Published online Nov 7, 2014. doi: 10.3748/wjg.v20.i41.15200
Revised: May 21, 2014
Accepted: June 14, 2014
Published online: November 7, 2014
Processing time: 239 Days and 0.8 Hours
Acute pancreatitis (AP) is one of the most common diseases of the gastrointestinal tract, bearing significant morbidity and mortality worldwide. Current treatment of AP remains unspecific and supportive and is mainly targeted to aggressively prevent systemic complications and organ failure by intensive care. As acute pancreatitis shares an indistinguishable profile of inflammation with sepsis, therapeutic approaches have turned towards modulating the systemic inflammatory response. Targets, among others, have included pro- and anti-inflammatory modulators, cytokines, chemokines, immune cells, adhesive molecules and platelets. Even though, initial results in experimental models have been encouraging, clinical implementation of immune-regulating therapies in acute pancreatitis has had a slow progress. Main reasons include difficulty in clinical translation of experimental data, poor understanding of inflammatory response time-course, flaws in experimental designs, need for multimodal approaches and commercial drawbacks. Whether immune-modulation in acute pancreatitis remains a fact or just fiction remains to be seen in the future.
Core tip: Acute pancreatitis is a common entity with significant mortality worldwide. Treatment remains non-specific and mainly supportive, mostly focusing on intensive care. Presence of inflammatory response syndrome during AP has driven recent immune-modulating therapeutic attempts in experimental models, including cytokine, chemokine, immune cell and other inflammatory mediator blockade. Although initial data are promising, translation to clinical routine has been less encouraging. The authors attempt to elucidate whether and to what extent tampering with the immune burst triggered by acute pancreatitis could actually ensure better outcomes, or that remains a farfetched expectation.
- Citation: Akinosoglou K, Gogos C. Immune-modulating therapy in acute pancreatitis: Fact or fiction. World J Gastroenterol 2014; 20(41): 15200-15215
- URL: https://www.wjgnet.com/1007-9327/full/v20/i41/15200.htm
- DOI: https://dx.doi.org/10.3748/wjg.v20.i41.15200
Acute pancreatitis (AP) is a common disease, posing a tremendous burden in health care systems globally[1,2]. The incidence of AP varies between 4.9 and 73.4 cases per 100000 worldwide[3,4]. Progression to multiple organ dysfunction syndrome (MODS), as a consequence of the systemic inflammatory response syndrome (SIRS) represents a major contributor to high mortality in the early phase of the disease[5,6]. Consequent breakdown of intestinal integrity, bacterial translocation and increased infection risk can further complicate outcome in the late phases of AP[7-9].
Current treatment of AP remains non-specific and supportive and is mainly targeted to aggressively prevent systemic complications by intensive care. During the last decade, a number of new therapeutic modalities have changed the management of acute pancreatitis, including enteral feeding in severe AP, the use of early antibiotic treatment in necrotizing pancreatitis and therapeutic endoscopic retrograde cholangiopancreatography (ERCP) with endoscopic sphincterotomy in severe biliary pancreatitis[10,11]. However, although the case fatality rate for AP has decreased over time, the overall population mortality rate has remained unchanged[12].
Recent data has come to show that in the early phase of AP, excessive leukocyte activation and consequent inflammatory mediator release are critical for development of early organ failure[13-16]. As a result, current experimental and clinical research has been driven by the need to inhibit the systemic inflammatory reaction thus; prevent the development of MODS. This article attempts to critically review recent data on immune-modulating strategies in AP.
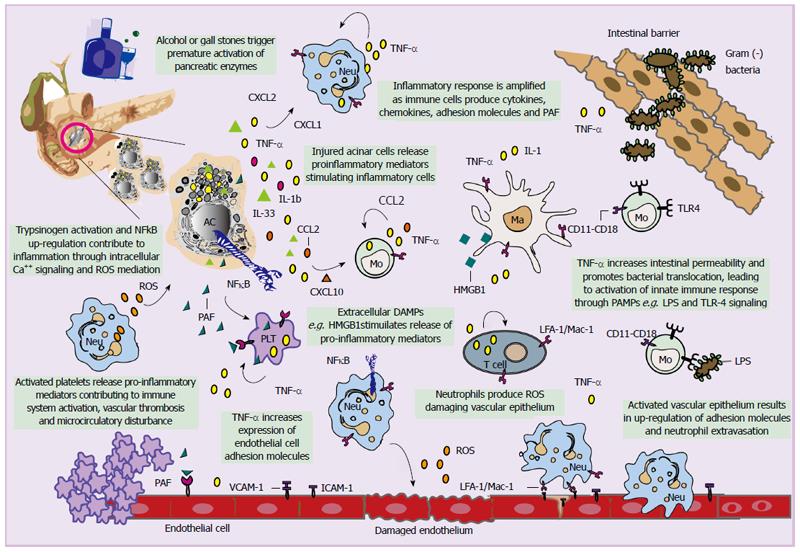
Acute pancreatitis represents an inflammatory disorder; hence, a complex cascade of immunologic events affects disease pathogenesis and progression. Alcohol and gallstones remain the major etiologic factors of AP. Irrespective of the cause, triggering events lead to premature activation of pancreatic proteases, as a result of intracellular co-localization with lysosomal enzymes[17,18]. An increase in intracellular calcium triggers activation of trypsinogen and induces local inflammation[19], further leading to auto-digestion, destruction of the parenchyma and finally necrosis of the pancreas[17,20-23](Figure 1).
The role of tumour necrosis factor alpha (TNF-α) in the potential activation of pancreas polypeptide and nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB)[24] has been recently investigated[25-29]. Whether there is a link between pancreatic NF-κB and trypsinogen activation remains controversial[27]. However, it seems that these processes may be unrelated and may both contribute to inflammation, possible through reactive O2 species (ROS) mediation and calcium (Ca2+) signalling[30,31] (Figure 1).
At this stage, it appears that the autophagy machinery interfaces with various cellular stress-response pathways including those involved in immune response and inflammation, entailing among others direct interactions between autophagy proteins and immune signalling molecules[32-34]. This complex interplay modulates both the induction and suppression of immune and inflammatory responses and vice versa, while it seems that the same genes that regulate autophagy are involved in xenophagy[35,36]. In this respect, a potential protective role for interleukin (IL)-22 against the autophagic pathway in pancreatitis is currently under investigation[37].
Endogenous molecules released as a result of tissue injury, referred to as damage-associated molecular patterns represent primary activators of the immune system[38-40]. Among those, high mobility group box 1 (HMGB1) protein, a nuclear DNA binding protein[41-43], has been recently suggested to act as a key mediator for inflammation and organ failure in AP[44-46]. Pancreatic-derived intracellular HMGB1 limits the severity of the disease by protecting cells from NF-κB activation, DNA damage, cell death, and release of nucleosomes from injured acinar cells[47]. On the other hand, extracellular HMGB1 released by necrotic cells, can, via members of the Toll-like receptor (TLR) family trigger acute lung injury[48,49] and a lethal systemic inflammatory process[50,51]. Extracellular HMGB1 can further stimulate the release of pro-inflammatory cytokines including TNF-α and IL-1β by inducing nuclear translocation of NF-κB and conversely, the pro-inflammatory cytokines can control further release of HMGB1 into the extracellular space (Figure 1)[52-54] .
Activated acinar cells also secrete pro-inflammatory factors including C-X-C motif chemokine (CXCL) 10, Chemokine (C-C motif) ligand 2 also referred to as monocyte chemotactic protein-1 (MCP-1), IL33[55,56], platelet activating factor (PAF), TNF-α and IL-1β leading to migration of monocytes and neutrophils into the pancreas[57,58]. Neutrophils are specifically activated by CXCL-1 and CXCL-2 (also called macrophage inflammatory protein 2-alpha, MIP2-α), while monocytes, eosinophils and T-cells are activated by CCL-2 (MCP-1) and CXCL-10[59] (Figure 1). However, monocyte and macrophage populations involved in AP are heterogeneous, with great phenotypic and functional plasticity[60]. Recently, a subtype of monocytes that derive from the bone marrow and express TNF-α has been identified, which appears to determine pancreatic oedema and acinar cell injury/necrosis[61]. T cells are also present in smaller numbers in the inflamed pancreas and appear to be necessary for progression of AP[62]. As AP progresses, changes in the number and ratio of CD4+ and CD8+ T cells has been noted, probably because CD4+ T cells contribute to activation of macrophage via antigen presentation and release of inflammatory cytokines[63]. In contrast to total depletion of CD4+ T cells, and consistent with functional heterogeneity of CD4+ T cells, recent data indicate that a subset of CD4+ IL22+ T cells likely protects against AP in mice, even though exact mechanisms remain elusive[64].
The magnitude of the inflammatory process is amplified following further secretion of inflammatory mediators by infiltrating immune-associated cells[65-67], and over-expression of adhesion molecules including intercellular adhesion molecule 1 (ICAM-1) and vascular adhesion molecule 1[68,69].The latter represent ligands for lymphocyte function-associated antigen 1[70] on leukocytes and lymphocytes, αLβ2 and CD11a-CD18 on monocytes and integrin macrophage 1 antigen (Mac-1) on neutrophils, while their secretion is promoted by ROS generation and TNF-α itself (Figure 1)[71-73]. Notably, ICAM-1 deficiency and systemic depletion of neutrophils were each shown to reduce the severity of AP and lung injury[71].
Except for regulation of cellular apoptosis, TNF-α was shown to increase intestinal paracellular permeability, by affecting tight junctions[74] and facilitating bacterial translocation from the epithelium[75]. It has been suggested that, pathogen-associated molecular patterns derived from the intestinal micro flora activate the host innate immune system via pattern recognition receptors, such as TLRs and nucleotide-binding domain and leucine-rich repeat-containing molecules[76] (Figure 1). Activation of TLRs and nucleotide-binding domain and leucine rich repeat-containing molecules likely mediates the mechanism by which bacterial translocation leads to severe AP. Consistent with this, mice that lack TLR4 develop less severe forms of AP[77], and polymorphisms in TLR genes have been associated with susceptibility to AP[78,79]. Interestingly, up-regulation of TLR4 has been associated with increased expression of TNF-α in peripheral blood mononuclear cells during early stages of AP[80].
Various molecules and mechanisms appear to complete the full spectra of manifestations in AP, mainly attributed to microcirculatory disturbance including nitric oxide, endothelin, oxygen free radicals, bradykinin, prostaglandin I2 and endothelin[81]. Inflammatory mediators induce microcirculatory disturbance mainly through increasing capillary permeability and decreasing capillary blood flow velocity (such as ICAM-1), promoting the contraction of arteries and veins (such as endothelin), as well as, promoting platelet aggregation and inducing thrombosis (such as PAF and TXA2). In the latter case, PAF exerts its biological activity through binding to its specific receptors on the surface of leukocytes, endothelial cells and platelets leading to microcirculatory disturbance in AP[82-85] (Figure 1). Furthermore, an increasing body of evidence reveals a pro-inflammatory role of platelets except for their established function in thrombosis and haemostasis[86-88]. During AP, data have come to show that platelets regulate neutrophil accumulation in the pancreatic tissue[89], even though exact mechanisms underlying platelet dependent leukocyte recruitment remain elusive. At the moment these mechanisms and various molecules[90], although important; surpass the purpose of this review and will not be discussed further. Figure 1 represents a schematic summary of innate and adaptive immune response mechanisms implicated in acute pancreatitis.
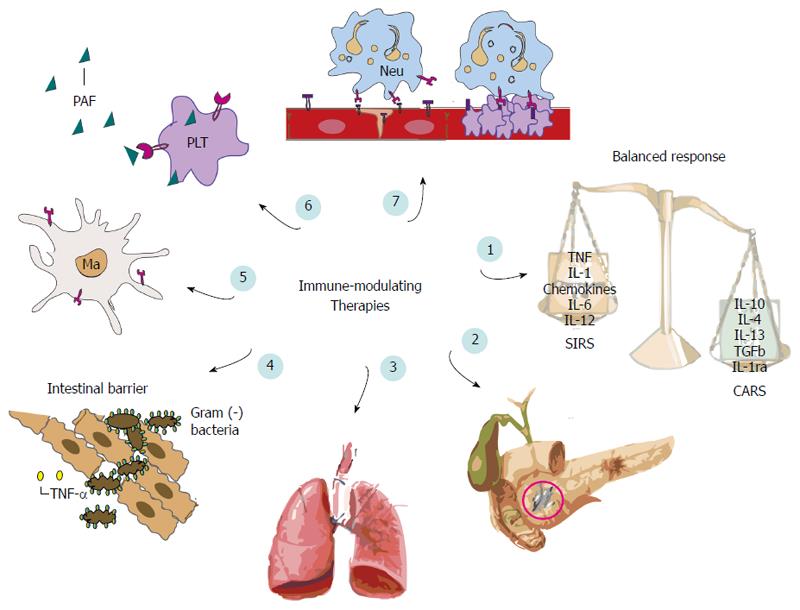
In mild AP, inflammation is regulated and confined by the host’s inflammatory response in the affected area. Although, most episodes of AP are mild, some patients proceed to SIRS, as a result of pro-inflammatory mediators’ release into the circulation[91,92], with local and extra-pancreatic complications[93], including respiratory, renal and hepatic dysfunction[94,95]. Systemic inflammation in AP is concomitantly associated with rapidly strengthening compensatory anti-inflammatory response syndrome (CARS)[96]. Even though, CARS may be sufficient to control SIRS and ensure favourable prognosis, excessive CARS may be overwhelming, leading to immune deficiency or suppression, which renders the host susceptible to secondary infections[97]. Increased serum concentrations of anti-inflammatory mediators including IL-10, IL-11, TNF-α receptors, and IL-1 receptors antagonist (IL-1ra) have been demonstrated in AP[98-102]. In immune-suppression monocytes are characterized by a significantly decreased human leukocyte antigen antigen-DR (HLA-DR) expression, process mostly attributed to IL-10 production[103]. Along with consequent impaired antigen presentation[104,105], monocytes show a profound reduction of their ability to produce pro-inflammatory cytokines e.g., TNF-α[106,107], facts associated with development of secondary infections[97] and organ failure[108,109]. IL-1ra and IL-6 are also important anti-inflammatory cytokines. IL1-ra blocks IL-1 mediated responses[110] while IL-6 appears to prevent synthesis of IL-1β and TNF-α[111].
Following understanding that, outcomes of our patients seem to be mostly dependent on pro- as well as, anti-inflammatory responses; in the last few years, a number of experimental and clinical studies have focused their interest in immune regulation during AP (Figure 2).
Recent data in animal models has come to show that, TNF-antagonism by either TNF-receptor blockade or anti TNF-antibodies protected from local intra-pancreatic damage, systemic complications and overall mortality in the vast majority of cases[29,112-117]. Administration of infliximab -a monoclonal TNF-antibody- appears to decrease serum amylase activity in both acute oedematous pancreatitis and severe necrotizing pancreatitis in a murine model[118]. In the latter case, a tendency to ameliorate both parenchymal and fatty tissue necrosis of the pancreas and alleviate acute respiratory distress syndrome-like pulmonary complications, was also noted. However, even though TNF-α has been clearly associated with failure of intestinal barrier, the latter study provided no information upon infliximab’s role on intestinal permeability since bacterial translocation was not considered a septic complication. However, previous reports that TNF-α blockage appears to correct intestinal permeability in people with Crohn’s disease[119], suggests that this effect may occur in other types of disorders and could represent a feasible therapeutic option. In a recent study, Aydin et al[120] showed that, infliximab administration 6 h after the induction of pancreatitis exerted beneficial effects on blood amylase levels and histopathologic changes in experimental necrotizing pancreatitis, while significant decrease in the degree of BT was noted. Even though, premature administration of anti-TNF 6 h post pancreatitis induction was beneficial, previous reports support that a prophylactic design starting the treatment before the induction could be superior, while the protective effect of TNF-antagonism on disease severity and mortality was still observed after the systemic effects had developed[116]. To date, no study has been conducted investigating TNF-a impact in clinical acute pancreatitis except for an isolated report in a patient with Crohn’s disease complicated with acute pancreatitis[121]. Interestingly, favourable outcome was observed following infliximab’s administration. However, data from two phase III sepsis trials[122,123] sharing an indistinguishable profile of inflammatory mediators with AP, have not been that optimistic. The use of an anti-TNF antibody in patients with sepsis failed to reduce 28-d mortality, suggesting that previous results from experimental studies or anecdotal reports should be interpreted with caution.
As previously described, pro-inflammatory cytokines such as IL-6, IL-1 and TNF are released in acute pancreatitis, while their plasma level correlates well with the severity of the disease and the occurrence of multi-organ failure[124-126]. By contrast, anti-inflammatory mediators such as IL-10, appear to mitigate the effects of inflammatory response and their level seems to be inversely proportional to the severity of pancreatitis[127,128].
IL-1: As observed for TNF, organ-specific expression of IL-1 is an early feature in experimental acute pancreatitis and is found in both the pancreas and distant organs[129,130]. Blockade of the IL-1 receptor by either targeted genetic disruption or pharmacological agents uniformly reduced the extent of intra-pancreatic damage, systemic complication, and mortality, similarly to TNF-α blockade[131-135]. Alternatively, the approach of inhibiting caspase-1, formerly termed interleukin 1β-converting enzyme (ICE) has been explored. Targeting ICE activity by a specific synthetic inhibitor dramatically attenuated both severity and mortality irrespective of the model used[136-139]. Interestingly, severity and mortality were still reduced even when a therapeutic window of 12 h following induction of severe acute pancreatitis was allowed[137]. Similarly to TNF-α, implementation of experimental findings to the clinical setting has been controversial. Even though, a post hoc analysis of a controlled trial of human recombinant IL-1ra in patients with sepsis showed a trend towards increased survival in patients with MODS[140], this observation could not be confirmed in a subsequent trial[141]. Technical reasons concerning the optimal dosage, duration and in vivo activity of the antibody could be responsible for these discrepancies [142].
IL-10: IL-10 - irrespectively of whether its activity has been blocked or augmented -has been shown to exert a protective effect in several models of acute pancreatitis in the past[143-149]. Cytokine manipulation appeared to significantly ameliorate organ specific damage in the pancreas and peripheral tissues, including the lung and the liver, while mortality was significantly reduced. Interestingly, IL-10 protective effect was still observed even when intervention occurred therapeutically after acute pancreatitis had already been induced[144,145,149]. However, this data has not been confirmed in the clinical setting. No significant differences were detected between IL-10 and placebo administration within 36 h of onset of symptoms, in days of hospital stay, CT scan score, organ failure score and local complications[150]. Conflicting results have come to light from randomized double-blind studies regarding the ability of IL-10 to prevent ERCP–induced AP. In his study, Devière et al[149] reported that, IL-10 decreased the incidence of post-ERCP pancreatitis, as well as, the length of hospital stay independently from other risk factors. This was not confirmed by a later American trial in which only a trend towards and not a significant decrease in the former parameters was noted[151]. A recent meta-analysis including patients receiving recombinant IL-10 or placebo before ERCP could show that, IL-10 significantly reduces the risk of post-ERCP pancreatitis[152]. Whether IL-10 treatment can ultimately prevent post-ERCP still remains under investigation.
IL-2: In contrast to the late effects of IL-2 deficiency and immune-paralysis, the excessive IL-2 mediated T-cell response during the early course of the disease can be deleterious[62,153-157] Following transcriptional regulation by administration of the FK506 agent which inhibits IL-2 production, decreased early local and systemic disease severity was noted even when given in a therapeutic fashion[155]. Similarly, early data have come to show that, sirolimus, an immune-modulatory agent, acting through inhibition of response to IL-2, thereby blocking activation of T and B cells, reduces acute pancreatic damage in the first week and less chronic changes in the further course of disease[157]. However, opposite results were shown by other studies. FK506 administration significantly worsened survival in diet-induced murine pancreatitis[156] while potentiation of IL-2 production through levamisole administration effectively decreased the incidence of pancreatic infections in a cat model of severe acute pancreatitis[153].
IL-18: IL-18 or interferon-inducing factor represents a novel key regulator of Th-1 response, through its ability to induce IFN-production in T and natural killer cells[158]. Scarce data on this interesting cytokine have shown that intra-pancreatic damage was decreased more effectively following neutralization of IL-18 activity by monoclonal antibodies than neutralizing IL-1 activity in cerulein-induced pancreatitis in mice[159]. Further research in IL-18 and its role during AP is currently in progress.
IL-6: Despite the numerous clinical studies investigating the role of IL-6 in the development of inflammatory syndrome, unfortunately only few reports have examined the role of this interleukin as potential target for modulating disease severity. Even though, IL-6 has been found to be a good predictor in predicting pancreatitis associated complications, including organ failure[160], limited experimental data have revealed that genetic deletion or prophylactic inhibition of IL-6 rather worsened outcomes than exerted a protective effect on disease severity and mortality[161-163].
Following previous observations implicating PAF in classical morphologic and biochemical derangements induced during AP[164], a number of groups have pursued the potential therapeutic role of this novel cytokine[165-171]. Except for one case[172], pathophysiological changes of acute pancreatitis were reduced in all established experimental models following PAF antagonism[168-171]. When PAF antagonists were applied therapeutically, local intra-pancreatic damage and micro-circulatory derangement was significantly ameliorated while systemic complications and mortality was considerably decreased[168,170,171]. One of the most promising PAF antagonists Lexipafant has been recently tested in two phase II trials including patients with acute[173] or predicted severe pancreatitis[174]. Results were encouraging, showing significant improvement of organ failure or organ failure scores. Subsequently though, in a randomized, double blind trial encountering patients with severe AP, intravenous administration of lexipafant for 7 d did not show any benefit in reducing MODS or mortality[175]. Nonetheless, systemic levels of IL-8 and E-selectin, sepsis or pseudocysts development were significantly lower in the treated than in the non-treated group, especially in patients treated within 48 h from the onset of symptoms[175]. In view of the multiple prior experimental studies suggesting that lexipafant is highly effective in reducing SIRS associated with AP[176,177], discrepancies among results finally came to challenge the actual role of PAF antagonism in the clinical setting. Discordance among studies can be partly attributed to the timing of each intervention and/or commercial influence and are further discussed.
So far, only a limited number of experimental and clinical studies have examined the efficacy of chemokine blockade in AP, mostly due to their more distal position within the inflammatory mediator cascade in comparison to cytokines.
In view of IL-8 detrimental effect in experimental AP[178], Osman et al[179] investigated the impact of prophylactic blockade of IL-8 in a rabbit model. Interestingly, significant reduction in systemic severity including lung injury, and mortality was observed, whereas the degree of local intrapancreatic damage remained unchanged. Even though, this study could not assess for potential therapeutic effects, the above data strongly supported the role of chemokines in mediating distant organ failure in AP.
Besides IL-8, high concentrations of other chemokines such as MCP-1, growth-related oncogene alpha, and epithelial neutrophil-activating protein 78 could be also found during the early stages of clinical acute pancreatitis. Blockade of specific CXC chemokines via specific antibodies, synthetic inhibitors or genetic deletion appeared to reduce pancreatitis associated pulmonary damage in several experimental studies[59,180,181]. Nonetheless, similar to IL-8, a complete absence of effect on local intrapancreatic damage was also observed[59,180,181].So far, only MCP-1 seemed to exert a detrimental role on the degree of local intrapancreatic damage[182]. Even though, in most of these studies, protective effects of chemokine blockade were observed even in a therapeutic design[59,180,182], no systematic study of their impact upon mortality has ever been carried out.
The expression of adhesion molecules is pivotal for the development of endothelial barrier dysfunction, transmigration of neutrophils and concomitant development of organ dysfunction. Treatment with antibodies against adhesion molecules like ICAM-1 and platelet endothelial cell adhesion molecule-1 (PECAM-1) has shown to be effective in the experimental setting[71,183-185]. Similarly, recent data demonstrate that platelets regulate leukocyte rolling in acute pancreatitis via induction of P-selectin, which was critical in supporting leukocyte rolling in inflamed venules of the pancreas[186]. It seems that inhibition of P-selectin protected against pancreatic tissue injury in experimental pancreatitis[187].
The roles of macrophages in the pathogenesis and progression of experimental AP make these cells interesting therapeutic targets since they exhibit both pro- and anti-inflammatory properties. Macrophages inhibitors (compounds such as gadolinium chloride, liposome-encapsulated dichloromethylene diphosphonate, and PAF antagonists) were shown to modulate the systemic inflammatory response[188]. However, most studies administered the inhibitors before AP was induced, which is clinically less relevant because most patients present after pancreatic injury. Other approaches to modify macrophages, either in vivo or ex vivo, into cells with anti-inflammatory properties have recently been tested. Currently, favourable effects of IL-4 and IL-13 have only been confirmed in vitro[189], while transfer of hemin-activated macrophages promoting production of anti-inflammatory agents seems promising[60,64,190,191]. In a study involving a xenogenic system, human bone marrow-derived clonal mesenchymal stem cells were administered to rats with mild or severe AP[192]. The human bone marrow–derived clonal mesenchymal stem cells induced Foxp3+T regulatory cells and suppressed pancreatic infiltration by T cells. Although more studies are needed in this area of research, stem cell–based immunosuppressive strategies could be developed as allogenic therapies for AP[193].
In rat models of AP, hydrocortisone has reduced mortality and blood cytokine levels[194-196]. At the moment, no human trials using steroids as treatment of AP have been published and attempts to show a beneficial effect of steroids as prophylaxis against post-ERCP pancreatitis in prospective placebo-controlled trials have been disappointing[197-199]. The use of glucocorticoids may, however, find a place as part of a combination therapy, as they suppress the inflammatory response, potentially through the inhibition NF-κB[200,201].
Increased levels of NF-κB during acute inflammation correlate well with AP severity[24,25], indicating that potential inhibition could improve outcomes[202,203]. NF-κB signalling seems to regulate autophagy during necrotising pancreatitis, while inhibition of NF-κB pathway reduced serum amylase and autophagosome formation in experimental models[204]. Similarly, lung injury and pancreatic destruction was lower in rats with acute necrotising pancreatitis following administration of NF-κB-N-acetylcysteine inhibitor[205].
The complex role of HMG1 in AP has not allowed the conduction of many studies[206]. Previous results have shown that anti-HMGB1 antibody improves lipopolysaccharide (LPS)-induced acute lung injury in mice[51], and ventilator-induced lung injury in rabbits[207]. In AP blockade of HMGB1 has been reported to attenuate the development of severe disease, as well as, associated organ dysfunction[208]. However, even though, HMGB1 can increase the permeability in enterocytic monolayers and bacterial translocation in mice[209], blockade of HMGB1 eventually deteriorated gut barrier function in this study[208].
Inhibiting pro-inflammatory mediators (e.g., PAF, IL-6, ICAM-1, TLR-4), enhancing anti- inflammatory mechanisms (e.g., IL-10) or modulating cellular immune responses, have all been found to be beneficial in experimental pancreatitis models[71,149,210,211]. Unfortunately, at the moment, they have all failed to find their way from the laboratory bench to the patient’s bedside[211,212], with the possible exception of preventing post-ERCP pancreatitis[213,214]. In this respect, a number of issues must be considered and addressed.
First, we have to keep in mind that discouraging results coming from the few representative clinical studies available on single anti-inflammatory agents does not necessarily mean that this approach is flawed in principle. The inflammatory mediators identified so far, most likely represent only the “tip of the iceberg”[215]; a million other mediators, underlying, interacting and regulatory mechanisms awaiting elucidation. Therefore, the concept of blocking single pro-inflammatory mediators could be an over-simplistic strategy to deal with the complex problem of acute pancreatitis, if there is any “ultimate” target at all. Of note, disastrous effects have been noted when single proximal mediators of the inflammatory response were blocked including development of anti-DNA, antinuclear or antithyroid antibodies, leading to various muscoloskeletal, neurological and skin manifestations[216,217].
In the complex network of inflammatory response, a multimodal strategy to inhibit several pro-inflammatory agents instead of one may be more useful[218,219]. The combination of the broad-acting antioxidant N-acetylcysteine, monoclonal antibodies against the adhesion molecule PECAM-1 and lexipafant was effective in animals with organ failure associated with AP[220]. The acute phase response and organ dysfunction was decreased, while gut barrier failure and translocation was prevented[220]. However, even in the case of multimodal strategy the example of TNF and anti IL-1 that share similar characteristics in both their pathophysiological functions, as well as, their regulation is worth noting[134,221]. As it was convincingly shown by Denham et al[131], no additive protective effects could be demonstrated by combined genetic disruption the IL-1 receptor and TNF in a murine model of AP, reflecting the huge challenges lying beyond the functional redundancy of the immune system[222].
However, even in such cases of multimodal management the timing of intervention remains critical. It is evident that the window for anti-inflammatory therapy to suppress excessive immune activation is very limited. Experimental and clinical evidence shows that the time limit for efficacious medical treatment is no more than 60 h from the onset of the symptoms of AP[223]. In fact, as stated by at least two controlled clinical trials including the European PAF-antagonist phase III trial[175,224], beneficial effect was achieved when treatment was introduced no later than 48 h after the onset of symptoms. Even though further trials are pivotal to clarify the proper timing for intervention, efforts are hampered by the fact that many patients present at a late stage of the disease, when organ failure may already be present and the patient may be already on his way to CARS or even immunosuppression[175].
This data challenges us to further explore and understand the tight balance and mechanisms underlying SIRS, CARS or even a mixed inflammatory response syndrome during the time-course of acute pancreatitis. In the last few years, an effort to monitor defects in monocyte function, as those reflected in reduced expression of HLA-DR during severe AP is under progress[107,225,226]. However, inflammatory stages may not be synchronous in the same patient, and even though immune-suppression may be evident in the peripheral blood, other end organs including the lungs may still be in the pro-inflammatory stage. Therefore, immune-stimulatory treatment must be used with caution and physicians should have the proper means to monitor the patients’ immune-inflammatory state, in order to most accurately identify patients who are at risk of organ failure. Signalling pathways and molecules of circulating leukocytes including HLA-DR, NF-κB, signal transducers and activators of transcription, and members of mitogen activated protein kinase family represent good candidates that could serve as future indices to identify the patients at risk for secondary infections and, thus, late organ failure[227-229].
Nonetheless, currently progress in AP research is slow, mainly due to the inaccessibility of the human pancreas to direct observation or biopsy, as well as, the self-destructive nature of the disease itself which does not allow distinguishing initiating events from the concomitant or consequent inflammatory response. Flaws in study design, including small sample sizes, variable tools to stratify disease severity and non-comparable study endpoints further hinder understanding of this complex disease. Consequently, most of our knowledge comes either from circulating inflammatory mediators and cells or animal models that are inevitably unable to simulate the complexity and individuality of human condition[230-232]. As a result, the authors of this review note a relative gap in experimental but mostly clinical research pertaining to AP during the last decade. The latter is reflected in the year of publication of many of our references, in contrast to the flourishing field of animal model pancreatitis during the 90’s and the dawn of the new millennium.
The key to future advances lies in obtaining data upon actual patients, making use of correct scientific methods and better design of clinical trials[233]. Recording of other meaningful parameters besides mortality including time from the onset of symptoms to type of intervention, permanent target organ damage, quality of life, pain scores or hospital stay should also be recorded. Improvement of the communication of the results is also pivotal. Scientific and editorial community must share the responsibility of publishing well-designed and well-conducted clinical studies irrespective of commercial or financial influence. Examples of poor management of these issues could be partly mirrored by the controversial efficacy of protease inhibitors in human AP[234] as well as, the highly debated results following lexipafant administration[235].
Treatment of AP by immune modulation currently represents an attractive and highly promising concept. However, further meticulous work lies ahead in order to overcome the fundamental conceptual problems surrounding the complex pathophysiology of this challenging disease. Individualized and timely management calls for close monitoring so that best possible outcomes are ensured for our patients.
P- Reviewer: Apte MV, Clemens DL, Cosen-Binker LI, Zamir DL S- Editor: Gou SX L- Editor: A E- Editor: Wang CH
| 1. | Myer PA, Mannalithara A, Singh G, Singh G, Pasricha PJ, Ladabaum U. Clinical and economic burden of emergency department visits due to gastrointestinal diseases in the United States. Am J Gastroenterol. 2013;108:1496-1507. [PubMed] |
| 2. | Fagenholz PJ, Fernández-del Castillo C, Harris NS, Pelletier AJ, Camargo CA. Direct medical costs of acute pancreatitis hospitalizations in the United States. Pancreas. 2007;35:302-307. [PubMed] |
| 3. | Fagenholz PJ, Castillo CF, Harris NS, Pelletier AJ, Camargo CA. Increasing United States hospital admissions for acute pancreatitis, 1988-2003. Ann Epidemiol. 2007;17:491-497. [PubMed] |
| 4. | Yadav D, Lowenfels AB. Trends in the epidemiology of the first attack of acute pancreatitis: a systematic review. Pancreas. 2006;33:323-330. [PubMed] |
| 5. | Tenner S, Sica G, Hughes M, Noordhoek E, Feng S, Zinner M, Banks PA. Relationship of necrosis to organ failure in severe acute pancreatitis. Gastroenterology. 1997;113:899-903. [PubMed] |
| 6. | Buter A, Imrie CW, Carter CR, Evans S, McKay CJ. Dynamic nature of early organ dysfunction determines outcome in acute pancreatitis. Br J Surg. 2002;89:298-302. [PubMed] |
| 7. | Büchler MW, Gloor B, Müller CA, Friess H, Seiler CA, Uhl W. Acute necrotizing pancreatitis: treatment strategy according to the status of infection. Ann Surg. 2000;232:619-626. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 533] [Cited by in RCA: 498] [Article Influence: 19.9] [Reference Citation Analysis (0)] |
| 8. | Medich DS, Lee TK, Melhem MF, Rowe MI, Schraut WH, Lee KK. Pathogenesis of pancreatic sepsis. Am J Surg. 1993;165:46-50; discussion 51-52. [PubMed] |
| 9. | Gianotti L, Munda R, Alexander JW, Tchervenkov JI, Babcock GF. Bacterial translocation: a potential source for infection in acute pancreatitis. Pancreas. 1993;8:551-558. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 50] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 10. | Kalfarentzos F, Kehagias J, Mead N, Kokkinis K, Gogos CA. Enteral nutrition is superior to parenteral nutrition in severe acute pancreatitis: results of a randomized prospective trial. Br J Surg. 1997;84:1665-1669. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 387] [Cited by in RCA: 293] [Article Influence: 10.9] [Reference Citation Analysis (1)] |
| 11. | Tenner S, Baillie J, DeWitt J, Vege SS. American College of Gastroenterology guideline: management of acute pancreatitis. Am J Gastroenterol. 2013;108:1400-1415; 1416. [PubMed] |
| 12. | Peery AF, Dellon ES, Lund J, Crockett SD, McGowan CE, Bulsiewicz WJ, Gangarosa LM, Thiny MT, Stizenberg K, Morgan DR. Burden of gastrointestinal disease in the United States: 2012 update. Gastroenterology. 2012;143:1179-1187.e1-3. [PubMed] |
| 13. | de Beaux AC, Goldie AS, Ross JA, Carter DC, Fearon KC. Serum concentrations of inflammatory mediators related to organ failure in patients with acute pancreatitis. Br J Surg. 1996;83:349-353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 181] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 14. | McKay CJ, Gallagher G, Brooks B, Imrie CW, Baxter JN. Increased monocyte cytokine production in association with systemic complications in acute pancreatitis. Br J Surg. 1996;83:919-923. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 110] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 15. | Ogawa M. Acute pancreatitis and cytokines: “second attack” by septic complication leads to organ failure. Pancreas. 1998;16:312-315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 90] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 16. | Lundberg AH, Granger DN, Russell J, Sabek O, Henry J, Gaber L, Kotb M, Gaber AO. Quantitative measurement of P- and E-selectin adhesion molecules in acute pancreatitis: correlation with distant organ injury. Ann Surg. 2000;231:213-222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 68] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 17. | Sah RP, Saluja A. Molecular mechanisms of pancreatic injury. Curr Opin Gastroenterol. 2011;27:444-451. [PubMed] |
| 18. | Saluja AK, Lerch MM, Phillips PA, Dudeja V. Why does pancreatic overstimulation cause pancreatitis? Annu Rev Physiol. 2007;69:249-269. [PubMed] |
| 19. | Mareninova OA, Hermann K, French SW, O’Konski MS, Pandol SJ, Webster P, Erickson AH, Katunuma N, Gorelick FS, Gukovsky I. Impaired autophagic flux mediates acinar cell vacuole formation and trypsinogen activation in rodent models of acute pancreatitis. J Clin Invest. 2009;119:3340-3355. [PubMed] |
| 20. | Gukovskaya AS, Gukovsky I. Autophagy and pancreatitis. Am J Physiol Gastrointest Liver Physiol. 2012;303:G993-G1003. [PubMed] |
| 21. | Gukovsky I, Pandol SJ, Mareninova OA, Shalbueva N, Jia W, Gukovskaya AS. Impaired autophagy and organellar dysfunction in pancreatitis. J Gastroenterol Hepatol. 2012;27 Suppl 2:27-32. [PubMed] |
| 22. | Grasso D, Ropolo A, Lo Ré A, Boggio V, Molejón MI, Iovanna JL, Gonzalez CD, Urrutia R, Vaccaro MI. Zymophagy, a novel selective autophagy pathway mediated by VMP1-USP9x-p62, prevents pancreatic cell death. J Biol Chem. 2011;286:8308-8324. [PubMed] |
| 23. | Saluja AK, Bhagat L, Lee HS, Bhatia M, Frossard JL, Steer ML. Secretagogue-induced digestive enzyme activation and cell injury in rat pancreatic acini. Am J Physiol. 1999;276:G835-G842. [PubMed] |
| 24. | Huang H, Liu Y, Daniluk J, Gaiser S, Chu J, Wang H, Li ZS, Logsdon CD, Ji B. Activation of nuclear factor-κB in acinar cells increases the severity of pancreatitis in mice. Gastroenterology. 2013;144:202-210. [PubMed] |
| 25. | Gukovsky I, Gukovskaya AS, Blinman TA, Zaninovic V, Pandol SJ. Early NF-kappaB activation is associated with hormone-induced pancreatitis. Am J Physiol. 1998;275:G1402-G1414. [PubMed] |
| 26. | Chen CC, Wang SS, Lu RH, Lu CC, Chang FY, Lee SD. Early changes of serum proinflammatory and anti-inflammatory cytokines after endoscopic retrograde cholangiopancreatography. Pancreas. 2003;26:375-380. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 27] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 27. | Rakonczay Z, Hegyi P, Takács T, McCarroll J, Saluja AK. The role of NF-kappaB activation in the pathogenesis of acute pancreatitis. Gut. 2008;57:259-267. [PubMed] |
| 28. | Zhang XP, Zhang L, Xu HM, Xu YP, Cheng QH, Wang JM, Shen HP. Application of tissue microarrays to study the influence of dexamethasone on NF-kappaB expression of pancreas in rat with severe acute pancreatitis. Dig Dis Sci. 2008;53:571-580. [PubMed] |
| 29. | Binker MG, Binker-Cosen AA, Richards D, Gaisano HY, de Cosen RH, Cosen-Binker LI. Chronic stress sensitizes rats to pancreatitis induced by cerulein: role of TNF-α. World J Gastroenterol. 2010;16:5565-5581. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 22] [Cited by in RCA: 21] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 30. | Booth DM, Mukherjee R, Sutton R, Criddle DN. Calcium and reactive oxygen species in acute pancreatitis: friend or foe? Antioxid Redox Signal. 2011;15:2683-2698. [PubMed] |
| 31. | Dawra R, Sah RP, Dudeja V, Rishi L, Talukdar R, Garg P, Saluja AK. Intra-acinar trypsinogen activation mediates early stages of pancreatic injury but not inflammation in mice with acute pancreatitis. Gastroenterology. 2011;141:2210-2217.e2. [PubMed] |
| 32. | Clarke R, Cook KL, Hu R, Facey CO, Tavassoly I, Schwartz JL, Baumann WT, Tyson JJ, Xuan J, Wang Y. Endoplasmic reticulum stress, the unfolded protein response, autophagy, and the integrated regulation of breast cancer cell fate. Cancer Res. 2012;72:1321-1331. [PubMed] |
| 33. | Kroemer G, Mariño G, Levine B. Autophagy and the integrated stress response. Mol Cell. 2010;40:280-293. [PubMed] |
| 34. | Saitoh T, Akira S. Regulation of innate immune responses by autophagy-related proteins. J Cell Biol. 2010;189:925-935. [PubMed] |
| 35. | Levine B, Mizushima N, Virgin HW. Autophagy in immunity and inflammation. Nature. 2011;469:323-335. [PubMed] |
| 36. | Deretic V, Saitoh T, Akira S. Autophagy in infection, inflammation and immunity. Nat Rev Immunol. 2013;13:722-737. [PubMed] |
| 37. | Feng D, Park O, Radaeva S, Wang H, Yin S, Kong X, Zheng M, Zakhari S, Kolls JK, Gao B. Interleukin-22 ameliorates cerulein-induced pancreatitis in mice by inhibiting the autophagic pathway. Int J Biol Sci. 2012;8:249-257. [PubMed] |
| 38. | Marichal T, Ohata K, Bedoret D, Mesnil C, Sabatel C, Kobiyama K, Lekeux P, Coban C, Akira S, Ishii KJ, Bureau F, Desmet CJ. DNA released from dying host cells mediates aluminum adjuvant activity. Nat Med. 2011;17:996-1002. [PubMed] |
| 39. | Zhang Q, Raoof M, Chen Y, Sumi Y, Sursal T, Junger W, Brohi K, Itagaki K, Hauser CJ. Circulating mitochondrial DAMPs cause inflammatory responses to injury. Nature. 2010;464:104-107. [PubMed] |
| 40. | Nakamura T, Ito T, Igarashi H, Uchida M, Hijioka M, Oono T, Fujimori N, Niina Y, Suzuki K, Jensen RT. Cytosolic double-stranded DNA as a damage-associated molecular pattern induces the inflammatory response in rat pancreatic stellate cells: a plausible mechanism for tissue injury-associated pancreatitis. Int J Inflam. 2012;2012:504128. [PubMed] |
| 41. | Melvin VS, Edwards DP. Coregulatory proteins in steroid hormone receptor action: the role of chromatin high mobility group proteins HMG-1 and -2. Steroids. 1999;64:576-586. [PubMed] |
| 42. | Bustin M. Regulation of DNA-dependent activities by the functional motifs of the high-mobility-group chromosomal proteins. Mol Cell Biol. 1999;19:5237-5246. [PubMed] |
| 43. | Goodwin GH, Sanders C, Johns EW. A new group of chromatin-associated proteins with a high content of acidic and basic amino acids. Eur J Biochem. 1973;38:14-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 558] [Cited by in RCA: 585] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 44. | Yasuda T, Ueda T, Takeyama Y, Shinzeki M, Sawa H, Nakajima T, Ajiki T, Fujino Y, Suzuki Y, Kuroda Y. Significant increase of serum high-mobility group box chromosomal protein 1 levels in patients with severe acute pancreatitis. Pancreas. 2006;33:359-363. [PubMed] |
| 45. | Ulloa L, Tracey KJ. The “cytokine profile”: a code for sepsis. Trends Mol Med. 2005;11:56-63. [PubMed] |
| 46. | Yu M, Wang H, Ding A, Golenbock DT, Latz E, Czura CJ, Fenton MJ, Tracey KJ, Yang H. HMGB1 signals through toll-like receptor (TLR) 4 and TLR2. Shock. 2006;26:174-179. [PubMed] |
| 47. | Kang R, Zhang Q, Hou W, Yan Z, Chen R, Bonaroti J, Bansal P, Billiar TR, Tsung A, Wang Q. Intracellular Hmgb1 inhibits inflammatory nucleosome release and limits acute pancreatitis in mice. Gastroenterology. 2014;146:1097-1107. [PubMed] |
| 48. | Reiss LK, Uhlig U, Uhlig S. Models and mechanisms of acute lung injury caused by direct insults. Eur J Cell Biol. 2012;91:590-601. [PubMed] |
| 49. | Kim JY, Park JS, Strassheim D, Douglas I, Diaz del Valle F, Asehnoune K, Mitra S, Kwak SH, Yamada S, Maruyama I. HMGB1 contributes to the development of acute lung injury after hemorrhage. Am J Physiol Lung Cell Mol Physiol. 2005;288:L958-L965. [PubMed] |
| 50. | Scaffidi P, Misteli T, Bianchi ME. Release of chromatin protein HMGB1 by necrotic cells triggers inflammation. Nature. 2002;418:191-195. [PubMed] |
| 51. | Ueno H, Matsuda T, Hashimoto S, Amaya F, Kitamura Y, Tanaka M, Kobayashi A, Maruyama I, Yamada S, Hasegawa N. Contributions of high mobility group box protein in experimental and clinical acute lung injury. Am J Respir Crit Care Med. 2004;170:1310-1316. [PubMed] |
| 52. | Gong Q, Xu JF, Yin H, Liu SF, Duan LH, Bian ZL. Protective effect of antagonist of high-mobility group box 1 on lipopolysaccharide-induced acute lung injury in mice. Scand J Immunol. 2009;69:29-35. [PubMed] |
| 53. | Barsness KA, Arcaroli J, Harken AH, Abraham E, Banerjee A, Reznikov L, McIntyre RC. Hemorrhage-induced acute lung injury is TLR-4 dependent. Am J Physiol Regul Integr Comp Physiol. 2004;287:R592-R599. [PubMed] |
| 54. | Andersson U, Wang H, Palmblad K, Aveberger AC, Bloom O, Erlandsson-Harris H, Janson A, Kokkola R, Zhang M, Yang H. High mobility group 1 protein (HMG-1) stimulates proinflammatory cytokine synthesis in human monocytes. J Exp Med. 2000;192:565-570. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1100] [Cited by in RCA: 1173] [Article Influence: 46.9] [Reference Citation Analysis (0)] |
| 55. | Kempuraj D, Twait EC, Williard DE, Yuan Z, Meyerholz DK, Samuel I. The novel cytokine interleukin-33 activates acinar cell proinflammatory pathways and induces acute pancreatic inflammation in mice. PLoS One. 2013;8:e56866. [PubMed] |
| 56. | Demyanets S, Konya V, Kastl SP, Kaun C, Rauscher S, Niessner A, Pentz R, Pfaffenberger S, Rychli K, Lemberger CE. Interleukin-33 induces expression of adhesion molecules and inflammatory activation in human endothelial cells and in human atherosclerotic plaques. Arterioscler Thromb Vasc Biol. 2011;31:2080-2089. [PubMed] |
| 57. | Mayerle J. A novel role for leucocytes in determining the severity of acute pancreatitis. Gut. 2009;58:1440-1441. [PubMed] |
| 58. | Schölmerich J, Schümichen C, Lausen M, Gross V, Leser HG, Lay L, Farthmann EH, Gerok W. Scintigraphic assessment of leukocyte infiltration in acute pancreatitis using technetium-99m-hexamethyl propylene amine oxine as leukocyte label. Dig Dis Sci. 1991;36:65-70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 59. | Bhatia M, Brady M, Zagorski J, Christmas SE, Campbell F, Neoptolemos JP, Slavin J. Treatment with neutralising antibody against cytokine induced neutrophil chemoattractant (CINC) protects rats against acute pancreatitis associated lung injury. Gut. 2000;47:838-844. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 127] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 60. | Gea-Sorlí S, Closa D. Role of macrophages in the progression of acute pancreatitis. World J Gastrointest Pharmacol Ther. 2010;1:107-111. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 44] [Cited by in RCA: 58] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 61. | Perides G, Weiss ER, Michael ES, Laukkarinen JM, Duffield JS, Steer ML. TNF-alpha-dependent regulation of acute pancreatitis severity by Ly-6C(hi) monocytes in mice. J Biol Chem. 2011;286:13327-13335. [PubMed] |
| 62. | Demols A, Le Moine O, Desalle F, Quertinmont E, Van Laethem JL, Devière J. CD4(+ )T cells play an important role in acute experimental pancreatitis in mice. Gastroenterology. 2000;118:582-590. [PubMed] |
| 63. | Pezzilli R, Billi P, Gullo L, Beltrandi E, Maldini M, Mancini R, Incorvaia L, Miglioli M. Behavior of serum soluble interleukin-2 receptor, soluble CD8 and soluble CD4 in the early phases of acute pancreatitis. Digestion. 1994;55:268-273. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 29] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 64. | Xue J, Nguyen DT, Habtezion A. Aryl hydrocarbon receptor regulates pancreatic IL-22 production and protects mice from acute pancreatitis. Gastroenterology. 2012;143:1670-1680. [PubMed] |
| 65. | Makhija R, Kingsnorth AN. Cytokine storm in acute pancreatitis. J Hepatobiliary Pancreat Surg. 2002;9:401-410. [PubMed] |
| 66. | Gukovskaya AS, Vaquero E, Zaninovic V, Gorelick FS, Lusis AJ, Brennan ML, Holland S, Pandol SJ. Neutrophils and NADPH oxidase mediate intrapancreatic trypsin activation in murine experimental acute pancreatitis. Gastroenterology. 2002;122:974-984. [PubMed] |
| 67. | Abdulla A, Awla D, Thorlacius H, Regnér S. Role of neutrophils in the activation of trypsinogen in severe acute pancreatitis. J Leukoc Biol. 2011;90:975-982. [PubMed] |
| 68. | Bernot D, Peiretti F, Canault M, Juhan-Vague I, Nalbone G. Upregulation of TNF-alpha-induced ICAM-1 surface expression by adenylate cyclase-dependent pathway in human endothelial cells. J Cell Physiol. 2005;202:434-441. [PubMed] |
| 69. | Hartwig W, Werner J, Warshaw AL, Antoniu B, Castillo CF, Gebhard MM, Uhl W, Büchler MW. Membrane-bound ICAM-1 is upregulated by trypsin and contributes to leukocyte migration in acute pancreatitis. Am J Physiol Gastrointest Liver Physiol. 2004;287:G1194-G1199. [PubMed] |
| 70. | Awla D, Abdulla A, Zhang S, Roller J, Menger MD, Regnér S, Thorlacius H. Lymphocyte function antigen-1 regulates neutrophil recruitment and tissue damage in acute pancreatitis. Br J Pharmacol. 2011;163:413-423. [PubMed] |
| 71. | Frossard JL, Saluja A, Bhagat L, Lee HS, Bhatia M, Hofbauer B, Steer ML. The role of intercellular adhesion molecule 1 and neutrophils in acute pancreatitis and pancreatitis-associated lung injury. Gastroenterology. 1999;116:694-701. [PubMed] |
| 72. | Lundberg AH, Granger N, Russell J, Callicutt S, Gaber LW, Kotb M, Sabek O, Gaber AO. Temporal correlation of tumor necrosis factor-alpha release, upregulation of pulmonary ICAM-1 and VCAM-1, neutrophil sequestration, and lung injury in diet-induced pancreatitis. J Gastrointest Surg. 2000;4:248-257. [PubMed] |
| 73. | Zaninovic V, Gukovskaya AS, Gukovsky I, Mouria M, Pandol SJ. Cerulein upregulates ICAM-1 in pancreatic acinar cells, which mediates neutrophil adhesion to these cells. Am J Physiol Gastrointest Liver Physiol. 2000;279:G666-G676. [PubMed] |
| 74. | Bruewer M, Luegering A, Kucharzik T, Parkos CA, Madara JL, Hopkins AM, Nusrat A. Proinflammatory cytokines disrupt epithelial barrier function by apoptosis-independent mechanisms. J Immunol. 2003;171:6164-6172. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 624] [Cited by in RCA: 666] [Article Influence: 31.7] [Reference Citation Analysis (0)] |
| 75. | Clark EC, Patel SD, Chadwick PR, Warhurst G, Curry A, Carlson GL. Glutamine deprivation facilitates tumour necrosis factor induced bacterial translocation in Caco-2 cells by depletion of enterocyte fuel substrate. Gut. 2003;52:224-230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 79] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 76. | Akira S, Takeda K. Toll-like receptor signalling. Nat Rev Immunol. 2004;4:499-511. [PubMed] |
| 77. | Sharif R, Dawra R, Wasiluk K, Phillips P, Dudeja V, Kurt-Jones E, Finberg R, Saluja A. Impact of toll-like receptor 4 on the severity of acute pancreatitis and pancreatitis-associated lung injury in mice. Gut. 2009;58:813-819. [PubMed] |
| 78. | Gao HK, Zhou ZG, Li Y, Chen YQ. Toll-like receptor 4 Asp299Gly polymorphism is associated with an increased risk of pancreatic necrotic infection in acute pancreatitis: a study in the Chinese population. Pancreas. 2007;34:295-298. [PubMed] |
| 79. | Takagi Y, Masamune A, Kume K, Satoh A, Kikuta K, Watanabe T, Satoh K, Hirota M, Shimosegawa T. Microsatellite polymorphism in intron 2 of human Toll-like receptor 2 gene is associated with susceptibility to acute pancreatitis in Japan. Hum Immunol. 2009;70:200-204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 23] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 80. | Li HG, Zhou ZG, Li Y, Zheng XL, Lei S, Zhu L, Wang Y. Alterations of Toll-like receptor 4 expression on peripheral blood monocytes during the early stage of human acute pancreatitis. Dig Dis Sci. 2007;52:1973-1978. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 28] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 81. | Zhang XP, Li ZJ, Zhang J. Inflammatory mediators and microcirculatory disturbance in acute pancreatitis. Hepatobiliary Pancreat Dis Int. 2009;8:351-357. [PubMed] |
| 82. | Flickinger BD, Olson MS. Localization of the platelet-activating factor receptor to rat pancreatic microvascular endothelial cells. Am J Pathol. 1999;154:1353-1358. [PubMed] |
| 83. | Fujimura K, Kubota Y, Ogura M, Yamaguchi T, Binnaka T, Tani K, Kitagawa S, Mizuno T, Inoue K. Role of endogenous platelet-activating factor in caerulein-induced acute pancreatitis in rats: protective effects of a PAF-antagonist. J Gastroenterol Hepatol. 1992;7:199-202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 18] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 84. | Wang WZ, Guo SZ, Tsai TM, Anderson GL, Miller FN. Platelet-activating factor contributes to postischemic vasospasm. J Surg Res. 2000;89:139-146. [PubMed] |
| 85. | Kingsnorth AN. Platelet-activating factor. Scand J Gastroenterol Suppl. 1996;219:28-31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 19] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 86. | McNicol A, Israels SJ. Beyond hemostasis: the role of platelets in inflammation, malignancy and infection. Cardiovasc Hematol Disord Drug Targets. 2008;8:99-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 102] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 87. | Nurden AT. Platelets, inflammation and tissue regeneration. Thromb Haemost. 2011;105 Suppl 1:S13-S33. [PubMed] |
| 88. | Akinosoglou K, Alexopoulos D. Use of antiplatelet agents in sepsis: a glimpse into the future. Thromb Res. 2014;133:131-138. [PubMed] |
| 89. | Abdulla A, Awla D, Hartman H, Rahman M, Jeppsson B, Regnér S, Thorlacius H. Role of platelets in experimental acute pancreatitis. Br J Surg. 2011;98:93-103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 27] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 90. | Shanmugam MK, Bhatia M. The role of pro-inflammatory molecules and pharmacological agents in acute pancreatitis and sepsis. Inflamm Allergy Drug Targets. 2010;9:20-31. [PubMed] |
| 91. | Malmstrøm ML, Hansen MB, Andersen AM, Ersbøll AK, Nielsen OH, Jørgensen LN, Novovic S. Cytokines and organ failure in acute pancreatitis: inflammatory response in acute pancreatitis. Pancreas. 2012;41:271-277. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 116] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 92. | Montravers P, Chollet-Martin S, Marmuse JP, Gougerot-Pocidalo MA, Desmonts JM. Lymphatic release of cytokines during acute lung injury complicating severe pancreatitis. Am J Respir Crit Care Med. 1995;152:1527-1533. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 65] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 94. | Polyzogopoulou E, Bikas C, Danikas D, Koutras A, Kalfarentzos F, Gogos CA. Baseline hypoxemia as a prognostic marker for pulmonary complications and outcome in patients with acute pancreatitis. Dig Dis Sci. 2004;49:150-154. [PubMed] |
| 95. | Closa D, Bardají M, Hotter G, Prats N, Gelpí E, Fernández-Cruz L, Roselló-Catafau J. Hepatic involvement in pancreatitis-induced lung damage. Am J Physiol. 1996;270:G6-13. [PubMed] |
| 96. | Bone RC. Sir Isaac Newton, sepsis, SIRS, and CARS. Crit Care Med. 1996;24:1125-1128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 717] [Cited by in RCA: 627] [Article Influence: 21.6] [Reference Citation Analysis (0)] |
| 97. | Mentula P, Kylänpää ML, Kemppainen E, Jansson SE, Sarna S, Puolakkainen P, Haapiainen R, Repo H. Plasma anti-inflammatory cytokines and monocyte human leucocyte antigen-DR expression in patients with acute pancreatitis. Scand J Gastroenterol. 2004;39:178-187. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 51] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 98. | Mayer JM, Rau B, Siech M, Beger HG. Local and systemic zymogen activation in human acute pancreatitis. Digestion. 2000;62:164-170. [PubMed] |
| 99. | De Beaux AC, Fearon KC. Circulating endotoxin, tumour necrosis factor-alpha, and their natural antagonists in the pathophysiology of acute pancreatitis. Scand J Gastroenterol Suppl. 1996;219:43-46. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 20] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 100. | Simovic MO, Bonham MJ, Abu-Zidan FM, Windsor JA. Anti-inflammatory cytokine response and clinical outcome in acute pancreatitis. Crit Care Med. 1999;27:2662-2665. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 46] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 101. | Chen CC, Wang SS, Lu RH, Chang FY, Lee SD. Serum interleukin 10 and interleukin 11 in patients with acute pancreatitis. Gut. 1999;45:895-899. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 56] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 102. | Mentula P, Kylänpää ML, Kemppainen E, Jansson SE, Sarna S, Puolakkainen P, Haapiainen R, Repo H. Early prediction of organ failure by combined markers in patients with acute pancreatitis. Br J Surg. 2005;92:68-75. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 92] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 103. | Fumeaux T, Pugin J. Role of interleukin-10 in the intracellular sequestration of human leukocyte antigen-DR in monocytes during septic shock. Am J Respir Crit Care Med. 2002;166:1475-1482. [PubMed] |
| 104. | Wolk K, Döcke WD, von Baehr V, Volk HD, Sabat R. Impaired antigen presentation by human monocytes during endotoxin tolerance. Blood. 2000;96:218-223. [PubMed] |
| 105. | Tonegawa S. Antibody and T-cell receptors. JAMA. 1988;259:1845-1847. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 106. | Hershman MJ, Appel SH, Wellhausen SR, Sonnenfeld G, Polk HC. Interferon-gamma treatment increases HLA-DR expression on monocytes in severely injured patients. Clin Exp Immunol. 1989;77:67-70. [PubMed] |
| 107. | Döcke WD, Randow F, Syrbe U, Krausch D, Asadullah K, Reinke P, Volk HD, Kox W. Monocyte deactivation in septic patients: restoration by IFN-gamma treatment. Nat Med. 1997;3:678-681. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 845] [Cited by in RCA: 847] [Article Influence: 30.3] [Reference Citation Analysis (0)] |
| 108. | Mentula P, Kylänpää-Bäck ML, Kemppainen E, Takala A, Jansson SE, Kautiainen H, Puolakkainen P, Haapiainen R, Repo H. Decreased HLA (human leucocyte antigen)-DR expression on peripheral blood monocytes predicts the development of organ failure in patients with acute pancreatitis. Clin Sci (Lond). 2003;105:409-417. [PubMed] |
| 109. | Richter A, Nebe T, Wendl K, Schuster K, Klaebisch G, Quintel M, Lorenz D, Post S, Trede M. HLA-DR expression in acute pancreatitis. Eur J Surg. 1999;165:947-951. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 31] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 110. | Dinarello CA. Interleukin-1, interleukin-1 receptors and interleukin-1 receptor antagonist. Int Rev Immunol. 1998;16:457-499. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 565] [Cited by in RCA: 549] [Article Influence: 20.3] [Reference Citation Analysis (0)] |
| 111. | Opal SM, DePalo VA. Anti-inflammatory cytokines. Chest. 2000;117:1162-1172. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1030] [Cited by in RCA: 1114] [Article Influence: 44.6] [Reference Citation Analysis (0)] |
| 112. | Guice KS, Oldham KT, Remick DG, Kunkel SL, Ward PA. Anti-tumor necrosis factor antibody augments edema formation in caerulein-induced acute pancreatitis. J Surg Res. 1991;51:495-499. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 52] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 113. | Grewal HP, Mohey el Din A, Gaber L, Kotb M, Gaber AO. Amelioration of the physiologic and biochemical changes of acute pancreatitis using an anti-TNF-alpha polyclonal antibody. Am J Surg. 1994;167:214-218; discussion 218-219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 125] [Cited by in RCA: 142] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 114. | Hughes CB, Gaber LW, Mohey el-Din AB, Grewal HP, Kotb M, Mann L, Gaber AO. Inhibition of TNF alpha improves survival in an experimental model of acute pancreatitis. Am Surg. 1996;62:8-13. [PubMed] |
| 115. | Hughes CB, Grewal HP, Gaber LW, Kotb M, El-din AB, Mann L, Gaber AO. Anti-TNFalpha therapy improves survival and ameliorates the pathophysiologic sequelae in acute pancreatitis in the rat. Am J Surg. 1996;171:274-280. [PubMed] |
| 116. | Norman JG, Fink GW, Messina J, Carter G, Franz MG. Timing of tumor necrosis factor antagonism is critical in determining outcome in murine lethal acute pancreatitis. Surgery. 1996;120:515-521. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 83] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 117. | Malleo G, Mazzon E, Siriwardena AK, Cuzzocrea S. TNF-alpha as a therapeutic target in acute pancreatitis--lessons from experimental models. ScientificWorldJournal. 2007;7:431-448. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 56] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 118. | Oruc N, Ozutemiz AO, Yukselen V, Nart D, Celik HA, Yuce G, Batur Y. Infliximab: a new therapeutic agent in acute pancreatitis? Pancreas. 2004;28:e1-e8. [PubMed] |
| 119. | Suenaert P, Bulteel V, Lemmens L, Noman M, Geypens B, Van Assche G, Geboes K, Ceuppens JL, Rutgeerts P. Anti-tumor necrosis factor treatment restores the gut barrier in Crohn’s disease. Am J Gastroenterol. 2002;97:2000-2004. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 278] [Cited by in RCA: 304] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 120. | Aydin S, Isik AT, Unal B, Comert B, Ozyurt M, Deveci S, Ozgur G, Cengiz O, Tasci I, Mas MR. Effects of infliximab on bacterial translocation in experimental acute necrotizing pancreatitis. Indian J Med Res. 2012;135:656-661. [PubMed] |
| 121. | Triantafillidis JK, Cheracakis P, Hereti IA, Argyros N, Karra E. Acute idiopathic pancreatitis complicating active Crohn’s disease: favorable response to infliximab treatment. Am J Gastroenterol. 2000;95:3334-3336. [PubMed] |
| 122. | Abraham E, Anzueto A, Gutierrez G, Tessler S, San Pedro G, Wunderink R, Dal Nogare A, Nasraway S, Berman S, Cooney R. Double-blind randomised controlled trial of monoclonal antibody to human tumour necrosis factor in treatment of septic shock. NORASEPT II Study Group. Lancet. 1998;351:929-933. [PubMed] |
| 123. | Cohen J, Carlet J. INTERSEPT: an international, multicenter, placebo-controlled trial of monoclonal antibody to human tumor necrosis factor-alpha in patients with sepsis. International Sepsis Trial Study Group. Crit Care Med. 1996;24:1431-1440. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 362] [Cited by in RCA: 303] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 124. | Leser HG, Gross V, Scheibenbogen C, Heinisch A, Salm R, Lausen M, Rückauer K, Andreesen R, Farthmann EH, Schölmerich J. Elevation of serum interleukin-6 concentration precedes acute-phase response and reflects severity in acute pancreatitis. Gastroenterology. 1991;101:782-785. [PubMed] |
| 125. | Viedma JA, Pérez-Mateo M, Domínguez JE, Carballo F. Role of interleukin-6 in acute pancreatitis. Comparison with C-reactive protein and phospholipase A. Gut. 1992;33:1264-1267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 114] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 126. | Berney T, Gasche Y, Robert J, Jenny A, Mensi N, Grau G, Vermeulen B, Morel P. Serum profiles of interleukin-6, interleukin-8, and interleukin-10 in patients with severe and mild acute pancreatitis. Pancreas. 1999;18:371-377. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 88] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 127. | Ohmoto K, Yamamoto S. Serum interleukin-6 and interleukin-10 in patients with acute pancreatitis: clinical implications. Hepatogastroenterology. 2005;52:990-994. [PubMed] |
| 128. | Han XC, Zhang YC, Wang Y, Jia MK. Clinical evaluation of serum interleukin 10 in patients with acute pancreatitis. Hepatobiliary Pancreat Dis Int. 2003;2:135-138. [PubMed] |
| 129. | Fink GW, Norman JG. Intrapancreatic interleukin-1beta gene expression by specific leukocyte populations during acute pancreatitis. J Surg Res. 1996;63:369-373. [PubMed] |
| 130. | Norman JG, Fink GW, Denham W, Yang J, Carter G, Sexton C, Falkner J, Gower WR, Franz MG. Tissue-specific cytokine production during experimental acute pancreatitis. A probable mechanism for distant organ dysfunction. Dig Dis Sci. 1997;42:1783-1788. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 168] [Cited by in RCA: 174] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 131. | Denham W, Yang J, Fink G, Denham D, Carter G, Ward K, Norman J. Gene targeting demonstrates additive detrimental effects of interleukin 1 and tumor necrosis factor during pancreatitis. Gastroenterology. 1997;113:1741-1746. [PubMed] |
| 132. | Tanaka N, Murata A, Uda K, Toda H, Kato T, Hayashida H, Matsuura N, Mori T. Interleukin-1 receptor antagonist modifies the changes in vital organs induced by acute necrotizing pancreatitis in a rat experimental model. Crit Care Med. 1995;23:901-908. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 48] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 133. | Norman J, Franz M, Messina J, Riker A, Fabri PJ, Rosemurgy AS, Gower WR. Interleukin-1 receptor antagonist decreases severity of experimental acute pancreatitis. Surgery. 1995;117:648-655. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 163] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 134. | Norman JG, Fink G, Franz M, Guffey J, Carter G, Davison B, Sexton C, Glaccum M. Active interleukin-1 receptor required for maximal progression of acute pancreatitis. Ann Surg. 1996;223:163-169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 63] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 135. | Norman JG, Franz MG, Fink GS, Messina J, Fabri PJ, Gower WR, Carey LC. Decreased mortality of severe acute pancreatitis after proximal cytokine blockade. Ann Surg. 1995;221:625-631; discussion 631-463. [PubMed] |
| 136. | Norman J, Yang J, Fink G, Carter G, Ku G, Denham W, Livingston D. Severity and mortality of experimental pancreatitis are dependent on interleukin-1 converting enzyme (ICE). J Interferon Cytokine Res. 1997;17:113-118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 54] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 137. | Paszkowski AS, Rau B, Mayer JM, Möller P, Beger HG. Therapeutic application of caspase 1/interleukin-1beta-converting enzyme inhibitor decreases the death rate in severe acute experimental pancreatitis. Ann Surg. 2002;235:68-76. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 64] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 138. | Rau B, Baumgart K, Paszkowski AS, Mayer JM, Beger HG. Clinical relevance of caspase-1 activated cytokines in acute pancreatitis: high correlation of serum interleukin-18 with pancreatic necrosis and systemic complications. Crit Care Med. 2001;29:1556-1562. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 51] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 139. | Rau B, Paszkowski A, Lillich S, Baumgart K, Möller P, Beger HG. Differential effects of caspase-1/interleukin-1beta-converting enzyme on acinar cell necrosis and apoptosis in severe acute experimental pancreatitis. Lab Invest. 2001;81:1001-1013. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 39] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 140. | Fisher CJ, Dhainaut JF, Opal SM, Pribble JP, Balk RA, Slotman GJ, Iberti TJ, Rackow EC, Shapiro MJ, Greenman RL. Recombinant human interleukin 1 receptor antagonist in the treatment of patients with sepsis syndrome. Results from a randomized, double-blind, placebo-controlled trial. Phase III rhIL-1ra Sepsis Syndrome Study Group. JAMA. 1994;271:1836-1843. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 647] [Cited by in RCA: 578] [Article Influence: 18.6] [Reference Citation Analysis (0)] |
| 141. | Opal SM, Fisher CJ, Dhainaut JF, Vincent JL, Brase R, Lowry SF, Sadoff JC, Slotman GJ, Levy H, Balk RA. Confirmatory interleukin-1 receptor antagonist trial in severe sepsis: a phase III, randomized, double-blind, placebo-controlled, multicenter trial. The Interleukin-1 Receptor Antagonist Sepsis Investigator Group. Crit Care Med. 1997;25:1115-1124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 621] [Cited by in RCA: 612] [Article Influence: 21.9] [Reference Citation Analysis (0)] |
| 142. | Marshall JC. Why have clinical trials in sepsis failed? Trends Mol Med. 2014;20:195-203. [PubMed] |
| 143. | Van Laethem JL, Marchant A, Delvaux A, Goldman M, Robberecht P, Velu T, Devière J. Interleukin 10 prevents necrosis in murine experimental acute pancreatitis. Gastroenterology. 1995;108:1917-1922. [PubMed] |
| 144. | Kusske AM, Rongione AJ, Ashley SW, McFadden DW, Reber HA. Interleukin-10 prevents death in lethal necrotizing pancreatitis in mice. Surgery. 1996;120:284-288; discussion 289. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 135] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 145. | Rongione AJ, Kusske AM, Kwan K, Ashley SW, Reber HA, McFadden DW. Interleukin 10 reduces the severity of acute pancreatitis in rats. Gastroenterology. 1997;112:960-967. [PubMed] |
| 146. | Osman MO, Jacobsen NO, Kristensen JU, Deleuran B, Gesser B, Larsen CG, Jensen SL. IT 9302, a synthetic interleukin-10 agonist, diminishes acute lung injury in rabbits with acute necrotizing pancreatitis. Surgery. 1998;124:584-592. [PubMed] |
| 147. | Gloor B, Todd KE, Lane JS, Rigberg DA, Reber HA. Mechanism of increased lung injury after acute pancreatitis in IL-10 knockout mice. J Surg Res. 1998;80:110-114. [PubMed] |
| 148. | Van Laethem JL, Eskinazi R, Louis H, Rickaert F, Robberecht P, Devière J. Multisystemic production of interleukin 10 limits the severity of acute pancreatitis in mice. Gut. 1998;43:408-413. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 80] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 149. | Devière J, Le Moine O, Van Laethem JL, Eisendrath P, Ghilain A, Severs N, Cohard M. Interleukin 10 reduces the incidence of pancreatitis after therapeutic endoscopic retrograde cholangiopancreatography. Gastroenterology. 2001;120:498-505. [PubMed] |
| 150. | Villoria A, Abadía de Barbará C, Molero X, Álvarez A, Antolín M, Guarner L, Malagelada JR. Early treatment with interleukin-10 (IL-10) in severe acute pancreatitis. Pancreatology. 2003;3:457-481. |
| 151. | Dumot JA, Conwell DL, Zuccaro G, Vargo JJ, Shay SS, Easley KA, Ponsky JL. A randomized, double blind study of interleukin 10 for the prevention of ERCP-induced pancreatitis. Am J Gastroenterol. 2001;96:2098-2102. [PubMed] |
| 152. | Singh P, Lee T, Davidoff S, Bank S. Efficacy of Interleukin 10 (IL10) in the prevention of post-ERCP pancreatitis: a meta-analysis. Gastrointest Endosc. 2002;55:AB150. |
| 153. | Widdison AL, Karanjia ND, Alvarez C, Reber HA. Influence of levamisole on pancreatic infection in acute pancreatitis. Am J Surg. 1992;163:100-103; discussion 103-104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 28] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 154. | Mayer J, Laine VJ, Rau B, Hotz HG, Foitzik T, Nevalainen TJ, Beger HG. Systemic lymphocyte activation modulates the severity of diet-induced acute pancreatitis in mice. Pancreas. 1999;19:62-68. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 25] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 155. | Mayer JM, Laine VJ, Gezgin A, Kolodziej S, Nevalainen TJ, Storck M, Beger HG. Single doses of FK506 and OKT3 reduce severity in early experimental acute pancreatitis. Eur J Surg. 2000;166:734-741. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 15] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 156. | Echigo Y, Inoue K, Kogire M, Doi R, Higashide S, Sumi S, Kaji H, Imamura M. Effects of cyclosporine and tacrolimus (FK 506) on acute pancreatitis in mice. Arch Surg. 1995;130:64-68. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 19] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 157. | Mayer JM, Kolodziej S, Jukka Laine V, Kahl S. Immunomodulation in a novel model of experimental chronic pancreatitis. Minerva Gastroenterol Dietol. 2012;58:347-354. [PubMed] |
| 158. | Dinarello CA. IL-18: A TH1-inducing, proinflammatory cytokine and new member of the IL-1 family. J Allergy Clin Immunol. 1999;103:11-24. [PubMed] |
| 159. | Rau B, Zhuang Y, Schöler C, Hans J, Tilton B, Schilling M. Caspase-1 activated cytokines in experimental acute pancreatitis: synergistic effects of IL-1 and IL-18 on the development of disease severity. Pancreas. 2003;27:403-404. |
| 160. | Mayer J, Rau B, Gansauge F, Beger HG. Inflammatory mediators in human acute pancreatitis: clinical and pathophysiological implications. Gut. 2000;47:546-552. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 262] [Cited by in RCA: 308] [Article Influence: 12.3] [Reference Citation Analysis (1)] |
| 161. | Liu Q, Djuricin G, Nathan C, Gattuso P, Weinstein RA, Prinz RA. The effect of interleukin-6 on bacterial translocation in acute canine pancreatitis. Int J Pancreatol. 2000;27:157-165. [PubMed] |
| 162. | Cuzzocrea S, Mazzon E, Dugo L, Centorrino T, Ciccolo A, McDonald MC, de Sarro A, Caputi AP, Thiemermann C. Absence of endogenous interleukin-6 enhances the inflammatory response during acute pancreatitis induced by cerulein in mice. Cytokine. 2002;18:274-285. [PubMed] |
| 163. | Suzuki S, Miyasaka K, Jimi A, Funakoshi A. Induction of acute pancreatitis by cerulein in human IL-6 gene transgenic mice. Pancreas. 2000;21:86-92. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 38] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 164. | Emanuelli G, Montrucchio G, Gaia E, Dughera L, Corvetti G, Gubetta L. Experimental acute pancreatitis induced by platelet activating factor in rabbits. Am J Pathol. 1989;134:315-326. [PubMed] |
| 165. | Dabrowski A, Gabryelewicz A, Chyczewski L. The effect of platelet activating factor antagonist (BN 52021) on cerulein-induced acute pancreatitis with reference to oxygen radicals. Int J Pancreatol. 1991;8:1-11. [PubMed] |
| 166. | Zhou W, McCollum MO, Levine BA, Olson MS. Role of platelet-activating factor in pancreatitis-associated acute lung injury in the rat. Am J Pathol. 1992;140:971-979. [PubMed] |
| 167. | Kald B, Kald A, Ihse I, Tagesson C. Release of platelet-activating factor in acute experimental pancreatitis. Pancreas. 1993;8:440-442. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 24] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 168. | Formela LJ, Wood LM, Whittaker M, Kingsnorth AN. Amelioration of experimental acute pancreatitis with a potent platelet-activating factor antagonist. Br J Surg. 1994;81:1783-1785. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 46] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 169. | Wang X, Sun Z, Börjesson A, Haraldsen P, Aldman M, Deng X, Leveau P, Andersson R. Treatment with lexipafant ameliorates the severity of pancreatic microvascular endothelial barrier dysfunction in rats with acute hemorrhagic pancreatitis. Int J Pancreatol. 1999;25:45-52. [PubMed] |
| 170. | Hofbauer B, Saluja AK, Bhatia M, Frossard JL, Lee HS, Bhagat L, Steer ML. Effect of recombinant platelet-activating factor acetylhydrolase on two models of experimental acute pancreatitis. Gastroenterology. 1998;115:1238-1247. [PubMed] |
| 171. | Foitzik T, Hotz HG, Eibl G, Hotz B, Kirchengast M, Buhr HJ. Therapy for microcirculatory disorders in severe acute pancreatitis: effectiveness of platelet-activating factor receptor blockade vs. endothelin receptor blockade. J Gastrointest Surg. 1999;3:244-251. [PubMed] |
| 172. | Rivera JA, Werner J, Warshaw AL, Lewandrowski KB, Rattner DW, Fernández del Castillo C. Lexipafant fails to improve survival in severe necrotizing pancreatitis in rats. Int J Pancreatol. 1998;23:101-106. [PubMed] |
| 173. | Kingsnorth AN, Galloway SW, Formela LJ. Randomized, double-blind phase II trial of Lexipafant, a platelet-activating factor antagonist, in human acute pancreatitis. Br J Surg. 1995;82:1414-1420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 138] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 174. | McKay CJ, Curran F, Sharples C, Baxter JN, Imrie CW. Prospective placebo-controlled randomized trial of lexipafant in predicted severe acute pancreatitis. Br J Surg. 1997;84:1239-1243. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 59] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 175. | Johnson CD, Kingsnorth AN, Imrie CW, McMahon MJ, Neoptolemos JP, McKay C, Toh SK, Skaife P, Leeder PC, Wilson P. Double blind, randomised, placebo controlled study of a platelet activating factor antagonist, lexipafant, in the treatment and prevention of organ failure in predicted severe acute pancreatitis. Gut. 2001;48:62-69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 245] [Cited by in RCA: 231] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 176. | Lane JS, Todd KE, Gloor B, Chandler CF, Kau AW, Ashley SW, Reber HA, McFadden DW. Platelet activating factor antagonism reduces the systemic inflammatory response in a murine model of acute pancreatitis. J Surg Res. 2001;99:365-370. [PubMed] |
| 177. | Liu Q, Djuricin G, Rossi H, Bewsey K, Nathan C, Gattuso P, Weinstein RA, Prinz RA. The effect of lexipafant on bacterial translocation in acute necrotizing pancreatitis in rats. Am Surg. 1999;65:611-616; discussion 617. [PubMed] |
| 178. | Osman MO, Lausten SB, Jakobsen NO, Kristensen JU, Deleuran B, Larsen CG, Jensen SL. Graded experimental acute pancreatitis: monitoring of a renewed rabbit model focusing on the production of interleukin-8 (IL-8) and CD11b/CD18. Eur J Gastroenterol Hepatol. 1999;11:137-149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 179. | Osman MO, Kristensen JU, Jacobsen NO, Lausten SB, Deleuran B, Deleuran M, Gesser B, Matsushima K, Larsen CG, Jensen SL. A monoclonal anti-interleukin 8 antibody (WS-4) inhibits cytokine response and acute lung injury in experimental severe acute necrotising pancreatitis in rabbits. Gut. 1998;43:232-239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 107] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 180. | Bhatia M, Proudfoot AE, Wells TN, Christmas S, Neoptolemos JP, Slavin J. Treatment with Met-RANTES reduces lung injury in caerulein-induced pancreatitis. Br J Surg. 2003;90:698-704. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 37] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 181. | Gerard C, Frossard JL, Bhatia M, Saluja A, Gerard NP, Lu B, Steer M. Targeted disruption of the beta-chemokine receptor CCR1 protects against pancreatitis-associated lung injury. J Clin Invest. 1997;100:2022-2027. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 171] [Cited by in RCA: 156] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 182. | Bhatia M, Ramnath RD, Chevali L, Guglielmotti A. Treatment with bindarit, a blocker of MCP-1 synthesis, protects mice against acute pancreatitis. Am J Physiol Gastrointest Liver Physiol. 2005;288:G1259-G1265. [PubMed] |
| 183. | Werner J, Z’graggen K, Fernández-del Castillo C, Lewandrowski KB, Compton CC, Warshaw AL. Specific therapy for local and systemic complications of acute pancreatitis with monoclonal antibodies against ICAM-1. Ann Surg. 1999;229:834-40; discussion 841-2. [PubMed] |
| 184. | Wang X, Sun Z, Börjesson A, Andersson R. Inhibition of platelet-activating factor, intercellular adhesion molecule 1 and platelet endothelial cell adhesion molecule 1 reduces experimental pancreatitis-associated gut endothelial barrier dysfunction. Br J Surg. 1999;86:411-416. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 40] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 185. | Hartwig W, Jimenez RE, Fernandez-del Castillo C, Kelliher A, Jones R, Warshaw AL. Expression of the adhesion molecules Mac-1 and L-selectin on neutrophils in acute pancreatitis is protease- and complement-dependent. Ann Surg. 2001;233:371-378. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 36] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 186. | Abdulla A, Awla D, Hartman H, Weiber H, Jeppsson B, Regnér S, Thorlacius H. Platelets regulate P-selectin expression and leukocyte rolling in inflamed venules of the pancreas. Eur J Pharmacol. 2012;682:153-160. [PubMed] |
| 187. | Hartman H, Abdulla A, Awla D, Lindkvist B, Jeppsson B, Thorlacius H, Regnér S. P-selectin mediates neutrophil rolling and recruitment in acute pancreatitis. Br J Surg. 2012;99:246-255. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 53] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 188. | Gloor B, Blinman TA, Rigberg DA, Todd KE, Lane JS, Hines OJ, Reber HA. Kupffer cell blockade reduces hepatic and systemic cytokine levels and lung injury in hemorrhagic pancreatitis in rats. Pancreas. 2000;21:414-420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 81] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 189. | Gea-Sorlí S, Closa D. In vitro, but not in vivo, reversibility of peritoneal macrophages activation during experimental acute pancreatitis. BMC Immunol. 2009;10:42. [PubMed] |
| 190. | Nakamichi I, Habtezion A, Zhong B, Contag CH, Butcher EC, Omary MB. Hemin-activated macrophages home to the pancreas and protect from acute pancreatitis via heme oxygenase-1 induction. J Clin Invest. 2005;115:3007-3014. [PubMed] |
| 191. | Habtezion A, Kwan R, Akhtar E, Wanaski SP, Collins SD, Wong RJ, Stevenson DK, Butcher EC, Omary MB. Panhematin provides a therapeutic benefit in experimental pancreatitis. Gut. 2011;60:671-679. [PubMed] |
| 192. | Jung KH, Song SU, Yi T, Jeon MS, Hong SW, Zheng HM, Lee HS, Choi MJ, Lee DH, Hong SS. Human bone marrow-derived clonal mesenchymal stem cells inhibit inflammation and reduce acute pancreatitis in rats. Gastroenterology. 2011;140:998-1008. [PubMed] |
| 193. | Schneider G, Saur D. Mesenchymal stem cells: therapeutic potential for acute pancreatitis. Gastroenterology. 2011;140:779-782. [PubMed] |
| 194. | Gloor B, Uhl W, Tcholakov O, Roggo A, Muller CA, Worni M, Büchler MW. Hydrocortisone treatment of early SIRS in acute experimental pancreatitis. Dig Dis Sci. 2001;46:2154-2161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 40] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 195. | Lázár G, Varga J, Lázár G, Duda E, Takács T, Balogh A, Lonovics J. The effects of glucocorticoids and a glucocorticoid antagonist (RU 38486) on experimental acute pancreatitis in rat. Acta Chir Hung. 1997;36:190-191. [PubMed] |
| 196. | Barzilai A, Ryback BJ, Medina JA, Toth L, Dreiling DA. The morphological changes of the pancreas in hypovolemic shock and the effect of pretreatment with steroids. Int J Pancreatol. 1987;2:23-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 197. | Budzyńska A, Marek T, Nowak A, Kaczor R, Nowakowska-Dulawa E. A prospective, randomized, placebo-controlled trial of prednisone and allopurinol in the prevention of ERCP-induced pancreatitis. Endoscopy. 2001;33:766-772. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 75] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 198. | De Palma GD, Catanzano C. Use of corticosteriods in the prevention of post-ERCP pancreatitis: results of a controlled prospective study. Am J Gastroenterol. 1999;94:982-985. [PubMed] |
| 199. | Sherman S, Blaut U, Watkins JL, Barnett J, Freeman M, Geenen J, Ryan M, Parker H, Frakes JT, Fogel EL. Does prophylactic administration of corticosteroid reduce the risk and severity of post-ERCP pancreatitis: a randomized, prospective, multicenter study. Gastrointest Endosc. 2003;58:23-29. [PubMed] |
| 200. | Matsumura M, Kakishita H, Suzuki M, Banba N, Hattori Y. Dexamethasone suppresses iNOS gene expression by inhibiting NF-kappaB in vascular smooth muscle cells. Life Sci. 2001;69:1067-1077. [PubMed] |
| 201. | van Leeuwen HJ, van der Bruggen T, van Asbeck BS, Boereboom FT. Effect of corticosteroids on nuclear factor-kappaB activation and hemodynamics in late septic shock. Crit Care Med. 2001;29:1074-1077. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 45] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 202. | Dunn JA, Li C, Ha T, Kao RL, Browder W. Therapeutic modification of nuclear factor kappa B binding activity and tumor necrosis factor-alpha gene expression during acute biliary pancreatitis. Am Surg. 1997;63:1036-1043; discussion 1043-1044. [PubMed] |
| 203. | Satoh A, Shimosegawa T, Fujita M, Kimura K, Masamune A, Koizumi M, Toyota T. Inhibition of nuclear factor-kappaB activation improves the survival of rats with taurocholate pancreatitis. Gut. 1999;44:253-258. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 109] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 204. | Yang S, Bing M, Chen F, Sun Y, Chen H, Chen W. Autophagy regulation by the nuclear factor κB signal axis in acute pancreatitis. Pancreas. 2012;41:367-373. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 16] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 205. | Sailai Y, Yu X, Baiheti P, Tang H, Li Y, Xu M. Influence of nuclear factor kappaB activation on inflammatory mediators of alveolar macrophages in rats with acute necrotizing pancreatitis. J Investig Med. 2010;58:38-42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 206. | Tsung A, Sahai R, Tanaka H, Nakao A, Fink MP, Lotze MT, Yang H, Li J, Tracey KJ, Geller DA. The nuclear factor HMGB1 mediates hepatic injury after murine liver ischemia-reperfusion. J Exp Med. 2005;201:1135-1143. [PubMed] |
| 207. | Ogawa EN, Ishizaka A, Tasaka S, Koh H, Ueno H, Amaya F, Ebina M, Yamada S, Funakoshi Y, Soejima J. Contribution of high-mobility group box-1 to the development of ventilator-induced lung injury. Am J Respir Crit Care Med. 2006;174:400-407. [PubMed] |
| 208. | Sawa H, Ueda T, Takeyama Y, Yasuda T, Shinzeki M, Nakajima T, Kuroda Y. Blockade of high mobility group box-1 protein attenuates experimental severe acute pancreatitis. World J Gastroenterol. 2006;12:7666-7670. [PubMed] |
| 209. | Sappington PL, Yang R, Yang H, Tracey KJ, Delude RL, Fink MP. HMGB1 B box increases the permeability of Caco-2 enterocytic monolayers and impairs intestinal barrier function in mice. Gastroenterology. 2002;123:790-802. [PubMed] |
| 210. | Sandoval D, Gukovskaya A, Reavey P, Gukovsky S, Sisk A, Braquet P, Pandol SJ, Poucell-Hatton S. The role of neutrophils and platelet-activating factor in mediating experimental pancreatitis. Gastroenterology. 1996;111:1081-1091. [PubMed] |
| 211. | Lankisch PG, Lerch MM. Pharmacological prevention and treatment of acute pancreatitis: where are we now? Dig Dis. 2006;24:148-159. [PubMed] |
| 212. | Andriulli A, Leandro G, Federici T, Ippolito A, Forlano R, Iacobellis A, Annese V. Prophylactic administration of somatostatin or gabexate does not prevent pancreatitis after ERCP: an updated meta-analysis. Gastrointest Endosc. 2007;65:624-632. [PubMed] |
| 213. | Pezzilli R, Miglioli M. Multicentre comparative study of two schedules of gabexate mesilate in the treatment of acute pancreatitis. Italian Acute Pancreatitis Study Group. Dig Liver Dis. 2001;33:49-57. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 214. | Büchler M, Malfertheiner P, Uhl W, Schölmerich J, Stöckmann F, Adler G, Gaus W, Rolle K, Beger HG. Gabexate mesilate in human acute pancreatitis. German Pancreatitis Study Group. Gastroenterology. 1993;104:1165-1170. [PubMed] |
| 215. | Matuschak GM. Circulating cytokine concentrations and outcome prediction in intensive care unit patients: still the tip of the iceberg? Crit Care Med. 1996;24:1769-1771. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 216. | Pezzilli R, Ceciliato R, Barakat B, Corinaldesi R. Immune-manipulation of the inflammatory response in acute pancreatitis. What can be expected? JOP. 2004;5:115-121. [PubMed] |
| 217. | Krause I, Valesini G, Scrivo R, Shoenfeld Y. Autoimmune aspects of cytokine and anticytokine therapies. Am J Med. 2003;115:390-397. [PubMed] |
| 218. | Werner J, Hartwig W, Hackert T, Kaiser H, Schmidt J, Gebhard MM, Büchler MW, Klar E. Multidrug strategies are effective in the treatment of severe experimental pancreatitis. Surgery. 2012;151:372-381. [PubMed] |
| 219. | Uomo G. Lesson from acute experimental pancreatitis: multidrug strategies is effective than single-target therapy. JOP. 2012;13:543-544. [PubMed] |
| 220. | Haraldsen P, Sun ZW, Börjesson A, Olanders K, Lasson A, Andersson R. Multimodal management - of value in fulminant acute pancreatitis? Pancreatology. 2003;3:14-25. [PubMed] |
| 221. | Denham W, Fink G, Yang J, Ulrich P, Tracey K, Norman J. Small molecule inhibition of tumor necrosis factor gene processing during acute pancreatitis prevents cytokine cascade progression and attenuates pancreatitis severity. Am Surg. 1997;63:1045-1049; discussion 1049-1050. [PubMed] |
| 222. | Pastor CM, Frossard JL. Are genetically modified mice useful for the understanding of acute pancreatitis? FASEB J. 2001;15:893-897. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 36] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 223. | Norman J. The role of cytokines in the pathogenesis of acute pancreatitis. Am J Surg. 1998;175:76-83. [PubMed] |
| 224. | Uhl W, Büchler MW, Malfertheiner P, Beger HG, Adler G, Gaus W. A randomised, double blind, multicentre trial of octreotide in moderate to severe acute pancreatitis. Gut. 1999;45:97-104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 119] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 225. | Kylanpaa ML, Mentula P, Kemppainen E, Puolakkainen P, Aittomaki S, Silvennoinen O, Haapiainen R, Repo H. Monocyte anergy is present in patients with severe acute pancreatitis and is significantly alleviated by granulocyte-macrophage colony-stimulating factor and interferon-gamma in vitro. Pancreas. 2005;31:23-27. [PubMed] |
| 226. | Nierhaus A, Montag B, Timmler N, Frings DP, Gutensohn K, Jung R, Schneider CG, Pothmann W, Brassel AK, Schulte Am Esch J. Reversal of immunoparalysis by recombinant human granulocyte-macrophage colony-stimulating factor in patients with severe sepsis. Intensive Care Med. 2003;29:646-651. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 102] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 227. | O’Reilly DA, Roberts JR, Cartmell MT, Demaine AG, Kingsnorth AN. Heat shock factor-1 and nuclear factor-kappaB are systemically activated in human acute pancreatitis. JOP. 2006;7:174-184. [PubMed] |
| 228. | Oiva J, Mustonen H, Kylänpää ML, Kyhälä L, Alanärä T, Aittomäki S, Siitonen S, Kemppainen E, Puolakkainen P, Repo H. Patients with acute pancreatitis complicated by organ failure show highly aberrant monocyte signaling profiles assessed by phospho-specific flow cytometry. Crit Care Med. 2010;38:1702-1708. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 28] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 229. | Oiva J, Mustonen H, Kylänpää ML, Kyhälä L, Kuuliala K, Siitonen S, Kemppainen E, Puolakkainen P, Repo H. Acute pancreatitis with organ dysfunction associates with abnormal blood lymphocyte signaling: controlled laboratory study. Crit Care. 2010;14:R207. [PubMed] |
| 230. | Imahara SD, O’Keefe GE. Genetic determinants of the inflammatory response. Curr Opin Crit Care. 2004;10:318-324. [PubMed] |
| 231. | Büchler M, Friess H, Uhl W, Beger HG. Clinical relevance of experimental acute pancreatitis. Eur Surg Res. 1992;24 Suppl 1:85-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 232. | Foitzik T, Hotz HG, Eibl G, Buhr HJ. Experimental models of acute pancreatitis: are they suitable for evaluating therapy? Int J Colorectal Dis. 2000;15:127-135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 31] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 233. | Mason J, Siriwardena AK. Designing future clinical trials in acute pancreatitis. Pancreatology. 2005;5:113-115. [PubMed] |
| 234. | Seta T, Noguchi Y, Shimada T, Shikata S, Fukui T. Treatment of acute pancreatitis with protease inhibitors: a meta-analysis. Eur J Gastroenterol Hepatol. 2004;16:1287-1293. [PubMed] |
| 235. | Abu-Zidan FM, Windsor JA. Lexipafant and acute pancreatitis: a critical appraisal of the clinical trials. Eur J Surg. 2002;168:215-219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 32] [Article Influence: 1.5] [Reference Citation Analysis (0)] |