Published online Dec 21, 2013. doi: 10.3748/wjg.v19.i47.8808
Revised: October 31, 2013
Accepted: November 18, 2013
Published online: December 21, 2013
Processing time: 112 Days and 5.7 Hours
Surveillance for hepatocellular carcinoma (HCC) is considered a standard of care for patients with chronic liver disease who are at risk of developing this malignancy. Several studies have shown that surveillance can improve the prognosis of patients diagnosed with HCC through an increased likelihood of application of curative or effective treatments. Repetition of liver ultrasonography (US) every 6 mo is the recommended surveillance program to detect early HCCs, and a positive US has to entrain a well-defined recall policy based on contrast-enhanced, dynamic radiological imaging or biopsy for the diagnosis of HCC. Although HCC fulfills the accepted criteria regarding cost-effective cancer screening and surveillance, the implementation of surveillance in clinical practice is defective and this has a negative impact on the cost-effectiveness of the procedure. Education of both physicians and patients is of paramount importance in order to improve the surveillance application and its benefits in patients at risk of HCC. The promotion of specific educational programs for practitioners, clinicians and patients is instrumental in order to expand the correct use of surveillance in clinical practice and eventually improve HCC prognosis.
Core tip: This article deals with the role of surveillance for early diagnosis of hepatocellular carcinoma in patients at risk. It addresses several topics on this issue, including how to best perform surveillance (tools and interval), its results in terms of cancer stage, patient survival, cost-effectiveness, pitfalls and actual under-(mis-)use.
- Citation: Giannini EG, Cucchetti A, Erroi V, Garuti F, Odaldi F, Trevisani F. Surveillance for early diagnosis of hepatocellular carcinoma: How best to do it? World J Gastroenterol 2013; 19(47): 8808-8821
- URL: https://www.wjgnet.com/1007-9327/full/v19/i47/8808.htm
- DOI: https://dx.doi.org/10.3748/wjg.v19.i47.8808
Hepatocellular carcinoma (HCC) is one of the leading malignancies worldwide, representing the fifth most common human cancer and the third cause of death from cancer[1,2]. Patients diagnosed with HCC often have a dismal prognosis as it is diagnosed at late stages, when therapeutic approaches are limited, if applicable at all. Conversely, an early diagnosis of HCC allows the application of curative or effective treatments in most cases, improving the survival of these patients[3]. Therefore there is a need for early diagnosis of this tumor. Screening and surveillance for HCC applied to patients with chronic liver disease who are at risk of developing this cancer can indeed identify malignancies at early stages and improve patient survival, although there is still debate regarding the optimal screening and surveillance tools and the actual yield of surveillance[3-5]. This review addresses the current evidence supporting surveillance programs in patients at risk of developing HCC, the best way to perform surveillance, and the still unsolved nuances of this topic.
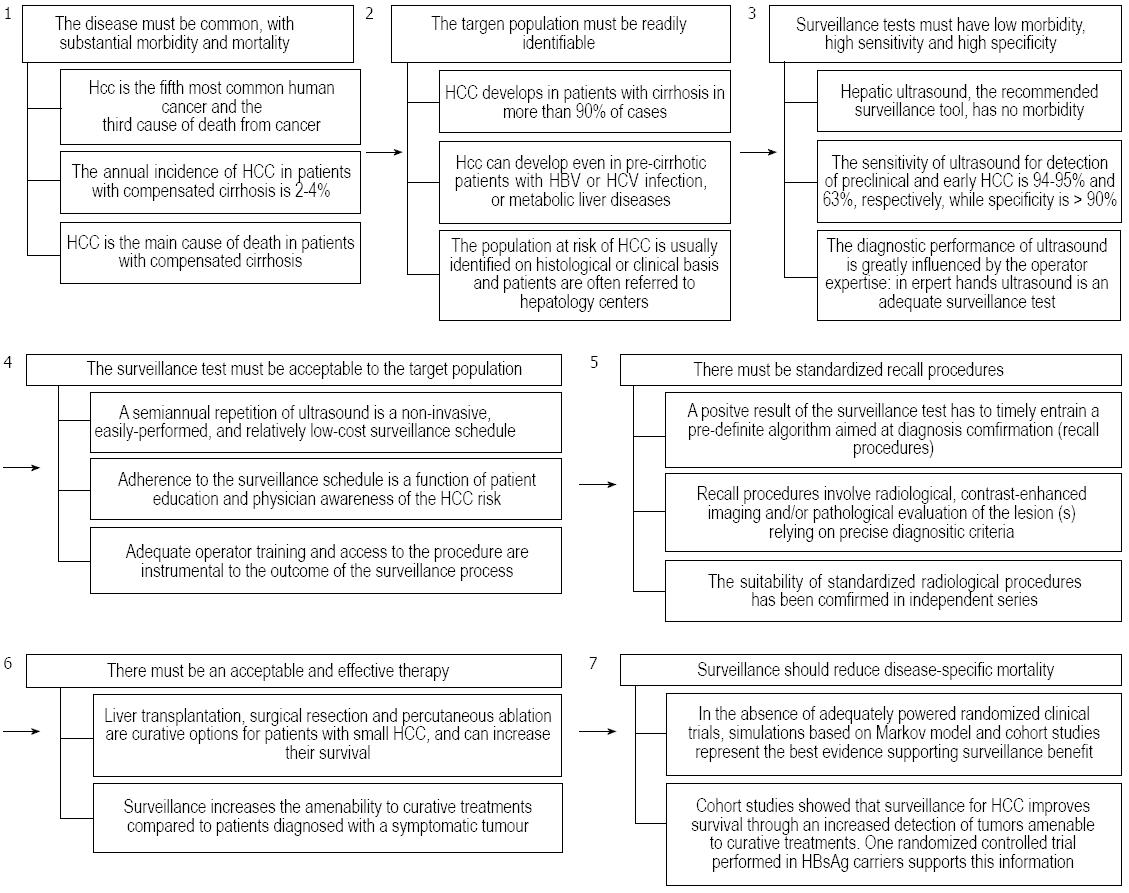
Screening is the application of a test to detect a disease in a population which has no signs or symptoms of that disease, while surveillance is the periodic repetition of the screening test in the same population. Both screening and surveillance have the aim of detecting a disease before it becomes symptomatic, at an early time point of its natural history and when treatment is more effective, with the ultimate goal of reducing disease-specific mortality. Positive findings of screening or surveillance tests must entrain a pre-defined recall policy aimed at identifying true positive cases with additional diagnostic procedures. Screening and surveillance must fulfill the seven Prorok postulates[6] and, as outlined below, this is the case for the surveillance of patients at risk of developing HCC.
HCC is a common malignancy worldwide and its incidence is expected to rise in most Western world areas due to the aging of patients with chronic hepatitis C virus (HCV) infection, which is the main etiological factor of this tumor in developed countries[7,8]. Moreover, HCC is currently the main cause of death of patients with initially compensated liver cirrhosis[9,10]. Noteworthy, the incidence and mortality rates of HCC are very similar all over the world, thus emphasizing the high lethality rate of this tumor in the short term, especially when it is diagnosed at late stages precluding any effective treatment[2], although a favorable mortality trend has recently been observed in Europe[11-13].
More than 90% of HCCs develop in a cirrhotic liver, and the main causes of chronic liver disease in these patients are hepatitis B virus (HBV) or HCV infections, alcohol abuse and non-alcoholic fatty liver disease[14]. These diseases can be detected on the basis of patient history and/or serological tests, thus making the target population for HCC surveillance readily identifiable.
The American and European guidelines for HCC management recommend surveillance to be carried out by ultrasound examination of the liver (US) repeated every 6 mo[15,16]. This surveillance schedule has no morbidity and, when US is properly carried out, a fairly high sensitivity and specificity[17]. In particular, a recent meta-regression analysis has shown that US can identify subclinical HCCs with a sensitivity of 94%-95%, but this drops to 63% for early HCC, while specificity ordinarily exceeded 90%[18]. However, series coming from referral centers reported remarkably higher sensitivity figures (82%), even for early HCC[19,20]. Therefore, the availability of sonographers with expertise in this field is a mandatory prerequisite for a useful US-based surveillance for HCC[21]. The use of serum alpha-fetoprotein (AFP) as a surveillance test has an acceptable specificity but a poor sensitivity for early HCC since only a small fraction (10%-20%) of early cancers is associated with elevated AFP serum levels[15,16,19,22-24]. The combination of US and serum AFP assessment slightly increases (6%) the sensitivity of surveillance but almost doubles the cost for each small HCC detected due to a high number of false positives[18,25].
A semiannual repetition of US is a non-invasive, easily-performed, and relatively low-cost surveillance schedule which is not a major obstacle for patient adherence. Rather, physician education and knowledge of the potential benefits of surveillance, and adequate operator training are areas where there is still room for improving the effectiveness of surveillance programs[26-29].
A positive result of the surveillance test must entrain a prompt activation of a pre-defined standardized algorithm (recall procedures) able to provide a definite diagnosis. Recall procedures for suspected lesions identified by US during screening or surveillance have to be consistently defined and involve radiological, contrast-enhanced imaging procedures or pathological evaluation of the lesion(s) relying on precise diagnostic criteria[15,16]. The diagnostic yield of these recall procedures has been independently confirmed, and allows an adequate evaluation of tumor extension that, in turn, has a pivotal role in driving the therapeutic strategy[21,30,31]. An inappropriate or delayed application of recall procedures is an important cause of surveillance failure[28].
The goal of surveillance for HCC is to identify tiny lesions, amenable to curative treatments with the aim of improving patient survival. Surgical resection, percutaneous ablation, and liver transplantation (LT) are considered curative options for patients with small HCCs. The results of a randomized study carried out in China in HBV infection active carriers and of several cohort studies carried out in Western and Japanese patients with cirrhosis support the use of surveillance as a way of identifying early tumors amenable to curative treatment, and therefore of improving patient survival[19,24,32-34]. Notably, refinements in diagnostic techniques and patient management led to a progressive improvement in the survival of patients diagnosed with HCC during surveillance[35].
The ideal methodology for confirming that surveillance reduces the disease-specific mortality would be to perform a randomized, controlled trial comparing surveillance vs care-on-demand in at-risk patients. Two such studies had been performed in Chinese chronic HBV carriers with contrasting results[32,36]. In particular, despite a 40% reduction in the disease-specific mortality, the first trial was affected by a low degree (< 60%) of patient adherence to the semiannual surveillance program and by the LT unavailability, indicating that the reported figure was probably the “minimal” benefit achievable with surveillance in HBV patients[32]. The negative study was instead methodologically flawed by the fact that patients diagnosed with early HCC did not receive an effective treatment[36]. It is unrealistic to expect results on this topic from new randomized controlled trials, at least in the Western world, due to several reasons: (1) subjects in the control arm would frequently undergo abdominal US due to extrahepatic or liver disease-related reasons; (2) almost all the patients, if adequately informed on the risk-benefits of surveillance, would refuse to participate in the study[37,38]; and (3) this position would likely be shared by most clinicians. Thus, the belief that surveillance for HCC reduces the disease-specific mortality and the pertinent recommendations released by Western and Eastern international guidelines mainly relies on the available proof-of-concept evidence, showing that US surveillance can detect small, asymptomatic tumors that are amenable to curative treatment while symptomatic HCCs are generally detected at an advanced stage, which greatly limits or even precludes any treatment. Pertinently, Western and Eastern cohort studies comparing the outcome of patients with HCC diagnosed during or outside surveillance programs consistently demonstrate that the assumed surveillance benefit holds true[4,24,32-34,38,39].
In the Western world, surveillance is recommended for subjects at high risk of developing HCC such as patients with cirrhosis and certain categories of patients with chronic hepatitis, while Japanese guidelines extend this recommendation to all patients with chronic hepatitis[15,16,40]. An essential pre-requisite to perform surveillance is the absence of contraindications to treatment-either curative or palliative-once HCC is diagnosed. Thus, surveillance is useless in patients with Child-Pugh class C cirrhosis not listed for LT[41], as an early detection of HCC does not improve their survival due to the inapplicability of therapeutic options for malignancy other than LT and a strong competitive effect with cancer by liver failure as the death cause[42].
As mentioned before, surveillance should be cost-effective and one crucial determinant of cost-effectiveness (CE) is the disease incidence in the target population (see also the specific chapter below). Therefore, the selection of patients who should enter into surveillance programs for HCC is driven by their oncologic risk, which can be inferred from the incidence of HCC (Table 1). The incidence threshold that should trigger surveillance in patients with cirrhosis is 1.5% per year, while for patients with chronic hepatitis this drops to 0.2% per year[15]. It is important to note that these thresholds are not derived from experimental data but they were proposed considering the results of CE analyses based on the Markov model showing an increase in survival of > 3 mo at a cost of less than 50000 USD per year of life gained[43,44].
| Group of patients | Threshold incidence to implement surveillance (% per year) | Incidence of HCC |
| Surveillance recommended | ||
| Asian male hepatitis B carriers over age 40 | 0.2 | 0.4%-0.6%/yr |
| Asian female hepatitis B carriers over age 50 | 0.2 | 0.3%-0.6%/yr |
| Hepatitis B carriers with family history of hepatocellular carcinoma | 0.2 | Incidence higher than without family history |
| African/North American Blacks with hepatitis B | 0.2 | Hepatocellular carcinoma occurs at a younger age |
| Hepatitis B virus carriers, cirrhosis | 0.2-1.5 | 3%-8%/yr |
| Hepatitis C virus infection, cirrhosis | 1.5 | 3%-5%/yr |
| Primary Biliary Cirrhosis, stage 4 | 1.5 | 3%-5%/yr |
| Genetic hemochromatosis, cirrhosis | 1.5 | Unknown, but probably > 1.5%/yr |
| Alpha 1-antitrypsin deficiency, cirrhosis | 1.5 | Unknown, but probably > 1.5%/yr |
| Other cirrhosis | 1.5 | Unknown |
| Surveillance benefit uncertain | ||
| Hepatitis B carriers younger than 40 (males) or 50 (females) | 0.2 | < 0.2%/yr |
| Hepatitis C virus infection, stage 3 fibrosis | 1.5 | < 1.5%/yr |
| Non-cirrhotic non-alcoholic fatty liver disease | 1.5 | < 1.5%/yr |
According to the above mentioned thresholds, patients with cirrhosis are appropriate candidates for a cost-effective surveillance, as the annual incidence of HCC in cirrhotic patients with HCV or HBV infection is 1.5%-4.5% and 2.2%-4.3%, respectively, and it is approximately 2.6% in both alcoholic and non-alcoholic steatohepatitis cirrhosis[14,45-47]. Even cirrhotic patients with genetic hemochromatosis or primary biliary cirrhosis have an HCC risk high enough to implement surveillance, whereas the annual incidence of HCC reported in cirrhotic patients with autoimmune hepatitis is 1.1%, thus questioning the CE of surveillance in this category of patients[48-50].
Patients with HCV-related cirrhosis who cleared the infection with antiviral treatment represent a subset of patients with a decreased, but not abolished, risk of HCC[51,52]. Namely, the incidence rate of HCC per 100 person-years in Japanese patients with cirrhosis who achieved a sustained virological response (SVR) to antiviral treatment was 0.5% compared to 5% in patients without SVR and 8% in untreated cirrhotic patients, while a retrospective Italian study showed figures of 0.7% after SVR and 2% in non-responder patients[51,52]. It should be pointed out that HCC incidences observed after SVR do not cross the suggested CE threshold for surveillance in cirrhosis. Nevertheless, non-viremic HCV cirrhotic patients represent a peculiar population where mortality due to the complications of cirrhosis or liver failure is negligible and the chance of applying aggressive treatments for HCC is high[51,52]. The same applies to HBV patients effectively treated with antiviral nucleos(t)ide drugs in whom the risk of HCC remains as high as 1.3 per 100 person-years despite undetectable viremia[53,54].
To conclude, non-viremic HCV and HBV patients should continue (or start) to undergo surveillance if they were at high risk of developing HCC before starting antiviral treatment.
Among pre-cirrhotic patients, those with chronic HBV infection have the highest risk of developing HCC, especially those with long-standing disease, who more likely acquired the infection perinatally, and those with persistent, high-load viral replication[55-57]. These features are frequent in Asian patients, and a study from China showed that patients with chronic hepatitis B without cirrhosis have an annual HCC incidence of 0.8%, thus exceeding the accepted threshold (0.2%) for a cost-effective surveillance[15,58]. African active HBV carriers or with a positive family history are also considered good candidates for surveillance, due to high HCC incidence. As the incidence of HCC in HBV-positive Western patients ranges from 0.1% to 0.4% per year[59,60], the latest European guidelines for HCC management recommend the implementation of surveillance programs in the subgroups of HBV-positive patients with active hepatitis or a family history of HCC[16].
Although Japanese guidelines recommend HCC surveillance in chronic hepatitis C patients whereas American and European guidelines propose this procedure in those with advanced fibrosis, the evidence supporting these suggestions is less robust[15,16,40]. Indeed, on the one hand, a study carried out in Japan showed that the annual incidence of HCC in untreated patients with chronic hepatitis C increased with increasing fibrosis stage, being 0.5% in patients without or with mild fibrosis, and 5% in those with severe fibrosis[52]. On the other hand, a large, prospective study carried out in the United States to assess the incidence of HCC in patients with bridging fibrosis (Ishak stage 3 and 4) reported an incidence of 0.8% per year[61,62]. Importantly, in this cohort the absence of cirrhosis was assessed at enrollment and HCC was diagnosed after a median of 46.5 mo of follow-up, when cirrhosis had developed in 65% of these patients (15/23 patients) and thrombocytopenia was present in all but one patient[62]. These findings emphasize the difficulties in identifying a clear hallmark indicating the transition from a low to a high oncologic risk status. In an attempt to overcome this problem, either bed-side clinical scores or transient hepatic elastography have been proposed to stratify patients according to HCC risk[62-65].
Lastly, although HCC may also occur in non-cirrhotic patients with a non-viral chronic liver disease, exhaustive data on its incidence in these categories are not currently available, but it is unlikely for surveillance to be cost-effective in these settings.
In general, a surveillance test has to have a high sensitivity (to miss very few cancers) and an adequate specificity (to avoid unnecessary confirmatory testing). There is universal agreement that US is the imaging tool to be used for surveillance of HCC. A meta-regression analysis of several cohort studies set the sensitivity of US, as a surveillance test for HCC, at 94% for asymptomatic tumors and 63% for early HCC, with a specificity of > 90%[18]. The relatively low sensitivity of US for tiny lesions may be explained by the fact that this technique is highly dependent on both the operator expertise and the quality of US equipment. In fact, the presence of regenerative nodules and fibrous septa conferring a coarse echo-pattern to the cirrhotic liver makes it difficult to identify minute nodules. Therefore, US examination should be performed by skilled operators and with adequate instruments. In this case, the sensitivity for early-stage or small HCC ranges from 82% to 91%, and the mean size of HCCs detected during surveillance is < 2 cm, with only 1.4% of tumors > 3 cm[19,20,66,67].
AFP is the serum tumoral marker most widely used in the surveillance for HCC[22]. Its levels are influenced by tumor size and aggressiveness, as well as by the etiology and activity of the liver disease[23,68-74]. These limitations affect the usefulness of AFP as a surveillance test for HCC, and its principal drawback is a poor sensitivity at cut-off levels ensuring an adequate specificity[22,75]. Namely, serum AFP levels are increased in a minority of early HCCs and, when elevated, tend to identify highly malignant cancers with a rapid growth rate[23,67-69,71-73]. Furthermore, AFP lacks specificity for HCC since abnormal levels can be caused by hepatitis activity flares in both HBV- and HCV-infected patients[70,74].
The combined use of US and AFP increases the sensitivity for early HCC by 6% compared to US alone, but also enhances the rate of false positive results, with detrimental consequences on direct and indirect costs for each early HCC detected[76-80]. In fact, while false positive results occur with US or AFP alone in 2.9% and 5.0% of cases, respectively, the figure rises to 7.5% on combining the two tests, and this drop of specificity translates into a cost of approximately 2000 USD per HCC identified with US alone as compared to 3000 USD with the combination of AFP plus US[25].
Inadequate sensitivity for early lesions and lack of specificity discourage the use of AFP as a screening and surveillance tool for HCC[23,78], so that the use of US alone in this setting has been recommended by Western guidelines for HCC management[15,16]. This suggestion, however, is not shared by the recently released Eastern guidelines[40,81], that continue to propose the combined use of US and sero-markers, such as AFP and des-gamma-carboxy prothrombin, aimed at maximizing the sensitivity of surveillance regardless of its negative impact on CE.
Patients on the LT waiting list represent a special subgroup where surveillance for HCC acquires additional clinical significance, as the identification of an HCC in these patients: (1) can hasten the urgency for LT by prioritizing the patient on the list; (2) alternatively, it may represent a reason for waiting list drop-out if the tumor burden exceeds the accepted criteria for LT[82-84]; (3) due to these reasons, it also impacts on the probability of the listed non-HCC patients to be transplanted[82,85]. Non-HCC patients listed for LT usually have an advanced cirrhosis which associates with a coarse liver echo-pattern, organ shrinkage and ascites, and these features may impair the US ability to detect (small) focal lesions[86]. Therefore, although there is no compelling evidence to support this suggestion, an HCC surveillance carried out with multiphasic computed tomography (CT) or magnetic resonance (MR) every 6 mo can be proposed for these patients; considering that the expected surveillance duration seldom exceeds 1 year, the detection of HCC is crucial to define the priority for LT (and hence to fairly allocate a limited therapeutic resource among oncologic and non-oncologic candidates), and the use of these techniques has been associated with a better CE ratio[21].
Patients with non-alcoholic liver cirrhosis represent a growing population at risk of HCC[87-89], and in most of them the presence of fatty liver and obesity may impair imaging resolution of liver US exploration. Although no formal studies have been carried out to address this issue, in some studies CT was purposely used instead of US for HCC surveillance in a minority of patients (3.3%) due to the presence of suboptimal US resolution because of a coarse liver echo-pattern or extreme obesity[67]. Instead, in a study carried out in the United States, the presence of an increased body mass index was not associated with a decreased sensitivity of US for HCC detection, although the robustness of this finding is flawed by the limited statistical power of the study and the overall poor quality of US results[90]. Thus, due to the growing prevalence of non-alcoholic liver disease, this is a field where prospective studies comparing US with other surveillance tools are urgently needed[91].
The surveillance interval should be dictated by the expected doubling volume time of the surveyed tumor, and not by the degree of the inherent risk of HCC. Median doubling volume time of untreated HCC is around 170 d, although there is a great inter-individual variability and the growth rate may be not constant over time[67,92]. This would indicate that the reference length of the surveillance interval is 6 mo. Increasing the length to 12 mo is indeed associated with a greater likelihood of missing early HCCs, reducing the applicability of effective treatments and thus worsening survival as compared to the semiannual surveillance schedule[34,93]. In fact, after correction for the lead-time bias, the survival of Child-Pugh class A or B patients with HCC identified during a semiannual surveillance was significantly improved as compared to patients undergoing 12 mo surveillance[34]. Similar findings were obtained in Asian patients, in whom the survival benefit adjusted for lead-time bias was significantly greater when surveillance was carried out with an interval ≤ 6 mo as compared to > 6 mo[93]. Conversely, a randomized study prevalently including patients with alcoholic cirrhosis demonstrated that shortening the surveillance schedule to 3 mo was detrimental as it did not significantly increase the likelihood to detect small (≤ 3 cm) HCCs (79% vs 70%), amenability to curative treatment (62% vs 58%) and 5-year survival (85% vs 86%), whereas it led to a greater cumulative incidence of detected focal lesions that proved non-malignant during the follow-up, thus leading to an increased cost of recall procedures[94]. In this regard, it should be emphasized that the proposal of the Japanese and Asian guidelines to shorten the surveillance interval to 3 mo in patients at very high risk of developing HCC does not rely on experimental results or CE study models[40,81]. Thus, on the basis of the currently available evidence, a 6-mo interval should be recommended for HCC surveillance[15,16].
Efficacy is a measure of the degree to which one procedure obtains the expected result under standardized conditions, generally chosen to maximize the chance to observe the expected result. Effectiveness, instead, measures the extent of the benefit when the procedure is applied in clinical practice. Effectiveness not only depends upon the efficacy of the procedure but also on “external” non-standardized factors, such as physicians’ (specific knowledge, convincement and recommendation) and patients’ (acceptance and adherence) behavior, health system organization (timeliness of the recall policy, availability and accessibility of appropriate diagnostic tools and treatments, adequate follow-up), as well as economic, cultural and social influences. In the case of surveillance for HCC, it can be optimistically hypothesized that its effectiveness is affected by the following drawbacks: missed/unconvincing doctor recommendation (80%), limitations to surveillance access (90%), patient refusal (90%) or inadequate adherence (90%), untimely recall (by 90%), untimely availability of appropriate diagnostic and therapeutic options (90%) and improper follow-up (90%). Thus, assuming that the mentioned limitations are independent probabilities and the reduction in overall mortality of cirrhotic patients with HCC diagnosed during surveillance is 40% - according to the Italian Liver Cancer data (ITA.LI.CA)[34] - it can be calculated (0.40 × 0.80 × 0.90 × 0.90 × 0.90 × 0.90 × 0.90 × 0.90) that the actual effectiveness of surveillance in cirrhosis drops to 17%.
Therefore, surveillance for early diagnosis of HCC is a typical example of “clinical nuance”, whose basics tenets are that medical services and providers differ in the clinical benefit provided; hence, the benefit of the service depends on the person using it, as well as where and by whom the service is provided. As previously pointed out by our group, besides limited economical resources, a major flaw of surveillance for HCC is the “behavior hazard” of both clinicians (prescription and organization) and patients (adherence)[79]. These shortcomings explain the large gap between efficacy and effectiveness of surveillance of patients at risk of HCC, and indicate the road for reducing this gap and greatly improving the CE of the procedure without the need for diagnostic and therapeutic advancements.
The economic aspect of HCC surveillance has also to be considered. Its CE is mainly determined by two features: the gain obtainable with surveillance in terms of quality-adjusted life-expectancy (effectiveness) and its total costs. In turn, these features are determined by two components each. Effectiveness strictly depends on HCC incidence and the actual possibility to submit patients diagnosed with HCC to potentially curative treatments; total costs result from the sum of the costs of surveillance test(s), tools utilized for tumor diagnosis and staging, and HCC treatment(s).
As mentioned above, from a CE standpoint US surveillance of cirrhotic patients should be started when the annual HCC incidence is expected to be at least 1.5%; however, it cannot be excluded that different surveillance strategies, and different surveillance intervals, can be more cost-effective in different clinical scenarios. For instance, available data suggest that the annual program of US surveillance (± AFP assessment) is cost-effective in patients with a tumor risk up to 3%-3.5% per year, while the semiannual program becomes more cost-effective in patients with a risk above these figures[44,76,95,96]. Indeed, the semiannual US strategy has been consistently reported to be the most effective program for an early tumor diagnosis but it inevitably increases direct and indirect costs with respect to programs with longer intervals. Considering this, a reasonable alternative from a CE perspective is the “AFP-triage strategy” that avoids US use in patients with normal AFP values. This strategy has been reported to be more cost-effective than semiannual US but with a lower efficiency in detecting HCC[97].
The second main determinant of surveillance effectiveness is the possibility to timely submit HCC patients to potentially curative treatments. While it is not possible to predict the tumor burden at presentation in the individual patient, it is intuitive that an advanced degree of liver dysfunction strongly limits-or even prevents-the therapeutic approach to the forthcoming HCC. The literature lacks specific analyses comparing the CE of surveillance vs no-surveillance in decompensated cirrhotic patients, also because surveillance is not currently recommended in patients with advanced cirrhosis not listed for LT. The only available evidence indicates that semiannual US surveillance can be more cost-effective than annual surveillance only if treatment can ensure a huge survival gain after HCC diagnosis, as in the case of LT[97], indirectly supporting the recommendation to keep, among Child-Pugh C patients, only candidates for LT under surveillance.
The direct costs of surveillance test(s) are relatively low, as both US examination and AFP dosage are not high-cost procedures. It has been reported that costs for surveillance and tumor diagnosis are around 18000 USD per each potentially curable HCC detected, accounting for only 10%-20% of total costs of cancer management since the main determinant of costs is treatment[19,98,99]. Nevertheless, the CE of surveillance programs based on CT, MR or contrast-enhanced US (CEUS) has been tested with Markov model analyses and most of the studies found that their use raised costs, without a parallel significant increase in HCC detection, resulting in a higher incremental cost-effectiveness ratio (ICER) compared to US surveillance[76,82,100,101]. Thus, there is not sufficient evidence for adopting CT or MR as surveillance tests, whereas the use of CEUS, although intriguing, requires further dedicated studies.
Another point that needs to be addressed is the use of AFP as a surveillance test. Sensitivity of AFP is reported to be around 60%, and its specificity is limited by the non-HCC related elevation of the marker due to hepatic necro-inflammation and regeneration occurring in active hepatitis or cirrhosis[15,16,70,71,74]. Consequently, the frequent false positive results of a periodic AFP measurement, entraining confirmatory tests, increase the total costs of surveillance based on serum AFP measurement[96].
As mentioned before, the main determinant of surveillance costs derives from the tumor treatment. For example, the inclusion of LT in the treatment algorithm, reimbursement of which can be up to 250000 USD (University of Alabama)[100], results in an up to 10-fold increase of the average cost-effectiveness when compared to scenarios where LT is not an option. Hepatic resection is another high-cost intervention, that can compete with percutaneous ablation in terms of both survival and CE. Available literature suggests a CE advantage for ablation in the case of single tumors ≤ 2 cm and 2-3 nodules each ≤ 3 cm, while surgery becomes more cost-effective for single tumors > 3 cm[102]. Thus, the type of treatment adopted, the proportion of patients undergoing each therapy and, more importantly, costs assumptions are the main sources of uncertainty for simulation models aimed at calculating the CE of surveillance for HCC. Therefore, it is advisable to propose prospective micro-costing analyses to refine this topic. Micro-costing studies collect data and values on the resources utilized for each patient so that, although time- and resource-consuming (expensive record keeping over time and use of database management), they allow a precise definition of costs. Only one prospective micro-costing study has been published, and this was more than 10 years ago[19]. Due to the changed scenario of HCC management since then, further similar studies are warranted.
To conclude, semiannual surveillance based on US achieves a higher detection rate of early HCC but at increased costs with respect to the annual program. From a CE perspective, alternative strategies, such as the semiannual AFP + annual US or the annual US (± AFP) schedules, could be proposed and tested in patients with a relatively low HCC incidence, such as young cirrhotic women or patients who have become non-viremic after (HCV- and HBV-infected) or during (HBV-infected) antiviral treatment.
Recall policy is instrumental to the success of surveillance, since an abnormal surveillance test must promptly entrain a pre-defined strategy aimed at ruling in/out the presence of HCC and staging it. The diagnostic algorithm that composes recall procedures should be carried out within a reasonable time interval to allow timely and adequate treatment. Recall procedures greatly concur in diagnosing HCC at a very early (solitary, ≤ 2 cm) or early stage (meeting the Milan criteria) that, in turn, allows application of curative treatment and eventually improves patient survival. It is recommended that patients are evaluated at a referral center with availability of all diagnostic techniques and therapeutic opportunities.
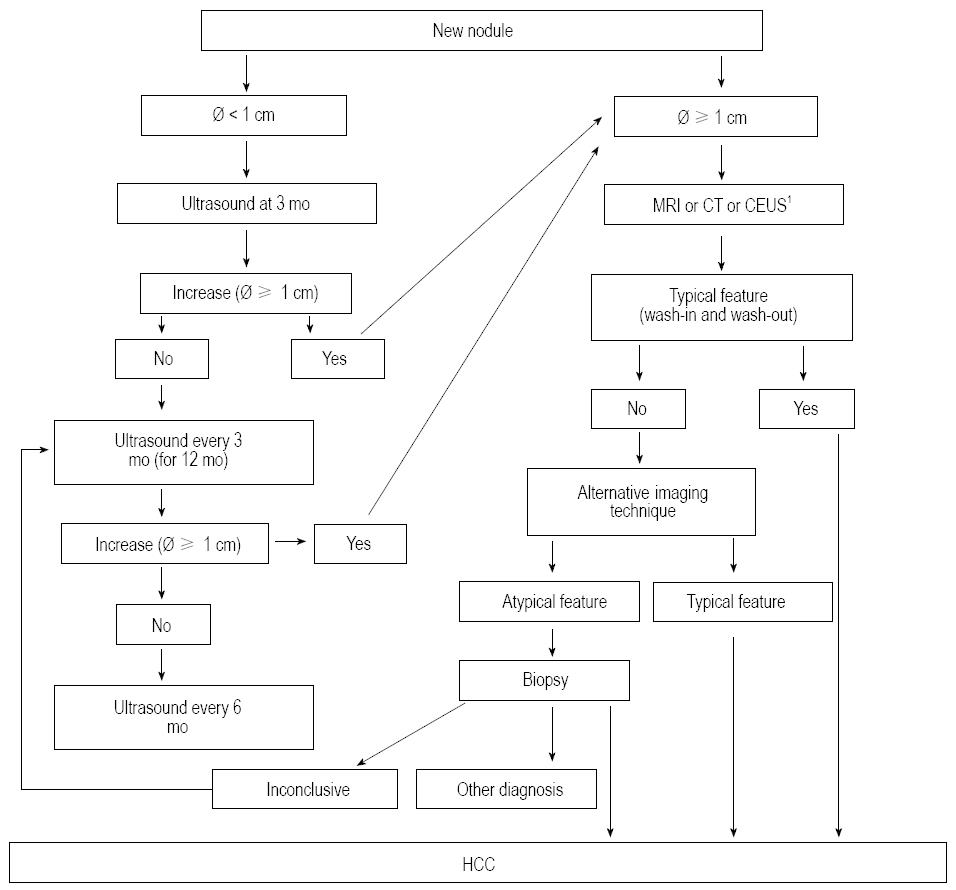
Importantly, any new lesion identified at screening or during surveillance as well as pre-existing lesions enlarging or changing their echo-pattern should be regarded as malignant unless otherwise demonstrated; however, as most nodules < 1 cm are non-malignant, the institution of recall procedures for these lesions would increase surveillance costs without clinical gain[94,103]. Therefore, these lesions should be strictly followed-up with US every 3 mo until an increase in size occurs (allowing a suitable definition of their nature with diagnostic techniques) or for one-two years[15,16,104]. This shortening of the interval between US scans (“enhanced” follow-up) is dictated by the knowledge that the volume doubling time of some HCCs may be as short as 30 d, and the main goal of surveillance is to detect HCCs ≤ 2 cm[89]. It has to be emphasized that the echo-pattern is not predictive of malignancy since, although HCC more often presents as a hypo-echoic lesion, it may be hyper-echoic or have a “target” appearance[105,106].
The recall strategy for lesions ≥ 1 cm relies on the use of dynamic, contrast-enhanced, multiphase, imaging techniques with vascular contrast media (CT, MR, CEUS) and overlaps with the diagnostic process. In cirrhotic patients, if the nodule shows the typical vascular patter i.e., homogeneous contrast enhancement in the arterial phase (wash-in) followed by hypo-enhancement in the portal or venous phase (wash-out) - it can be regarded as HCC with no need for histological confirmation[20,21,30,31,107,108]. If the lesion does not display this typical pattern at the first imaging procedure, an alternative imaging technique can be performed, and if an atypical vascular pattern is found again, the lesion should undergo biopsy. It is recommended that histological samples are evaluated by an expert in liver pathology and, in the case of non-diagnostic pathological results, a follow-up with US every 3 mo should be implemented and the recall procedures repeated as soon as a nodule enlargement is observed (Figure 1).
When selecting the most rewarding imaging technique to be firstly performed in a patient with suspected HCC, it should be considered that MR has the highest sensitivity to detect the typical vascular pattern in tiny HCC (< 2 cm) and, using hepatocyte-specific contrast agents, it can provide important additional information in the so called “hepato-biliary phase” (hypo-intensity of the nodule) to suspect malignancy even in the absence of the wash-in phenomenon, a feature quite frequent in tiny lesions[21,109-112]. Discovering the malignant nature of nodules < 2 cm is indeed of paramount importance as, above this size, the prevalence of unfavorable prognostic factors, such as microscopic vascular invasion and satellites, greatly increases[59,112] (Figure 2).
The inclusion of CEUS among the imaging techniques of the recall policy is currently debated, due to the risk of misdiagnosis between HCC and intrahepatic cholangiocarcinoma (ICC)[15,16,113-115]. However, the recently released Italian recommendations for HCC management[104] have included CEUS in the recall algorithm due to: (1) its positive predictive value for HCC > 95% when a typical vascular pattern is observed; (2) the fairly low incidence of ICC in cirrhosis (1%-3% of newly diagnosed tumors); and (3) the fact that only half of small ICCs display a pattern typical for HCC at CEUS[115-118]. From a CE standpoint, however, it should be pointed out that, since a “panoramic” imaging technique is mandatory to correctly stage the tumor, CT or MR should be preferred, using CEUS as a second-line procedure in the case of inconclusive findings at radiological imaging techniques[105,114].
Despite the available evidence that surveillance increases the survival of patients diagnosed with HCC, expanding the possibility to perform effective therapies, there is still controversy on its actual usefulness in clinical practice. Some recent studies, coming from the United States, have helped frame the receipt of HCC surveillance in everyday practice in this country and reported the obstacles to its utilization, providing hints on how to improve its uptake and outcome[116-121]. Indeed, initial reports showed that no more than 28% of patients diagnosed with HCC underwent at least 1 screening test in the 3 years preceding the diagnosis and, among them, 36% received AFP testing alone as a screening test[117]. However, this study did not report a measure of receipt of surveillance in the whole population of patients at risk. A subsequent study, performed on a larger and more representative sample, confirmed a low uptake of surveillance in patients diagnosed with HCC, showing that 17% and 38% of patients received consistent and inconsistent surveillance, respectively, before HCC detection, and demonstrated that being followed up by a gastroenterologist/hepatologist or an academic physician was associated with a higher likelihood of receiving surveillance as compared to patients followed by primary care physicians[118]. Thus, being followed by a specialist in liver disease is a key factor for the likelihood of receiving HCC screening and surveillance, a finding indirectly supported by the result of a self-reported use of surveillance ranging from 71% to 84% among members of the American Association for the Study of the Liver and the Veteran Health Administration[121,122]. Moreover, an adequate surveillance was strongly associated with the local availability of all possible treatments for HCC[121], thus emphasizing the concept that patients at risk should be followed up and managed at referral centers with availability of multi-disciplinary resources to optimize the effectiveness of surveillance. These findings underscore that the patient’s probability to be maintained under surveillance is strictly connected with specialist care and the possibility to receive treatment for HCC, and that effectiveness of surveillance is modest in decreasing HCC mortality when surveillance uptake is markedly low[119,121].
Lastly, longitudinal evaluation of the ITA.LI.CA database over 20 years showed an increase in the proportion of patients diagnosed with HCC during surveillance until 2002, followed by stationary figures over the subsequent 6 years, accounting for approximately 53% of these cases, but with a significant continuous shift to preference of the 6-mo interval[89]. These data, as well as those coming from the United States, clearly reveal an insufficient and suboptimal use of surveillance in the real world of health care and should stimulate educational policies aimed at expanding the knowledge and the correct use of this tool for secondary prevention of HCC. Indeed, audits with identification of barriers to the application of surveillance and implementation of measures able to improve physician and patient education, together with system re-design, have led to a great increase in the application of adequate surveillance protocols for an early diagnosis of HCC[122].
P- Reviewers: Goglia F, Marin JJG, Mazzanti R, Rajeshwari K S- Editor: Qi Y L- Editor: A E- Editor: Liu XM
| 1. | Parkin DM. Global cancer statistics in the year 2000. Lancet Oncol. 2001;2:533-543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1727] [Cited by in RCA: 1710] [Article Influence: 71.3] [Reference Citation Analysis (0)] |
| 2. | Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer. 2010;127:2893-2917. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11128] [Cited by in RCA: 11836] [Article Influence: 845.4] [Reference Citation Analysis (4)] |
| 3. | de Lope CR, Tremosini S, Forner A, Reig M, Bruix J. Management of HCC. J Hepatol. 2012;56 Suppl 1:S75-S87. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 409] [Cited by in RCA: 484] [Article Influence: 37.2] [Reference Citation Analysis (0)] |
| 4. | Noda I, Kitamoto M, Nakahara H, Hayashi R, Okimoto T, Monzen Y, Yamada H, Imagawa M, Hiraga N, Tanaka J. Regular surveillance by imaging for early detection and better prognosis of hepatocellular carcinoma in patients infected with hepatitis C virus. J Gastroenterol. 2010;45:105-112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 5. | Lederle FA, Pocha C. Screening for liver cancer: the rush to judgment. Ann Intern Med. 2012;156:387-389. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 74] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 6. | Prorok PC. Epidemiologic approach for cancer screening. Problems in design and analysis of trials. Am J Pediatr Hematol Oncol. 1992;14:117-128. [PubMed] |
| 7. | El-Serag HB. Hepatocellular carcinoma: recent trends in the United States. Gastroenterology. 2004;127:S27-S34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 738] [Cited by in RCA: 723] [Article Influence: 34.4] [Reference Citation Analysis (0)] |
| 8. | Kanwal F, Hoang T, Kramer JR, Asch SM, Goetz MB, Zeringue A, Richardson P, El-Serag HB. Increasing prevalence of HCC and cirrhosis in patients with chronic hepatitis C virus infection. Gastroenterology. 2011;140:1182-1188.e1. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 301] [Cited by in RCA: 308] [Article Influence: 22.0] [Reference Citation Analysis (0)] |
| 9. | Benvegnù L, Gios M, Boccato S, Alberti A. Natural history of compensated viral cirrhosis: a prospective study on the incidence and hierarchy of major complications. Gut. 2004;53:744-749. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 328] [Cited by in RCA: 318] [Article Influence: 15.1] [Reference Citation Analysis (0)] |
| 10. | Talwalkar JA, Kamath PS. Influence of recent advances in medical management on clinical outcomes of cirrhosis. Mayo Clin Proc. 2005;80:1501-1508. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 38] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 11. | Bosetti C, Levi F, Boffetta P, Lucchini F, Negri E, La Vecchia C. Trends in mortality from hepatocellular carcinoma in Europe, 1980-2004. Hepatology. 2008;48:137-145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 194] [Cited by in RCA: 181] [Article Influence: 10.6] [Reference Citation Analysis (1)] |
| 12. | Bosetti C, Bianchi C, Negri E, Colombo M, La Vecchia C. Estimates of the incidence and prevalence of hepatocellular carcinoma in Italy in 2002 and projections for the years 2007 and 2012. Tumori. 2009;95:23-27. [PubMed] |
| 13. | Bertuccio P, Bosetti C, Levi F, Decarli A, Negri E, La Vecchia C. A comparison of trends in mortality from primary liver cancer and intrahepatic cholangiocarcinoma in Europe. Ann Oncol. 2013;24:1667-1674. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 113] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 14. | Fattovich G, Stroffolini T, Zagni I, Donato F. Hepatocellular carcinoma in cirrhosis: incidence and risk factors. Gastroenterology. 2004;127:S35-S50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1691] [Cited by in RCA: 1793] [Article Influence: 85.4] [Reference Citation Analysis (2)] |
| 15. | Bruix J, Sherman M. Management of hepatocellular carcinoma: an update. Hepatology. 2011;53:1020-1022. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5972] [Cited by in RCA: 6573] [Article Influence: 469.5] [Reference Citation Analysis (1)] |
| 16. | European Association For The Study Of The Liver; European Organisation For Research And Treatment Of Cancer. EASL-EORTC clinical practice guidelines: management of hepatocellular carcinoma. J Hepatol. 2012;56:908-943. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4059] [Cited by in RCA: 4520] [Article Influence: 347.7] [Reference Citation Analysis (2)] |
| 17. | Bolondi L. Screening for hepatocellular carcinoma in cirrhosis. J Hepatol. 2003;39:1076-1084. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 134] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 18. | Singal A, Volk ML, Waljee A, Salgia R, Higgins P, Rogers MA, Marrero JA. Meta-analysis: surveillance with ultrasound for early-stage hepatocellular carcinoma in patients with cirrhosis. Aliment Pharmacol Ther. 2009;30:37-47. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 519] [Cited by in RCA: 573] [Article Influence: 35.8] [Reference Citation Analysis (0)] |
| 19. | Bolondi L, Sofia S, Siringo S, Gaiani S, Casali A, Zironi G, Piscaglia F, Gramantieri L, Zanetti M, Sherman M. Surveillance programme of cirrhotic patients for early diagnosis and treatment of hepatocellular carcinoma: a cost effectiveness analysis. Gut. 2001;48:251-259. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 473] [Cited by in RCA: 457] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 20. | Sangiovanni A, Manini MA, Iavarone M, Romeo R, Forzenigo LV, Fraquelli M, Massironi S, Della Corte C, Ronchi G, Rumi MG. The diagnostic and economic impact of contrast imaging techniques in the diagnosis of small hepatocellular carcinoma in cirrhosis. Gut. 2010;59:638-644. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 330] [Cited by in RCA: 309] [Article Influence: 20.6] [Reference Citation Analysis (0)] |
| 21. | Lee JM, Trevisani F, Vilgrain V, Wald C. Imaging diagnosis and staging of hepatocellular carcinoma. Liver Transpl. 2011;17 Suppl 2:S34-S43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 35] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 22. | Sherman M. Current status of alpha-fetoprotein testing. Gastroenterol Hepatol (N Y). 2011;7:113-114. [PubMed] |
| 23. | Giannini EG, Marenco S, Borgonovo G, Savarino V, Farinati F, Del Poggio P, Rapaccini GL, Anna Di Nolfo M, Benvegnù L, Zoli M. Alpha-fetoprotein has no prognostic role in small hepatocellular carcinoma identified during surveillance in compensated cirrhosis. Hepatology. 2012;56:1371-1379. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 115] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 24. | Trevisani F, De Notariis S, Rapaccini G, Farinati F, Benvegnù L, Zoli M, Grazi GL, Del PP, Di N, Bernardi M. Semiannual and annual surveillance of cirrhotic patients for hepatocellular carcinoma: effects on cancer stage and patient survival (Italian experience). Am J Gastroenterol. 2002;97:734-744. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 230] [Cited by in RCA: 234] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 25. | Zhang B, Yang B. Combined alpha fetoprotein testing and ultrasonography as a screening test for primary liver cancer. J Med Screen. 1999;6:108-110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 106] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 26. | Davila JA, Morgan RO, Richardson PA, Du XL, McGlynn KA, El-Serag HB. Use of surveillance for hepatocellular carcinoma among patients with cirrhosis in the United States. Hepatology. 2010;52:132-141. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 313] [Cited by in RCA: 312] [Article Influence: 20.8] [Reference Citation Analysis (0)] |
| 27. | El-Serag HB, Kramer JR, Chen GJ, Duan Z, Richardson PA, Davila JA. Effectiveness of AFP and ultrasound tests on hepatocellular carcinoma mortality in HCV-infected patients in the USA. Gut. 2011;60:992-997. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 77] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 28. | Singal AG, Nehra M, Adams-Huet B, Yopp AC, Tiro JA, Marrero JA, Lok AS, Lee WM. Detection of hepatocellular carcinoma at advanced stages among patients in the HALT-C trial: where did surveillance fail? Am J Gastroenterol. 2013;108:425-432. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 165] [Cited by in RCA: 185] [Article Influence: 15.4] [Reference Citation Analysis (0)] |
| 29. | Giannini EG, Trevisani F. Surveillance for hepatocellular carcinoma: just do it! Am J Gastroenterol. 2013;108:1013-1014. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 30. | Forner A, Vilana R, Ayuso C, Bianchi L, Solé M, Ayuso JR, Boix L, Sala M, Varela M, Llovet JM. Diagnosis of hepatic nodules 20 mm or smaller in cirrhosis: Prospective validation of the noninvasive diagnostic criteria for hepatocellular carcinoma. Hepatology. 2008;47:97-104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 724] [Cited by in RCA: 727] [Article Influence: 42.8] [Reference Citation Analysis (1)] |
| 31. | Leoni S, Piscaglia F, Golfieri R, Camaggi V, Vidili G, Pini P, Bolondi L. The impact of vascular and nonvascular findings on the noninvasive diagnosis of small hepatocellular carcinoma based on the EASL and AASLD criteria. Am J Gastroenterol. 2010;105:599-609. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 150] [Cited by in RCA: 151] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 32. | Zhang BH, Yang BH, Tang ZY. Randomized controlled trial of screening for hepatocellular carcinoma. J Cancer Res Clin Oncol. 2004;130:417-422. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 960] [Cited by in RCA: 944] [Article Influence: 45.0] [Reference Citation Analysis (1)] |
| 33. | Stravitz RT, Heuman DM, Chand N, Sterling RK, Shiffman ML, Luketic VA, Sanyal AJ, Habib A, Mihas AA, Giles HC. Surveillance for hepatocellular carcinoma in patients with cirrhosis improves outcome. Am J Med. 2008;121:119-126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 115] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 34. | Santi V, Trevisani F, Gramenzi A, Grignaschi A, Mirici-Cappa F, Del Poggio P, Di Nolfo MA, Benvegnù L, Farinati F, Zoli M. Semiannual surveillance is superior to annual surveillance for the detection of early hepatocellular carcinoma and patient survival. J Hepatol. 2010;53:291-297. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 210] [Cited by in RCA: 237] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 35. | Sangiovanni A, Del Ninno E, Fasani P, De Fazio C, Ronchi G, Romeo R, Morabito A, De Franchis R, Colombo M. Increased survival of cirrhotic patients with a hepatocellular carcinoma detected during surveillance. Gastroenterology. 2004;126:1005-1014. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 437] [Cited by in RCA: 429] [Article Influence: 20.4] [Reference Citation Analysis (0)] |
| 36. | Chen JG, Parkin DM, Chen QG, Lu JH, Shen QJ, Zhang BC, Zhu YR. Screening for liver cancer: results of a randomised controlled trial in Qidong, China. J Med Screen. 2003;10:204-209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 209] [Cited by in RCA: 222] [Article Influence: 24.7] [Reference Citation Analysis (0)] |
| 37. | Poustchi H, Farrell GC, Strasser SI, Lee AU, McCaughan GW, George J. Feasibility of conducting a randomized control trial for liver cancer screening: is a randomized controlled trial for liver cancer screening feasible or still needed? Hepatology. 2011;54:1998-2004. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 118] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 38. | Sherman M, Bruix J, Porayko M, Tran T. Screening for hepatocellular carcinoma: the rationale for the American Association for the Study of Liver Diseases recommendations. Hepatology. 2012;56:793-796. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 114] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 39. | Kuo YH, Lu SN, Chen CL, Cheng YF, Lin CY, Hung CH, Chen CH, Changchien CS, Hsu HC, Hu TH. Hepatocellular carcinoma surveillance and appropriate treatment options improve survival for patients with liver cirrhosis. Eur J Cancer. 2010;46:744-751. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 78] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 40. | Kudo M, Izumi N, Kokudo N, Matsui O, Sakamoto M, Nakashima O, Kojiro M, Makuuchi M. Management of hepatocellular carcinoma in Japan: Consensus-Based Clinical Practice Guidelines proposed by the Japan Society of Hepatology (JSH) 2010 updated version. Dig Dis. 2011;29:339-364. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 549] [Cited by in RCA: 664] [Article Influence: 47.4] [Reference Citation Analysis (0)] |
| 41. | Pugh RN, Murray-Lyon IM, Dawson JL, Pietroni MC, Williams R. Transection of the oesophagus for bleeding oesophageal varices. Br J Surg. 1973;60:646-649. [PubMed] |
| 42. | Trevisani F, Santi V, Gramenzi A, Di Nolfo MA, Del Poggio P, Benvegnù L, Rapaccini G, Farinati F, Zoli M, Borzio F. Surveillance for early diagnosis of hepatocellular carcinoma: is it effective in intermediate/advanced cirrhosis? Am J Gastroenterol. 2007;102:2448-2457; quiz 2458. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 80] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 43. | Laupacis A, Feeny D, Detsky AS, Tugwell PX. How attractive does a new technology have to be to warrant adoption and utilization? Tentative guidelines for using clinical and economic evaluations. CMAJ. 1992;146:473-481. [PubMed] |
| 44. | Sarasin FP, Giostra E, Hadengue A. Cost-effectiveness of screening for detection of small hepatocellular carcinoma in western patients with Child-Pugh class A cirrhosis. Am J Med. 1996;101:422-434. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 268] [Cited by in RCA: 264] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 45. | Gomez EV, Rodriguez YS, Bertot LC, Gonzalez AT, Perez YM, Soler EA, Garcia AY, Blanco LP. The natural history of compensated HCV-related cirrhosis: a prospective long-term study. J Hepatol. 2013;58:434-444. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 59] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 46. | Ascha MS, Hanouneh IA, Lopez R, Tamimi TA, Feldstein AF, Zein NN. The incidence and risk factors of hepatocellular carcinoma in patients with nonalcoholic steatohepatitis. Hepatology. 2010;51:1972-1978. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 893] [Cited by in RCA: 965] [Article Influence: 64.3] [Reference Citation Analysis (1)] |
| 47. | Mancebo A, González-Diéguez ML, Cadahía V, Varela M, Pérez R, Navascués CA, Sotorríos NG, Martínez M, Rodrigo L, Rodríguez M. Annual incidence of hepatocellular carcinoma among patients with alcoholic cirrhosis and identification of risk groups. Clin Gastroenterol Hepatol. 2013;11:95-101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 115] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 48. | Caballería L, Parés A, Castells A, Ginés A, Bru C, Rodés J. Hepatocellular carcinoma in primary biliary cirrhosis: similar incidence to that in hepatitis C virus-related cirrhosis. Am J Gastroenterol. 2001;96:1160-1163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 26] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 49. | Fracanzani AL, Conte D, Fraquelli M, Taioli E, Mattioli M, Losco A, Fargion S. Increased cancer risk in a cohort of 230 patients with hereditary hemochromatosis in comparison to matched control patients with non-iron-related chronic liver disease. Hepatology. 2001;33:647-651. [PubMed] |
| 50. | Yeoman AD, Al-Chalabi T, Karani JB, Quaglia A, Devlin J, Mieli-Vergani G, Bomford A, O’Grady JG, Harrison PM, Heneghan MA. Evaluation of risk factors in the development of hepatoscellular carcinoma in autoimmune hepatitis: Implications for follow-up and screening. Hepatology. 2008;48:863-870. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 118] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 51. | Bruno S, Stroffolini T, Colombo M, Bollani S, Benvegnù L, Mazzella G, Ascione A, Santantonio T, Piccinino F, Andreone P. Sustained virological response to interferon-alpha is associated with improved outcome in HCV-related cirrhosis: a retrospective study. Hepatology. 2007;45:579-587. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 506] [Cited by in RCA: 475] [Article Influence: 26.4] [Reference Citation Analysis (0)] |
| 52. | Yoshida H, Shiratori Y, Moriyama M, Arakawa Y, Ide T, Sata M, Inoue O, Yano M, Tanaka M, Fujiyama S. Interferon therapy reduces the risk for hepatocellular carcinoma: national surveillance program of cirrhotic and noncirrhotic patients with chronic hepatitis C in Japan. IHIT Study Group. Inhibition of Hepatocarcinogenesis by Interferon Therapy. Ann Intern Med. 1999;131:174-181. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 833] [Cited by in RCA: 780] [Article Influence: 30.0] [Reference Citation Analysis (0)] |
| 53. | Liaw YF. Impact of therapy on the outcome of chronic hepatitis B. Liver Int. 2013;33 Suppl 1:111-115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 57] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 54. | Singal AK, Salameh H, Kuo YF, Fontana RJ. Meta-analysis: the impact of oral anti-viral agents on the incidence of hepatocellular carcinoma in chronic hepatitis B. Aliment Pharmacol Ther. 2013;38:98-106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 127] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 55. | Chen CJ, Yang HI, Su J, Jen CL, You SL, Lu SN, Huang GT, Iloeje UH. Risk of hepatocellular carcinoma across a biological gradient of serum hepatitis B virus DNA level. JAMA. 2006;295:65-73. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2309] [Cited by in RCA: 2365] [Article Influence: 124.5] [Reference Citation Analysis (0)] |
| 56. | Chen JD, Yang HI, Iloeje UH, You SL, Lu SN, Wang LY, Su J, Sun CA, Liaw YF, Chen CJ. Carriers of inactive hepatitis B virus are still at risk for hepatocellular carcinoma and liver-related death. Gastroenterology. 2010;138:1747-1754. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 236] [Cited by in RCA: 254] [Article Influence: 16.9] [Reference Citation Analysis (0)] |
| 57. | Yang HI, Lu SN, Liaw YF, You SL, Sun CA, Wang LY, Hsiao CK, Chen PJ, Chen DS, Chen CJ. Hepatitis B e antigen and the risk of hepatocellular carcinoma. N Engl J Med. 2002;347:168-174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 924] [Cited by in RCA: 915] [Article Influence: 39.8] [Reference Citation Analysis (0)] |
| 58. | Liaw YF, Tai DI, Chu CM, Lin DY, Sheen IS, Chen TJ, Pao CC. Early detection of hepatocellular carcinoma in patients with chronic type B hepatitis. A prospective study. Gastroenterology. 1986;90:263-267. [PubMed] |
| 59. | Fattovich G, Bortolotti F, Donato F. Natural history of chronic hepatitis B: special emphasis on disease progression and prognostic factors. J Hepatol. 2008;48:335-352. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 909] [Cited by in RCA: 967] [Article Influence: 56.9] [Reference Citation Analysis (0)] |
| 60. | Sánchez-Tapias JM, Costa J, Mas A, Bruguera M, Rodés J. Influence of hepatitis B virus genotype on the long-term outcome of chronic hepatitis B in western patients. Gastroenterology. 2002;123:1848-1856. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 296] [Cited by in RCA: 312] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 61. | Ishak K, Baptista A, Bianchi L, Callea F, De Groote J, Gudat F, Denk H, Desmet V, Korb G, MacSween RN. Histological grading and staging of chronic hepatitis. J Hepatol. 1995;22:696-699. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3521] [Cited by in RCA: 3783] [Article Influence: 126.1] [Reference Citation Analysis (1)] |
| 62. | Lok AS, Seeff LB, Morgan TR, di Bisceglie AM, Sterling RK, Curto TM, Everson GT, Lindsay KL, Lee WM, Bonkovsky HL. Incidence of hepatocellular carcinoma and associated risk factors in hepatitis C-related advanced liver disease. Gastroenterology. 2009;136:138-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 453] [Cited by in RCA: 452] [Article Influence: 28.3] [Reference Citation Analysis (0)] |
| 63. | Masuzaki R, Tateishi R, Yoshida H, Goto E, Sato T, Ohki T, Imamura J, Goto T, Kanai F, Kato N. Prospective risk assessment for hepatocellular carcinoma development in patients with chronic hepatitis C by transient elastography. Hepatology. 2009;49:1954-1961. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 294] [Cited by in RCA: 315] [Article Influence: 19.7] [Reference Citation Analysis (0)] |
| 64. | Ikeda K, Arase Y, Saitoh S, Kobayashi M, Someya T, Hosaka T, Akuta N, Suzuki Y, Suzuki F, Sezaki H. Prediction model of hepatocarcinogenesis for patients with hepatitis C virus-related cirrhosis. Validation with internal and external cohorts. J Hepatol. 2006;44:1089-1097. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 36] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 65. | Kurosaki M, Hiramatsu N, Sakamoto M, Suzuki Y, Iwasaki M, Tamori A, Matsuura K, Kakinuma S, Sugauchi F, Sakamoto N. Data mining model using simple and readily available factors could identify patients at high risk for hepatocellular carcinoma in chronic hepatitis C. J Hepatol. 2012;56:602-608. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 22] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 66. | Zoli M, Magalotti D, Bianchi G, Gueli C, Marchesini G, Pisi E. Efficacy of a surveillance program for early detection of hepatocellular carcinoma. Cancer. 1996;78:977-985. [PubMed] |
| 67. | Sato T, Tateishi R, Yoshida H, Ohki T, Masuzaki R, Imamura J, Goto T, Kanai F, Obi S, Kato N. Ultrasound surveillance for early detection of hepatocellular carcinoma among patients with chronic hepatitis C. Hepatol Int. 2009;3:544-550. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 55] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 68. | Bayati N, Silverman AL, Gordon SC. Serum alpha-fetoprotein levels and liver histology in patients with chronic hepatitis C. Am J Gastroenterol. 1998;93:2452-2456. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 88] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 69. | Farinati F, Marino D, De Giorgio M, Baldan A, Cantarini M, Cursaro C, Rapaccini G, Del Poggio P, Di Nolfo MA, Benvegnù L. Diagnostic and prognostic role of alpha-fetoprotein in hepatocellular carcinoma: both or neither? Am J Gastroenterol. 2006;101:524-532. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 259] [Cited by in RCA: 312] [Article Influence: 16.4] [Reference Citation Analysis (0)] |
| 70. | Chen TM, Huang PT, Tsai MH, Lin LF, Liu CC, Ho KS, Siauw CP, Chao PL, Tung JN. Predictors of alpha-fetoprotein elevation in patients with chronic hepatitis C, but not hepatocellular carcinoma, and its normalization after pegylated interferon alfa 2a-ribavirin combination therapy. J Gastroenterol Hepatol. 2007;22:669-675. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 72] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 71. | Di Bisceglie AM, Sterling RK, Chung RT, Everhart JE, Dienstag JL, Bonkovsky HL, Wright EC, Everson GT, Lindsay KL, Lok AS. Serum alpha-fetoprotein levels in patients with advanced hepatitis C: results from the HALT-C Trial. J Hepatol. 2005;43:434-441. [PubMed] |
| 72. | Nomura F, Ohnishi K, Tanabe Y. Clinical features and prognosis of hepatocellular carcinoma with reference to serum alpha-fetoprotein levels. Analysis of 606 patients. Cancer. 1989;64:1700-1707. [PubMed] |
| 73. | Peng SY, Chen WJ, Lai PL, Jeng YM, Sheu JC, Hsu HC. High alpha-fetoprotein level correlates with high stage, early recurrence and poor prognosis of hepatocellular carcinoma: significance of hepatitis virus infection, age, p53 and beta-catenin mutations. Int J Cancer. 2004;112:44-50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 213] [Cited by in RCA: 253] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 74. | Di Bisceglie AM, Hoofnagle JH. Elevations in serum alpha-fetoprotein levels in patients with chronic hepatitis B. Cancer. 1989;64:2117-2120. [PubMed] |
| 75. | Trevisani F, D’Intino PE, Morselli-Labate AM, Mazzella G, Accogli E, Caraceni P, Domenicali M, De Notariis S, Roda E, Bernardi M. Serum alpha-fetoprotein for diagnosis of hepatocellular carcinoma in patients with chronic liver disease: influence of HBsAg and anti-HCV status. J Hepatol. 2001;34:570-575. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 512] [Cited by in RCA: 515] [Article Influence: 21.5] [Reference Citation Analysis (4)] |
| 76. | Andersson KL, Salomon JA, Goldie SJ, Chung RT. Cost effectiveness of alternative surveillance strategies for hepatocellular carcinoma in patients with cirrhosis. Clin Gastroenterol Hepatol. 2008;6:1418-1424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 187] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 77. | Sherman M. Alphafetoprotein: an obituary. J Hepatol. 2001;34:603-605. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 193] [Cited by in RCA: 186] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 78. | Sherman M. Serological surveillance for hepatocellular carcinoma: time to quit. J Hepatol. 2010;52:614-615. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 32] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 79. | Giannini EG, Farinati F, Trevisani F. Alpha-fetoprotein in hepatocellular carcinoma surveillance: wake not the dead. Hepatology. 2011;54:376-377. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 80. | Giannini EG, Erroi V, Trevisani F. Effectiveness of α-fetoprotein for hepatocellular carcinoma surveillance: the return of the living-dead? Expert Rev Gastroenterol Hepatol. 2012;6:441-444. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 81. | Poon D, Anderson BO, Chen LT, Tanaka K, Lau WY, Van Cutsem E, Singh H, Chow WC, Ooi LL, Chow P. Management of hepatocellular carcinoma in Asia: consensus statement from the Asian Oncology Summit 2009. Lancet Oncol. 2009;10:1111-1118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 283] [Cited by in RCA: 318] [Article Influence: 19.9] [Reference Citation Analysis (0)] |
| 82. | Saab S, Ly D, Nieto J, Kanwal F, Lu D, Raman S, Amado R, Nuesse B, Durazo F, Han S. Hepatocellular carcinoma screening in patients waiting for liver transplantation: a decision analytic model. Liver Transpl. 2003;9:672-681. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 54] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 83. | Mazzaferro V, Regalia E, Doci R, Andreola S, Pulvirenti A, Bozzetti F, Montalto F, Ammatuna M, Morabito A, Gennari L. Liver transplantation for the treatment of small hepatocellular carcinomas in patients with cirrhosis. N Engl J Med. 1996;334:693-699. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5110] [Cited by in RCA: 5310] [Article Influence: 183.1] [Reference Citation Analysis (0)] |
| 84. | Northup PG, Berg CL. Hepatocellular carcinoma and model for end-stage liver disease exceptions: the more we understand, the more challenging the allocation gets. Liver Transpl. 2012;18:381-383. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 85. | Vitale A, Volk ML, Gambato M, Zanus G, D’Amico F, Carraro A, Pauletto A, Bonsignore P, Scopelliti M, Polacco M. Estimation of the harm to the waiting list as a crucial factor in the selection of patients with hepatocellular carcinoma for liver transplantation. Transplant Proc. 2010;42:1194-1196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 16] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 86. | Kim CK, Lim JH, Lee WJ. Detection of hepatocellular carcinomas and dysplastic nodules in cirrhotic liver: accuracy of ultrasonography in transplant patients. J Ultrasound Med. 2001;20:99-104. [PubMed] |
| 87. | Marrero JA, Fontana RJ, Su GL, Conjeevaram HS, Emick DM, Lok AS. NAFLD may be a common underlying liver disease in patients with hepatocellular carcinoma in the United States. Hepatology. 2002;36:1349-1354. [PubMed] |
| 88. | Giannini EG, Marabotto E, Savarino V, Trevisani F, di Nolfo MA, Del Poggio P, Benvegnù L, Farinati F, Zoli M, Borzio F. Hepatocellular carcinoma in patients with cryptogenic cirrhosis. Clin Gastroenterol Hepatol. 2009;7:580-585. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 38] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 89. | Santi V, Buccione D, Di Micoli A, Fatti G, Frigerio M, Farinati F, Del Poggio P, Rapaccini G, Di Nolfo MA, Benvegnù L. The changing scenario of hepatocellular carcinoma over the last two decades in Italy. J Hepatol. 2012;56:397-405. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 78] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 90. | Singal AG, Conjeevaram HS, Volk ML, Fu S, Fontana RJ, Askari F, Su GL, Lok AS, Marrero JA. Effectiveness of hepatocellular carcinoma surveillance in patients with cirrhosis. Cancer Epidemiol Biomarkers Prev. 2012;21:793-799. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 290] [Reference Citation Analysis (0)] |
| 91. | Vernon G, Baranova A, Younossi ZM. Systematic review: the epidemiology and natural history of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis in adults. Aliment Pharmacol Ther. 2011;34:274-285. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2405] [Cited by in RCA: 2293] [Article Influence: 163.8] [Reference Citation Analysis (0)] |
| 92. | Barbara L, Benzi G, Gaiani S, Fusconi F, Zironi G, Siringo S, Rigamonti A, Barbara C, Grigioni W, Mazziotti A. Natural history of small untreated hepatocellular carcinoma in cirrhosis: a multivariate analysis of prognostic factors of tumor growth rate and patient survival. Hepatology. 1992;16:132-137. [PubMed] |
| 93. | Han KH, Kim do Y, Park JY, Ahn SH, Kim J, Kim SU, Kim JK, Lee KS, Chon CY. Survival of hepatocellular carcinoma patients may be improved in surveillance interval not more than 6 mo compared with more than 6 mo: a 15-year prospective study. J Clin Gastroenterol. 2013;47:538-544. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 36] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 94. | Trinchet JC, Chaffaut C, Bourcier V, Degos F, Henrion J, Fontaine H, Roulot D, Mallat A, Hillaire S, Cales P. Ultrasonographic surveillance of hepatocellular carcinoma in cirrhosis: a randomized trial comparing 3- and 6-mo periodicities. Hepatology. 2011;54:1987-1997. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 245] [Cited by in RCA: 294] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 95. | Nouso K, Tanaka H, Uematsu S, Shiraga K, Okamoto R, Onishi H, Nakamura S, Kobayashi Y, Araki Y, Aoki N. Cost-effectiveness of the surveillance program of hepatocellular carcinoma depends on the medical circumstances. J Gastroenterol Hepatol. 2008;23:437-444. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 40] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 96. | Cucchetti A, Trevisani F, Cescon M, Ercolani G, Farinati F, Poggio PD, Rapaccini G, Nolfo MA, Benvegnù L, Zoli M. Cost-effectiveness of semi-annual surveillance for hepatocellular carcinoma in cirrhotic patients of the Italian Liver Cancer population. J Hepatol. 2012;56:1089-1096. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 58] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 97. | Thompson Coon J, Rogers G, Hewson P, Wright D, Anderson R, Jackson S, Ryder S, Cramp M, Stein K. Surveillance of cirrhosis for hepatocellular carcinoma: a cost-utility analysis. Br J Cancer. 2008;98:1166-1175. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 81] [Cited by in RCA: 91] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 98. | Qian MY, Yuwei J R, Angus P, Schelleman T, Johnson L, Gow P. Efficacy and cost of a hepatocellular carcinoma screening program at an Australian teaching hospital. J Gastroenterol Hepatol. 2010;25:951-956. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 27] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 99. | Patel D, Terrault NA, Yao FY, Bass NM, Ladabaum U. Cost-effectiveness of hepatocellular carcinoma surveillance in patients with hepatitis C virus-related cirrhosis. Clin Gastroenterol Hepatol. 2005;3:75-84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 56] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 100. | Arguedas MR, Chen VK, Eloubeidi MA, Fallon MB. Screening for hepatocellular carcinoma in patients with hepatitis C cirrhosis: a cost-utility analysis. Am J Gastroenterol. 2003;98:679-690. [PubMed] |
| 101. | Tanaka H, Iijima H, Nouso K, Aoki N, Iwai T, Takashima T, Sakai Y, Aizawa N, Iwata K, Ikeda N. Cost-effectiveness analysis on the surveillance for hepatocellular carcinoma in liver cirrhosis patients using contrast-enhanced ultrasonography. Hepatol Res. 2012;42:376-384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 46] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 102. | Cucchetti A, Piscaglia F, Cescon M, Colecchia A, Ercolani G, Bolondi L, Pinna AD. Cost-effectiveness of hepatic resection versus percutaneous radiofrequency ablation for early hepatocellular carcinoma. J Hepatol. 2013;59:300-307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 249] [Cited by in RCA: 313] [Article Influence: 26.1] [Reference Citation Analysis (0)] |
| 103. | Roskams T. Anatomic pathology of hepatocellular carcinoma: impact on prognosis and response to therapy. Clin Liver Dis. 2011;15:245-259, vii-x. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 34] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 104. | Italian Association for the Study of the Liver (AISF); AISF Expert Panel; AISF Coordinating Committee; Bolondi L, Cillo U, Colombo M, Craxì A, Farinati F, Giannini EG, Golfieri R, Levrero M, Pinna AD, Piscaglia F, Raimondo G, Trevisani F, Bruno R, Caraceni P, Ciancio A, Coco B, Fraquelli M, Rendina M, Squadrito G, Toniutto P. Position paper of the Italian Association for the Study of the Liver (AISF): the multidisciplinary clinical approach to hepatocellular carcinoma. Dig Liver Dis. 2013;45:712-723. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 143] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 105. | Bolondi L, Gaiani S, Benzi G, Zironi G, Rigamonti A, Fusconi F, Barbara L. Ultrasonography and guided biopsy in the diagnosis of hepatocellular carcinoma. Ital J Gastroenterol. 1992;24:46-49. [PubMed] |
| 106. | Trevisani F, Cantarini MC, Wands JR, Bernardi M. Recent advances in the natural history of hepatocellular carcinoma. Carcinogenesis. 2008;29:1299-1305. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 121] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 107. | Colli A, Fraquelli M, Casazza G, Massironi S, Colucci A, Conte D, Duca P. Accuracy of ultrasonography, spiral CT, magnetic resonance, and alpha-fetoprotein in diagnosing hepatocellular carcinoma: a systematic review. Am J Gastroenterol. 2006;101:513-523. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 433] [Cited by in RCA: 387] [Article Influence: 20.4] [Reference Citation Analysis (0)] |
| 108. | Lencioni R. Surveillance and early diagnosis of hepatocellular carcinoma. Dig Liver Dis. 2010;42 Suppl 3:S223-S227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 109. | Bolondi L, Gaiani S, Celli N, Golfieri R, Grigioni WF, Leoni S, Venturi AM, Piscaglia F. Characterization of small nodules in cirrhosis by assessment of vascularity: the problem of hypovascular hepatocellular carcinoma. Hepatology. 2005;42:27-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 324] [Cited by in RCA: 309] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 110. | Golfieri R, Renzulli M, Lucidi V, Corcioni B, Trevisani F, Bolondi L. Contribution of the hepatobiliary phase of Gd-EOB-DTPA-enhanced MRI to Dynamic MRI in the detection of hypovascular small (≤ 2 cm) HCC in cirrhosis. Eur Radiol. 2011;21:1233-1242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 165] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 111. | Nakashima Y, Nakashima O, Hsia CC, Kojiro M, Tabor E. Vascularization of small hepatocellular carcinomas: correlation with differentiation. Liver. 1999;19:12-18. [PubMed] |
| 112. | Kojiro M, Roskams T. Early hepatocellular carcinoma and dysplastic nodules. Semin Liver Dis. 2005;25:133-142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 212] [Cited by in RCA: 190] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 113. | Vilana R, Forner A, Bianchi L, García-Criado A, Rimola J, de Lope CR, Reig M, Ayuso C, Brú C, Bruix J. Intrahepatic peripheral cholangiocarcinoma in cirrhosis patients may display a vascular pattern similar to hepatocellular carcinoma on contrast-enhanced ultrasound. Hepatology. 2010;51:2020-2029. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 218] [Cited by in RCA: 228] [Article Influence: 15.2] [Reference Citation Analysis (0)] |
| 114. | Barreiros AP, Piscaglia F, Dietrich CF. Contrast enhanced ultrasound for the diagnosis of hepatocellular carcinoma (HCC): comments on AASLD guidelines. J Hepatol. 2012;57:930-932. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 55] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 115. | Galassi M, Iavarone M, Rossi S, Bota S, Vavassori S, Rosa L, Leoni S, Venerandi L, Marinelli S, Sangiovanni A. Patterns of appearance and risk of misdiagnosis of intrahepatic cholangiocarcinoma in cirrhosis at contrast enhanced ultrasound. Liver Int. 2013;33:771-779. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 91] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 116. | Sersté T, Barrau V, Ozenne V, Vullierme MP, Bedossa P, Farges O, Valla DC, Vilgrain V, Paradis V, Degos F. Accuracy and disagreement of computed tomography and magnetic resonance imaging for the diagnosis of small hepatocellular carcinoma and dysplastic nodules: role of biopsy. Hepatology. 2012;55:800-806. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 104] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 117. | Davila JA, Weston A, Smalley W, El-Serag HB. Utilization of screening for hepatocellular carcinoma in the United States. J Clin Gastroenterol. 2007;41:777-782. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 99] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 118. | Davila JA, Henderson L, Kramer JR, Kanwal F, Richardson PA, Duan Z, El-Serag HB. Utilization of surveillance for hepatocellular carcinoma among hepatitis C virus-infected veterans in the United States. Ann Intern Med. 2011;154:85-93. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 241] [Cited by in RCA: 236] [Article Influence: 16.9] [Reference Citation Analysis (0)] |
| 119. | Davila JA, Kramer JR, Duan Z, Richardson PA, Tyson GL, Sada YH, Kanwal F, El-Serag HB. Referral and receipt of treatment for hepatocellular carcinoma in United States veterans: effect of patient and nonpatient factors. Hepatology. 2013;57:1858-1868. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 47] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 120. | El-Serag HB, Alsarraj A, Richardson P, Davila JA, Kramer JR, Durfee J, Kanwal F. Hepatocellular carcinoma screening practices in the Department of Veterans Affairs: findings from a national facility survey. Dig Dis Sci. 2013;58:3117-3126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 17] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 121. | Chalasani N, Said A, Ness R, Hoen H, Lumeng L. Screening for hepatocellular carcinoma in patients with cirrhosis in the United States: results of a national survey. Am J Gastroenterol. 1999;94:2224-2229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 96] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 122. | Kennedy NA, Rodgers A, Altus R, McCormick R, Wundke R, Wigg AJ. Optimisation of hepatocellular carcinoma surveillance in patients with viral hepatitis: a quality improvement study. Intern Med J. 2013;43:772-777. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 28] [Article Influence: 2.5] [Reference Citation Analysis (0)] |