: 524] [Article Influence: 27.6] [Reference Citation Analysis (0)] |
| 28. | Watari K, Nakaya M, Kurose H. Multiple functions of G protein-coupled receptor kinases. J Mol Signal. 2014;9:1. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 72] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 29. | Liu S, Premont RT, Kontos CD, Zhu S, Rockey DC. A crucial role for GRK2 in regulation of endothelial cell nitric oxide synthase function in portal hypertension. Nat Med. 2005;11:952-958. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 196] [Cited by in RCA: 195] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 30. | Liu S, Rockey DC. Cicletanine stimulates eNOS phosphorylation and NO production via Akt and MAP kinase/Erk signaling in sinusoidal endothelial cells. Am J Physiol Gastrointest Liver Physiol. 2013;305:G163-G171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 14] [Article Influence: 1.2] [Reference Citation Analysis (0)] |