Published online Sep 15, 1996. doi: 10.3748/wjg.v2.i3.152
Revised: July 25, 1996
Accepted: August 16, 1996
Published online: September 15, 1996
AIM: The relationship between Helicobacter pylori (Hp) and gastric epithelia in chronic gastritis and in peptic ulcers was studied by transmission electron microscopy (TEM).
METHODS: Seventy-five patients were screened for Hp. Gastric antral biopsy specimens were fixed in glutaraldehyde and treated with tannic acid before OsO4 staining. Samples were routinely processed for TEM studies (at least four semi-thin sections oriented for ultrathin sections in each sample).
RESULTS: The bacilli were detected by TEM within the gastric mucosa in 53 of 55 patients infected with Hp. Ultrathin sections revealed clear glycocalyx by which the bacillus was connected to the epithelium. As the bacilli colonized, the adjacent mucous cells degenerated. They were characterized by erosion of the juxtaluminal cytoplasm, vacuolation or blebbing, and desquamation of the cell membrane. The bacilli located in the lumen attracted neutrophils, which migrated into intercellular space of the epithelia or into the lumen to begin phagocytosis of Hp.
CONCLUSION: The sensitivity and specificity of TEM diagnosis is 96% and 95%, respectively. Tannic acid is suitable for the preservation of the glycocalyx of a cell. The colonized bacilli, usually with the wide periplasm, contributed to the degeneration of epithelia, including mucous neck cells. If Hp infection persists, the degeneration and regeneration of mucous neck cells occurs alternatively. Ultimately the generative stem cells were damaged, and as a result chronic atrophic gastritis could occur.
- Citation: Yang SM, Lin BZ, Fang Y, Zheng Y. Ultrastructural observation of Helicobacter pylori to the gastric epithelia in chronic gastritis and peptic ulcers. World J Gastroenterol 1996; 2(3): 152-154
- URL: https://www.wjgnet.com/1007-9327/full/v2/i3/152.htm
- DOI: https://dx.doi.org/10.3748/wjg.v2.i3.152
Ultrastructural studies have demonstrated a loss of apical microvilli and depletion of mucin granules in Helicobacter pylori (Hp) infected cells[1,2]. Further investigation of Hp adherence, cell penetration, and immune response of the gastric epithelia is needed to delineate the relationship between the bacilli and the epithelia or the monocytes.
Biopsies of the gastric antrum (four biopsies), the body (one biopsy), and serum from 75 patients with chronic gastritis or peptic ulcers were individually tested for Hp by culture, histopathology, cytology of smear, Hp rapid analytical chemistry urease kit (Hp-RACU), Hp enzyme linked immunosorbent analysis kit (Hp-EIA), and Hp polymerase chain reaction kit (Hp-PCR) (Cancer Research Center, Xiamen University). Positivity in more than three methods was considered the standard for detection of Hp infection. One of antral biopsies was also examined by a JEM 100CX/II transmission electron microscope (TEM). Biopsies were prefixed in 2.5% glutaraldehyde, treated for 4 h in 1% tannic acid (except for ten samples), then post-fixed and stained in a 1% osmium tetroxide and a 1% potassium ferricyanide mixture. Ultrathin sections (90 nm) were selected from the oriented semi-thin sections (at least four blocks/sample), and were examined by TEM for the relationship between the bacilli and the gastric epithelia.
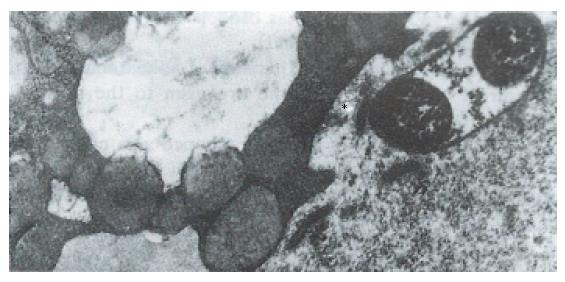
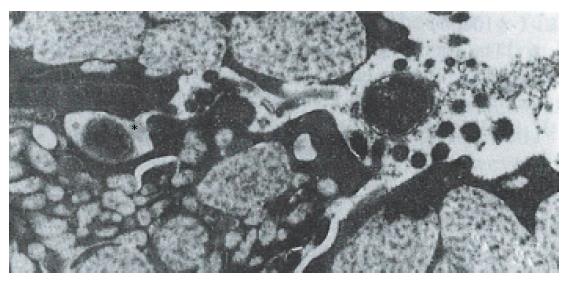
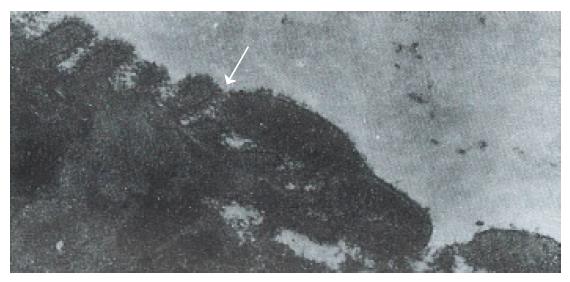
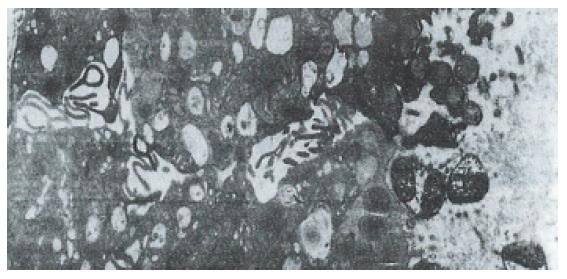
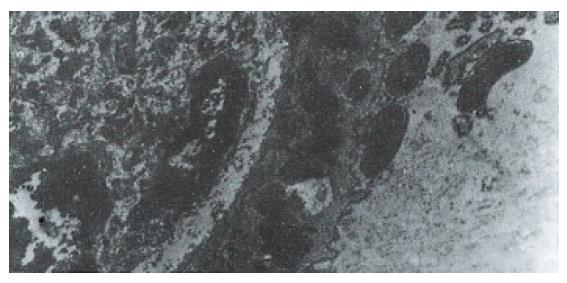
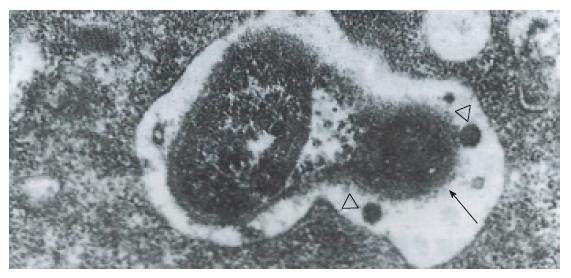
Hp appeared dense and opaque under the microscope. An electron-lucent zone in the mucin pool usually surrounded curved or spiral bodies. They were also located near the microvilli of epithelial cells, typically in the gastric neck region or adjacent to a depression in the plasma membrane that resulted from a lost microvillus (Figure 1) or apocrine secretion. Hp bacilli were found disrupting the cellular junction of rolled microvilli and fragmentated cells, and were found to be insinuated deep in between the epithelial cells (Figure 2). Hp infection was confirmed in 55 (30 chronic gastritis and 25 peptic ulcer) patients out of 75 patients. Curved or spiral organisms were detected in 53 patients (with one false positive) by TEM in the gastric mucosa (96%). Specificity of TEM was 95%, with 19 true negative and one false negative. After tannic acid staining, the glycocalyx was observed at the surface of the organisms, especially on the tip of the microvilli (Figure 3). Where the bacilli colonized, the adjacent mucous cells degenerated and were characterized by vacuolation (Figure 4), accumulation of lysosomes, and the appearance of mucous apocrine or blebs displaying a myeline figure. The bacilli located in the lumen attracted polymorphonuclear leukocytes (PMNL) that migrated into the intercellular space of the epithelia (Figure 5), or into the lumen to exert the effect of Hp phagocytosis (Figure 6).
In order to preserve delicate structures typically destroyed by osmium tetroxide, we fixed biopsies in a tannic acid solution. We found that some fragile glycocalyx on the bacilli and the epithelia remained intact (Figure 3). Based on our observation, where the bacilli colonized, mitotic figures and vacuolation of adjacent epithelial cells were present. A similar observation was noted by Caselli et al[3]. The bacilli firmly attached to the epithelium or hiding in the niche of injured cells may have released cytotoxins or vacuolotoxin to impair the cells[4]. As the organisms colonized, ammonia was released in order for the bacilli to escape the microbicidal effect for a long period of time.
Hp can be cleared by neutrophils from the site of infection most effectively through the opsonophagocytotic process in vivo and in vitro. Caselli et al[5] demonstrated the specific IgG antibody-promoted complement-dependent phagocytosis and killing of Hp by PMNL in vitro. Until now, the evidence of Hp phagocytosis by PMNL in antrum in vivo was scant. It is known that a certain protein or chemotactic factor of Hp[6] can attract neutrophils to migrate into the epithelia or the gland and begin phagocytosis. Degranulation of proteolytic enzymes from neutrophils may also contribute to gastric mucosa damage, especially in the cells of the generative mucous neck region. As a result of persistent Hp infection, the degeneration and regeneration of the mucous neck region alternatively occurs and gives rise to atrophic gastritis. Regeneration of the stem cells of the mucous neck region would ultimately promote gene instability, which accounts for genetic mutation in neoplastic transformation in gastric stem cells.
Original title:
S- Editor: Yang ZD L- Editor: Filipodia E- Editor: Li RF
| 1. | Kazi JL, Sinniah R, Zaman V, Ng ML, Jafarey NA, Alam SM, Zuberi SJ, Kazi AM. Ultrastructural study of Helicobacter pylori-associated gastritis. J Pathol. 1990;161:65-70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 34] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 2. | Hopwood D, Milne G, Penston J. Leakiness of gastric superficial and foveolar cells. A quantitative electron microscopic study using tannic acid. J Pathol. 1991;165:119-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 3. | Caselli M, Aleotti A, Boldrini P, Ruina M, Alvisi V. Ultrastructural patterns of Helicobacter pylori. Gut. 1993;34:1507-1509. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 4. | Cover TL, Cao P, Lind CD, Tham KT, Blaser MJ. Correlation between vacuolating cytotoxin production by Helicobacter pylori isolates in vitro and in vivo. Infect Immun. 1993;61:5008-5012. [PubMed] |
| 5. | Caselli M, Figura N, Trevisani L, Pazzi P, Guglielmetti P, Bovolenta MR, Stabellini G. Patterns of physical modes of contact between Campylobacter pylori and gastric epithelium: implications about the bacterial pathogenicity. Am J Gastroenterol. 1989;84:511-513. [PubMed] |
| 6. | Kozol R, McCurdy B, Czanko R. A neutrophil chemotactic factor present in H. pylori but absent in H. mustelae. Dig Dis Sci. 1993;38:137-141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 20] [Article Influence: 0.6] [Reference Citation Analysis (0)] |