Published online Apr 21, 2014. doi: 10.3748/wjg.v20.i15.4407
Revised: December 31, 2013
Accepted: February 17, 2014
Published online: April 21, 2014
Processing time: 285 Days and 8.1 Hours
AIM: To investigate hepatitis B surface antigen (HBsAg) levels in patients with HBeAg-positive chronic hepatitis B (CHB) and different immune conditions.
METHODS: HBeAg-positive CHB patients with different immune conditions were enrolled in this cross-sectional study. These patients were grouped according to the following criteria: immune-tolerant patients, IT group; patients with a mild immune response in the immune clearance phase, IC-Mild group; and patients with a dramatic immune response in the immune clearance phase and exhibiting acute on chronic liver failure (ACLF), ACLF group. All these patients had not previously received antiviral therapy and were enrolled at a pre-settled ratio of 2:2:1. Serum HBsAg levels and the correlation between serum HBsAg level and serum hepatitis B virus (HBV) DNA level were evaluated in these groups.
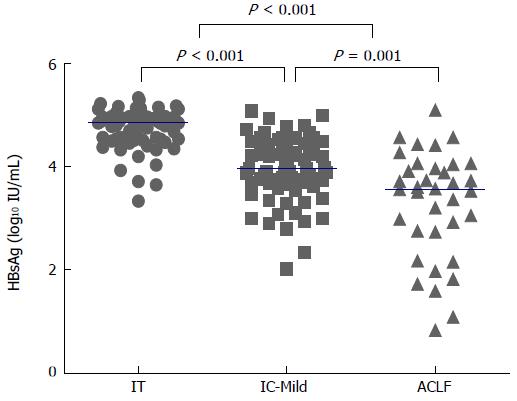
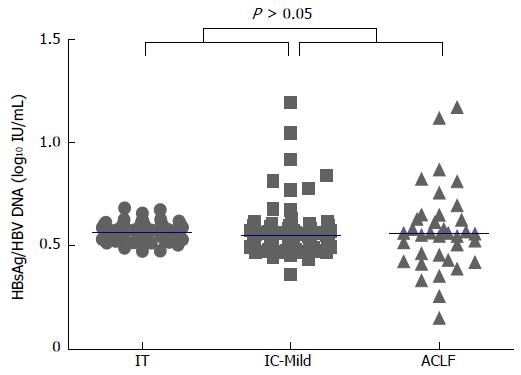
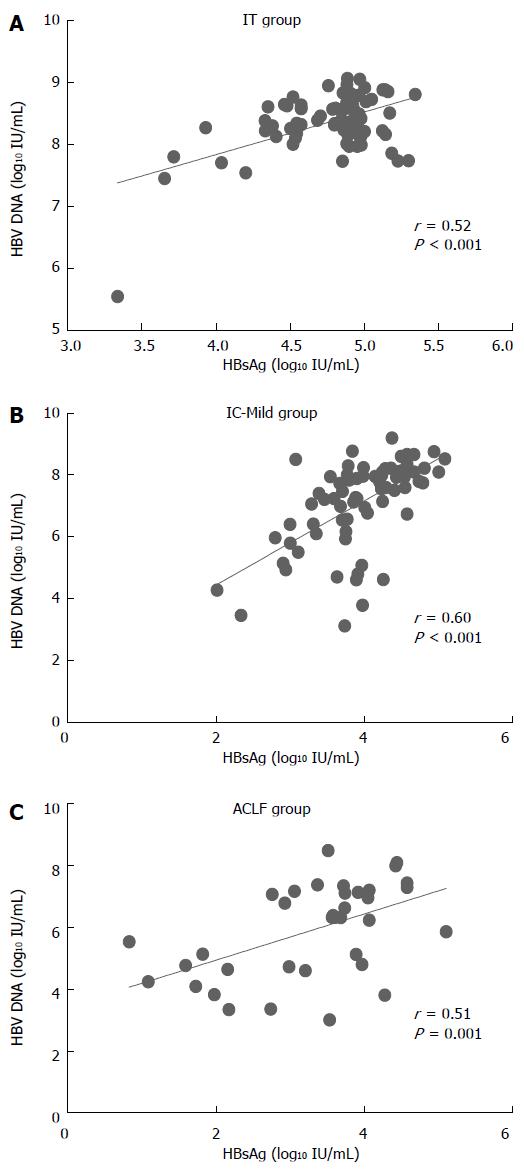
RESULTS: In total, 180 HBeAg-positive CHB patients [IT group (n = 72), IC-Mild group (n = 72), and ACLF group (n = 36)] were enrolled in this study. The median serum HBsAg levels varied among the groups (P < 0.001): IT, 4.86 log10 IU/mL; IC-Mild, 3.97 log10 IU/mL; and ACLF, 3.57 log10 IU/mL. Serum HBsAg level showed a moderate positive correlation with serum HBV-DNA level in the IC-Mild group (r = 0.60, P < 0.001), but exhibited a weaker correlation in the IT (r = 0.52, P < 0.001) and ACLF groups (r = 0.51, P = 0.001). The ratio of HBsAg/HBV DNA did not differ significantly among the IT, IC-Mild, and ACLF groups (medians: 0.56, 0.55, and 0.56, respectively; P = 0.179).
CONCLUSION: Serum HBsAg levels varied significantly in HBeAg-positive patients with different immune conditions. These findings may have important implications for understanding the immune clearance of HBV in HBeAg-positive CHB patients.
Core tip: This study showed that a dramatic immune response demonstrated that acute on chronic liver failure results in lower serum hepatitis B surface antigen levels compared with a mild immune response in patients in the immune clearance phase. These findings may have important implications for understanding the immune clearance of hepatitis B virus in these patients.
- Citation: Zhang YM, Yang YD, Jia HY, Zeng LY, Yu W, Zhou N, Li LJ. HBsAg levels in HBeAg-positive chronic hepatitis B patients with different immune conditions. World J Gastroenterol 2014; 20(15): 4407-4413
- URL: https://www.wjgnet.com/1007-9327/full/v20/i15/4407.htm
- DOI: https://dx.doi.org/10.3748/wjg.v20.i15.4407
Hepatitis B surface antigen (HBsAg) is considered a milestone in the research history of hepatitis B virus (HBV) and has been used for more than 40 years to confirm HBV infections[1]. With the second generation of standard HBsAg samples introduced by the World Health Organization, quantitative HBsAg tests have been performed[2,3].
Studies have shown the potential application of HBsAg quantification in predicting responses to interferon-based therapy and nucleos(t)ide therapy[4-7]. In addition, the quantified serum HBsAg level has been used as a potential surrogate or supplemental marker of HBV DNA to identify the phase of the natural history of the disease and to predict seroconversion in CHB[8,9].
In the HBeAg-positive condition, the natural course of CHB patients is divided into the immune tolerance and immune clearance phases. The serum HBsAg level is higher in the immune tolerance phase and declines in the immune clearance phase[10-12], indicating different immune conditions associated with various HBsAg levels.
In addition to the different immune conditions between the immune tolerance and immune clearance phases, the degree of host immune response is different when immune clearance begins[13,14]. In general, patients show moderately increased alanine aminotransferase (ALT) levels without hepatic decompensation, indicating a mild immune response. However, when a dramatic immune response occurs in the immune clearance phase, liver function decompensates or deteriorates to acute on chronic liver failure (ACLF), which is usually described as acute hepatic injury manifested as jaundice and coagulopathy[15,16]. Although all patients in the IC phase have been evaluated as a whole, the serum HBsAg levels in these patients with different immune responses in the immune clearance phase have not been evaluated individually. Lee et al[17] showed that baseline serum HBsAg levels can be used as a prognostic factor of the anti-HBV treatment response in HBeAg-positive CHB patients during the immune clearance phase. Thus, studies should be conducted to investigate whether a dramatic immune response exhibiting ACLF will yield different HBsAg levels compared with a mild immune response in patients in the immune clearance phase, which may indicate anti-viral therapy results. In addition, the HBsAg levels should be compared with those in immune-tolerant patients to determine whether different characteristics are observed when patients in the immune clearance phase are subdivided according to different immune clearance conditions.
Therefore, three groups of HBeAg-positive CHB patients were enrolled in this study: immune-tolerant patients, IT group; patients with a mild immune response in the immune clearance phase, IC-Mild group; and patients with a dramatic immune response in the immune clearance phase and exhibiting ACLF, ACLF group. The HBsAg levels were compared among these three groups.
In different phases of the HBV infection, the connection between HBsAg production and HBV replication is not stable[2]. This study also aimed to evaluate the correlation between serum HBsAg levels and serum HBV DNA levels among these three groups.
Three groups of HBeAg-positive CHB patients with different immune conditions were recruited into this cross-sectional study. Each group was classified as follows: IT group, high HBV DNA levels and ALT < upper limit of normal (ULN); IC-Mild group, increased HBV DNA levels, 2 ULN < ALT < 10 ULN, total bilirubin (TB) < 2 ULN (42 μmol/L), international normalized ratio (INR) (a laboratory test representing coagulopathy) < 1.27, and approximate prothrombin time activity percentage (PTA) > 60%; ACLF group, increased HBV DNA levels, ALT > 2 ULN, extreme fatigue with severe digestive symptoms, serum TB level > 171 μmol/L, INR > 1.50, and approximate PTA < 40%.
Patient history showed that all patients had not received antiviral therapy and were HBsAg positive for longer than six months. The patients had no clinical or ultrasonographic findings of cirrhosis. The patients were recruited from the First Affiliated Hospital, College of Medicine, Zhejiang University with a pre-settled ratio of 2:2:1 in the IT, IC-Mild, and ACLF groups, respectively, from October 2011 to May 2013. The target number of patients in each group was estimated according to the serum HBsAg levels of the first 50 patients enrolled (data not shown). In this study, the ULN of ALT was 40 U/L, whereas the ULN of TB was 21 μmol/L.
Patients in the IT group met the criteria of the IT phase according to the Asian-Pacific Consensus Statement on the Management of Chronic Hepatitis B: A 2008 Update[14]. Patients in the IC-Mild group represented patients without liver function decompensation in the IC phase, and this definition is consistent with the same consensus statement regarding CHB. Patients in the ACLF group met the criteria according to the ACLF consensus recommendations of the Asian Pacific Association for the Study of the Liver[15].
None of the enrolled patients suffered from co-infection with viruses, such as hepatitis A, hepatitis C, hepatitis D, hepatitis E, or human immunodeficiency viruses. Patients with cirrhosis, drug-induced liver injury, autoimmune hepatitis, alcoholic liver diseases, genetic and other liver diseases, chemo/immunotherapy, or cancer were excluded. All pediatric patients were also excluded. For the IT and ACLF patients, serum HBsAg and HBV DNA levels were determined once at the time of enrollment. IC-Mild patients had a median follow-up of three weeks (minimum 2 wk, maximum 8 wk); serum HBsAg and HBV DNA levels were determined once at the time of enrollment and repeated at least once during the follow-up period. Serum samples for the HBsAg and HBV DNA tests were collected at the same time points during enrollment or follow-up. The patients who experienced liver function decompensation, but did not meet the criteria of the IC-Mild group were excluded.
Serum HBsAg levels were compared among the three groups in this study. The correlation between serum HBsAg level and serum HBV DNA level was determined.
This study was conducted according to the guidelines of the Declaration of Helsinki and was approved by the Review Board of the Ethics Committee of the First Affiliated Hospital, College of Medicine, Zhejiang University. Written informed consent was obtained from the patients or their relatives.
Biochemical parameters were analyzed by routine automated methods (Hitachi 7600, Japan). HBeAg was qualitatively analyzed, and HBsAg levels were quantitatively analyzed using the Abbott ARCHITECT assay (Abbott Diagnostics, Germany) according to the manufacturer’s instructions. The dynamic range of HBsAg levels was 0.05-250 IU/mL. The samples were diluted to 1:500 or 1:1000 using ARCHITECT HBsAg Manual Diluent (Abbott Diagnostics) if the HBsAg levels were greater than 250 IU/mL. Serum HBV DNA was quantified using a determination kit for HBV DNA (Life River, China) at a detection range of 5 × 102-1 × 108 IU/mL; for the samples with > 108 IU/mL HBV DNA levels, the HBV DNA assay was repeated after dilution to 1:1000.
The data are presented as medians (interquartile range), except when stated otherwise. The Kruskal-Wallis test was performed for multivariate comparisons. The Mann-Whitney test and Fisher’s exact test were performed for univariate comparisons, as appropriate. The Spearman rank test was conducted for correlation analyses. Multivariate linear regression with the HBsAg concentration as an independent factor was performed. Statistical calculations were performed using GraphPad Prism (Version 5.0, GraphPad Software, San Diego, United States). A P value of 0.05 (two-tailed) was considered significant.
In total, 180 HBeAg-positive CHB patients were enrolled in this cross-sectional study (72 in the IT group, 72 in the IC-Mild group, and 36 in the ACLF group). A predominance of males (131/180, 72.8%) was observed in the study population. The patient characteristics, including age, gender, serum HBV DNA levels, serum HBsAg levels, ALT, TB, INR, and platelets, are described in detail in Table 1. All these parameters were significantly different among the IT, IC-Mild, and ACLF groups.
| Variables | IT group (n = 72) | IC-Mild group (n = 72) | ACLF group (n = 36) | Kruskal-Wallis test P value |
| Age (yr) | 25 (22-29) | 30 (24-37) | 34 (28-41) | < 0.001 |
| Gender (male/female) | 44/28 | 55/17 | 32/4 | 0.006 |
| HBV DNA (log10 IU/mL) | 8.39 (8.15-8.65) | 7.58 (7.11-8.79) | 6.33 (4.67-7.17) | < 0.001 |
| HBsAg (log10 IU/mL) | 4.86 (4.54-4.97) | 3.97 (3.67-4.43) | 3.57 (2.74-4.03) | < 0.001 |
| ALT (IU/mL) | 25 (19-32) | 220 (118-282) | 468 (244-779) | < 0.001 |
| TB (μmol/L) | 11 (9-15) | 16 (10-29) | 314 (248-443) | < 0.001 |
| INR | 1.01 (0.81-1.07) | 1.08 (0.85-1.17) | 2.24 (1.74-2.71) | < 0.001 |
| PLT (109/L) | 210 (187-273) | 172 (152-298) | 124 (103-167) | < 0.001 |
The distribution of serum HBsAg levels in the three groups was evaluated as described in Figure 1. The median serum HBsAg levels in each group were as follows: IT group, 4.86 log10 IU/mL; IC-Mild group, 3.97 log10 IU/mL; and ACLF group, 3.57 log10 IU/mL. The serum HBsAg levels were significantly different in each group (IT vs IC-Mild, P < 0.001; IT vs ACLF, P < 0.001; IC-Mild vs ACLF, P = 0.001).
The HBsAg/HBV DNA ratio was evaluated (Figure 2). The median levels of the HBsAg/HBV DNA ratio in the IT, IC-Mild, and ACLF groups were 0.56, 0.55, and 0.56, respectively. The results showed no significant differences among these three groups (P = 0.179).
The correlations between serum HBsAg levels and HBV DNA levels in each group are shown in Figure 3. A modest correlation was found between HBsAg and HBV DNA in the IC-Mild group (r = 0.60, P < 0.001). Serum HBsAg level was weakly correlated with HBV DNA in the IT group (r = 0.52, P < 0.001) and the ACLF group (r = 0.51, P < 0.001) compared with the IC-Mild group.
Several research programs have been established to investigate HBsAg levels in the natural history of CHB in Asian and European ethnicities[10-12]. With regard to HBeAg-positive status, a higher and more stable titer of serum HBsAg (median 4.5-4.9 log10 IU/mL) is observed in the IT phase when the host’s immune response against HBV-infected hepatocytes has not been triggered[10-12]. When immune clearance has been triggered and a fraction of the HBV-infected hepatocytes has been destroyed, decreased serum HBsAg levels have been found in the IC phase (median 3.7-4.3 log10 IU/mL)[10-12], which was proven in our study. The degree of a host’s immune response has been shown to be different when immune clearance begins. However, scientists have often considered all patients in the IC phase as a whole or have excluded ACLF patients from their studies[10-12]. This study is the first to subdivide HBeAg-positive CHB patients into groups according to the different immune responses. The results revealed that the median level of serum HBsAg was significantly lower in the ACLF group than in the IC-Mild group (3.57 log10 IU/mL vs 3.97 log10 IU/mL). The explanation for this difference may be the dramatic immune response in the ACLF group compared with the mild immune response in the IC-Mild group. Although the HBsAg clearance mechanism is complex in HBeAg-positive ACLF patients, several clues have revealed that the mechanism appears related to the remarkable immune response in which pro-inflammatory cytokines play an important role[18].
Changes in serum HBsAg levels in HBeAg-positive CHB patients with different immune conditions may provide useful information regarding the HBV life cycle. Chan et al[10] performed a longitudinal study and showed that the median serum HBsAg level in IC patients was similar before HBeAg seroconversion was achieved. Another study demonstrated that the early decrease in the amount of HBsAg in HBeAg-positive patients can predict sustained response to pegylated interferon. Pegylation enhances the half-life of interferon compared with its native form[5]. In this study, a dramatic immune response in the ACLF group caused a significant reduction in HBsAg levels compared with the IC-Mild group. The decreased HBsAg levels in the ACLF group suggest that HBeAg seroconversion may be achieved after this dramatic immune response. For these reasons, longitudinal follow-up is recommended.
HBsAg synthesis during persistent HBV infection is complex and differs from HBV DNA synthesis. HBsAg is synthesized in the endoplasmic reticulum and from HBV DNA integrated into a host’s genome[19]. Three forms of HBsAg are found in serum HBsAg: part of an HBV virion, the filament, and spheroid particles; the amount of HBsAg produced is usually 102-105 greater[19] than the value required for virion assembly[2]. The methods used to quantify serum HBsAg cannot differentiate these three forms. Furthermore, the synthesis ratio of these three forms of HBsAg is complex in the natural history of CHB. Previous studies showed a strong correlation between HBsAg production and HBV DNA replication in the early phase of immune clearance, such as in acute hepatitis B[11] or in patients responding to Peg-IFN treatments[20]. In addition, a positive association was observed between serum HBsAg and HBV DNA in the IC phase in patients with genotypes B, C, and D according to previous studies[2]. Patients in China predominantly exhibit genotype B or C, and thus, we also found a positive correlation between serum HBsAg and HBV DNA in the IC-Mild group (r = 0.60, P < 0.001). Studies demonstrating the relationship between HBsAg and HBV DNA in the IT phase are limited. For example, Nguyen et al[12] reported that HBsAg production is poorly correlated with viral replication (r = 0.30, P = 0.09). Our study showed a stronger correlation between HBsAg and HBV DNA in the IT patients (r = 0.52, P < 0.001). This result may be attributed to the unavailable HBV genotype composition of the patients and the relatively large number of patients enrolled in the IT group compared with those in previous studies.
Early studies described rapid viral clearance with undetectable serum HBsAg and HBV DNA in HBV-related liver failure[21,22]. However, the test methods for HBsAg and HBV DNA are qualitative. A recent study noted a consequential, rapid decrease in the HBV viral load within four days in chronic hepatitis B-induced liver failure[23]. A positive correlation between HBsAg and HBV DNA in ACLF patients was found (r = 0.51, P = 0.001) in our study. This result is possible evidence that the serum HBsAg level rapidly decreases in ACLF as the HBV DNA rapidly declines.
The ratio of HBsAg/HBV DNA is stable in HBeAg-positive patients before HBeAg seroconversion, indicating a stable correlation between virus replication and HBsAg secretion[24]. This stable correlation was proven in our study. The HBsAg/HBV DNA ratios were approximately 0.55 in the three groups in this study.
This study also has certain limitations. First, it is a cross-sectional study, whereas a longitudinal investigation would be more useful. However, long-term follow-up without the influence of anti-HBV therapy is difficult because antiviral therapy was administered to almost all patients in the IC-Mild or ACLF groups. Second, the HBV genotype cannot be used in this study. Investigations should also be conducted to determine the influence of genotypes on the production of HBsAg under different immune conditions. In addition, the distribution of serum HBsAg levels does not show significant differences in genotype B or C HBeAg-positive patients[12]. This finding partly resolves such limitations.
In conclusion, serum HBsAg levels are significantly different in HBeAg-positive CHB patients with different immune conditions. In particular, the highest, moderately high, and lowest HBsAg levels were found in the IT, IC-Mild, and ACLF groups, respectively. These findings may have important implications in elucidating the immune clearance and sequential immune control of HBV infection in HBeAg-positive CHB.
Quantitative serum Hepatitis B surface antigen (HBsAg) levels have been characterized in four typical phases of the natural history of chronic hepatitis B (CHB), indicating the dynamic interaction between hepatitis B virus and its host’s immune system. However, the severity of the immune response is different in the immune clearance phase, and HBsAg levels have not been evaluated in these subdivided patients and compared with those in immune-tolerant patients.
Studies on HBsAg quantification should focus on predicting the responses to antiviral therapy according to the baseline HBsAg levels or according to changes in the HBsAg levels during therapy. Therefore, studies should be conducted to investigate the HBsAg levels under different immune conditions during the natural history of CHB. In this way, a link between the immune reaction and antiviral efficacy might be revealed.
In this study, HBsAg levels in HBeAg-positive patients who met the criteria of acute on chronic liver failure were evaluated for the first time. The HBsAg levels were lower in these patients than in patients without evident liver decompensation in the immune clearance phase.
These findings may have important implications in elucidating the immune clearance and sequential immune control of HBV infection in HBeAg-positive CHB.
HBsAg is a surface antigen of the hepatitis B virus. HBsAg has been used as a hallmark of hepatitis B infection for more than 40 years.
This paper reported different serum HBsAg levels in patients with HBeAg-positive chronic hepatitis B under different immune conditions. It also showed a modest correlation between the HBsAg levels and HBV DNA levels under these different immune conditions. Basically, the manuscript is well written, and the conclusion is clear.
P- Reviewers: Marcos M, Nagata T, Sanai FM S- Editor: Zhai HH L- Editor: Webster JR E- Editor: Zhang DN
| 1. | Blumberg BS, Alter HJ, Visnich S. A “new” antigen in leukemia sera. JAMA. 1965;191:541-546. [PubMed] |
| 2. | Brunetto MR. A new role for an old marker, HBsAg. J Hepatol. 2010;52:475-477. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 3. | Sonneveld MJ, Zoutendijk R, Janssen HL. Hepatitis B surface antigen monitoring and management of chronic hepatitis B. J Viral Hepat. 2011;18:449-457. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 43] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 4. | Wursthorn K, Jung M, Riva A, Goodman ZD, Lopez P, Bao W, Manns MP, Wedemeyer H, Naoumov NV. Kinetics of hepatitis B surface antigen decline during 3 years of telbivudine treatment in hepatitis B e antigen-positive patients. Hepatology. 2010;52:1611-1620. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 163] [Cited by in RCA: 174] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 5. | Sonneveld MJ, Rijckborst V, Boucher CA, Hansen BE, Janssen HL. Prediction of sustained response to peginterferon alfa-2b for hepatitis B e antigen-positive chronic hepatitis B using on-treatment hepatitis B surface antigen decline. Hepatology. 2010;52:1251-1257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 227] [Cited by in RCA: 226] [Article Influence: 15.1] [Reference Citation Analysis (0)] |
| 6. | Rijckborst V, Hansen BE, Cakaloglu Y, Ferenci P, Tabak F, Akdogan M, Simon K, Akarca US, Flisiak R, Verhey E, Van Vuuren AJ, Boucher CA, ter Borg MJ, Janssen HL. Early on-treatment prediction of response to peginterferon alfa-2a for HBeAg-negative chronic hepatitis B using HBsAg and HBV DNA levels. Hepatology. 2010;52:454-461. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 192] [Cited by in RCA: 185] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 7. | Chen J, Wang Z, Guo Y, Peng J, Sun J, Ahmed CS, Zhou Y, Hou J. Serum HBsAg changes in HBeAg positive chronic hepatitis B patients with continuous viral load reductions during treatment with adefovir or peg-interferon-alpha-2a. Antiviral Res. 2009;81:88-91. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 8. | Brunetto MR, Oliveri F, Colombatto P, Moriconi F, Ciccorossi P, Coco B, Romagnoli V, Cherubini B, Moscato G, Maina AM. Hepatitis B surface antigen serum levels help to distinguish active from inactive hepatitis B virus genotype D carriers. Gastroenterology. 2010;139:483-490. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 284] [Cited by in RCA: 306] [Article Influence: 20.4] [Reference Citation Analysis (0)] |
| 9. | Tseng TC, Liu CJ, Su TH, Wang CC, Chen CL, Chen PJ, Chen DS, Kao JH. Serum hepatitis B surface antigen levels predict surface antigen loss in hepatitis B e antigen seroconverters. Gastroenterology. 2011;141:517-525, 525.e1-2. [PubMed] |
| 10. | Chan HL, Wong VW, Wong GL, Tse CH, Chan HY, Sung JJ. A longitudinal study on the natural history of serum hepatitis B surface antigen changes in chronic hepatitis B. Hepatology. 2010;52:1232-1241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 192] [Cited by in RCA: 201] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 11. | Jaroszewicz J, Calle Serrano B, Wursthorn K, Deterding K, Schlue J, Raupach R, Flisiak R, Bock CT, Manns MP, Wedemeyer H. Hepatitis B surface antigen (HBsAg) levels in the natural history of hepatitis B virus (HBV)-infection: a European perspective. J Hepatol. 2010;52:514-522. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 313] [Cited by in RCA: 313] [Article Influence: 20.9] [Reference Citation Analysis (0)] |
| 12. | Nguyen T, Thompson AJ, Bowden S, Croagh C, Bell S, Desmond PV, Levy M, Locarnini SA. Hepatitis B surface antigen levels during the natural history of chronic hepatitis B: a perspective on Asia. J Hepatol. 2010;52:508-513. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 264] [Cited by in RCA: 280] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 13. | Liaw YF. Hepatitis flares and hepatitis B e antigen seroconversion: implication in anti-hepatitis B virus therapy. J Gastroenterol Hepatol. 2003;18:246-252. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 125] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 14. | Liaw YF, Leung N, Kao JH, Piratvisuth T, Gane E, Han KH, Guan R, Lau GK, Locarnini S. Asian-Pacific consensus statement on the management of chronic hepatitis B: a 2008 update. Hepatol Int. 2008;2:263-283. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 666] [Cited by in RCA: 743] [Article Influence: 43.7] [Reference Citation Analysis (0)] |
| 15. | Sarin SK, Kumar A, Almeida JA, Chawla YK, Fan ST, Garg H, de Silva HJ, Hamid SS, Jalan R, Komolmit P. Acute-on-chronic liver failure: consensus recommendations of the Asian Pacific Association for the study of the liver (APASL). Hepatol Int. 2009;3:269-282. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 630] [Cited by in RCA: 643] [Article Influence: 40.2] [Reference Citation Analysis (0)] |
| 16. | Liver failure and Artificial Liver Group CSoID, Severe Liver Disease and Artificial Liver Group, Chinese Society of Hepatology. Guideline for diagnosis and treatment of liver failure: update 2012. Zhonghua Linchuang Ganranbing Zazhi. 2012;5:321-327. |
| 17. | Lee JM, Ahn SH, Kim HS, Park H, Chang HY, Kim do Y, Hwang SG, Rim KS, Chon CY, Han KH. Quantitative hepatitis B surface antigen and hepatitis B e antigen titers in prediction of treatment response to entecavir. Hepatology. 2011;53:1486-1493. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 113] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 18. | Rolando N, Wade J, Davalos M, Wendon J, Philpott-Howard J, Williams R. The systemic inflammatory response syndrome in acute liver failure. Hepatology. 2000;32:734-739. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 535] [Cited by in RCA: 507] [Article Influence: 20.3] [Reference Citation Analysis (0)] |
| 19. | Seeger C, Mason WS. Hepatitis B virus biology. Microbiol Mol Biol Rev. 2000;64:51-68. [PubMed] |
| 20. | Moucari R, Mackiewicz V, Lada O, Ripault MP, Castelnau C, Martinot-Peignoux M, Dauvergne A, Asselah T, Boyer N, Bedossa P. Early serum HBsAg drop: a strong predictor of sustained virological response to pegylated interferon alfa-2a in HBeAg-negative patients. Hepatology. 2009;49:1151-1157. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 40] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 21. | Krogsgaard K, Kryger P, Aldershvile J, Andersson P, Brechot C. Hepatitis B virus DNA in serum from patients with acute hepatitis B. Hepatology. 1985;5:10-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 40] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 22. | Trepo CG, Robert D, Motin J, Trepo D, Sepetjian M, Prince AM. Hepatitis B antigen (HBSAg) and/or antibodies (anti-HBS and anti-HBC) in fulminant hepatitis: pathogenic and prognostic significance. Gut. 1976;17:10-13. [PubMed] |
| 23. | Dao DY, Hynan LS, Yuan HJ, Sanders C, Balko J, Attar N, Lok AS, Word RA, Lee WM. Two distinct subtypes of hepatitis B virus-related acute liver failure are separable by quantitative serum immunoglobulin M anti-hepatitis B core antibody and hepatitis B virus DNA levels. Hepatology. 2012;55:676-684. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 43] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 24. | Chan HL, Wong VW, Tse AM, Tse CH, Chim AM, Chan HY, Wong GL, Sung JJ. Serum hepatitis B surface antigen quantitation can reflect hepatitis B virus in the liver and predict treatment response. Clin Gastroenterol Hepatol. 2007;5:1462-1468. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 270] [Cited by in RCA: 286] [Article Influence: 15.9] [Reference Citation Analysis (0)] |