Published online Jun 28, 2011. doi: 10.3748/wjg.v17.i24.2958
Revised: March 11, 2011
Accepted: March 18, 2011
Published online: June 28, 2011
AIM: To evaluate bacterial cytosine deaminase (bCD) mutant D314A and 5-fluorocytosine (5-FC) for treatment of colon cancer in a mouse model.
METHODS: Recombinant lentivirus vectors that contained wild-type bCD gene (bCDwt), and bCD mutant D314A gene (bCD-D314A) with green fluorescence protein gene were constructed and used to infect human colon carcinoma LoVo cells, to generate stable transfected cells, LoVo/null, LoVo/bCDwt or LoVo/bCD-D314A. These were injected subcutaneously into Balb/c nude mice to establish xenograft models. Two weeks post-LoVo cell inoculation, PBS or 5-FC (500 mg/kg) was administered by intraperitoneal (i.p.) injection once daily for 14 d. On the day after LoVo cell injection, mice were monitored daily for tumor volume and survival.
RESULTS: Sequence analyses confirmed the construction of recombinant lentiviral plasmids that contained bCDwt or bCD-D314A. The lentiviral vector had high efficacy for gene delivery, and RT-PCR showed that bCDwt or bCD-D314A gene was transferred to LoVo cells. Among these treatment groups, gene delivery or 5-FC administration alone had no effect on tumor growth. However, bCDwt/5-FC or bCD-D314A/5-FC treatment inhibited tumor growth and prolonged survival of mice significantly (P < 0.05). Importantly, the tumor volume in the bCD-D314A/5-FC-treated group was lower than that in the bCDwt/5-FC group (P < 0.05), and bCD-D314A plus 5-FC significantly prolonged survival of mice in comparison with bCDwt plus 5-FC (P < 0.05).
CONCLUSION: The bCD mutant D314A enhanced significantly antitumor activity in human colon cancer xenograft models, which provides a promising approach for human colon carcinoma therapy.
- Citation: Deng LY, Wang JP, Gui ZF, Shen LZ. Antitumor activity of mutant bacterial cytosine deaminase gene for colon cancer. World J Gastroenterol 2011; 17(24): 2958-2964
- URL: https://www.wjgnet.com/1007-9327/full/v17/i24/2958.htm
- DOI: https://dx.doi.org/10.3748/wjg.v17.i24.2958
Colorectal cancer has one of the highest mortalities worldwide[1]. Conventional therapies consist of surgery, chemotherapy, radiotherapy, and biotherapy. Despite advances in the treatment of colorectal cancer, the prognosis for locally advanced or metastatic disease remains relatively poor[2]. Therefore, it is crucial to develop novel therapeutic strategies, not only to completely cure cancer, but also to prevent its recurrence. Among these approaches, gene-directed enzyme-prodrug therapy (GDEPT) has received considerable attention[3,4].
Suicide gene therapy for cancer is an appealing alternative to standard methods of chemotherapy because most chemotherapeutic agents lack tumor specificity. Classic chemotherapeutic agents are often unable to distinguish tumor cells from normal dividing cells, which results in indiscriminate toxic effects. In contrast, suicide gene therapy allows for specific targeting of the tumor while preventing damage to normal cells. This is accomplished by introducing a gene that encodes a prodrug-activating enzyme into cancer cells. Although delivery systems have not been fully optimized, delivery of the gene is typically done using either a viral vector (retrovirus or adenovirus) or by other non-viral means[5-7]. Once the gene is delivered into the cancer cell, a non-toxic prodrug is administered. The enzyme converts the non-toxic prodrug into its active and lethal form that results in cancer cell death. Escherichia coli or bacterial cytosine deaminase (bCD) is responsible for the activation of the non-toxic prodrug 5-fluorocytosine (5-FC) to its toxic form, 5-fluorouracil (5-FU)[8]. The absence of an endogenous cytosine deaminase in mammalian cells provides for deamination of 5-FC only in cells that express bCD. This is followed by the conversion of 5-FU into its deoxyribonucleoside, fluorodeoxyuridine (FUdR) by thymidine phosphorylase. Upon phosphorylation of FUdR by endogenous thymidine kinase, thymidylate synthase is irreversibly inhibited by the product, 5FdUMP, thereby preventing dTTP formation and ultimately leading to inhibition of DNA synthesis.
One of the key advantages of the bCD/5-FC enzyme/prodrug system is the phenomenon known as the “bystander effect”, which is defined as the killing of untransfected cells neighboring those cells transfected with the suicide gene[8]. This type of killing has been described extensively with regard to the herpes simplex virus-1 thymidine kinase and ganciclovir enzyme/prodrug system, and occurs primarily by the transfer of toxic antimetabolites through gap junctions[9,10]. Unlike phosphorylated ganciclovir, 5-FU is a small, uncharged molecule that can pass freely in and out of the cell by diffusion. Consequently, cell-cell contact is not required for the bystander effect with bCD/5-FC; an advantage for those cell types with limited gap junctions[11]. bCD has been used successfully in gene therapy for a variety of animal tumor models and is currently under investigation for the treatment of human cancers[12-15].
The limiting factors for successful suicide GDEPT are transfection efficiency and the ability of the enzyme to turn over the prodrug, which is an analog of its natural substrate. From a kinetic perspective, 5-FC is a poor substrate for bCD (Km = 3.3 mmol/L) compared with its native substrate, cytosine (Km = 0.2 mmol/L)[16]. Thus, high doses of this prodrug must be administered to achieve cell killing. The plasma levels of 5-FC required to obtain a significant amount of active metabolites may lead to adverse effects. This is observed with 5-FC, whereas deamination by CD of bacterial intestinal microflora into 5-FU is responsible for side effects[17]. Other studies have suggested that the CD from Saccharomyces cerevisiae (yCD) displays a kinetic advantage towards 5-FC over bCD[18]. However, yCD is considerably less thermostable than bCD; a characteristic that may make the bacterial enzyme a preferable catalytic system for gene therapy.
Fortunately, Mahan et al[16,19] have used random mutagenesis to create novel bCDs that demonstrate an increased preference for 5-FC over cytosine. Among these mutants isolated, the mutant D314A [substitution of an alanine (A) for the aspartic acid (D) at position 314 of bCD] enzyme demonstrates a dramatic decrease in cytosine activity (17-fold), as well as a slight increase in activity toward 5-FC (twofold), which indicates that mutant D314A prefers the prodrug over cytosine by almost 20-fold. Despite the thermolability of yCD, others have suggested that yCD is superior to bCD in gene therapy settings because of a 23-fold relative substrate preference for 5-FC that is displayed by yCD[18]. However, given the thermostability of bCD and the 19-fold relative substrate preference that the bCD mutant D314A displays towards 5-FC, bCD D314A may be a superior suicide gene to yCD. These results indicate that bCD mutant D314A is a superior candidate for suicide gene therapy. Recently, this mutant D314A has been demonstrated to enhance cytotoxicity of human glioma and pancreatic cancer cells, and to increase therapeutic efficacy against human glioma and human pancreatic tumor xenografts, especially combined with radiotherapy[20,21]. However, there have been only a few studies of bCD mutants in colorectal cancer.
Previously, we have used the bCD gene to treat colon cancer, and have found that the efficacy of wild-type bCD is not sufficient to abolish cancer cells in vitro or in vivo, therefore, combination therapy with other genes, such as interleukin-2 or interferon-γ, is needed to improve the cytotoxicity of bCD[7,22,23]. Recently, we have constructed bCD-D314A using site-directed mutagenesis[19], and have demonstrated that it has significantly increased cytotoxicity in human colon cancer cell line LoVo, compared with wild-type bCD (bCDwt) in vitro[24].
The aim of the present study was to investigate whether bCD-D314A suicide gene and 5-FC prodrug therapy produce increased therapeutic efficacy in vivo for human colon cancer in nude mice using lentiviral vectors. The results presented here indicated that mutant bCD-D314A was able to significantly enhance antitumor efficacy in human colon cancer xenograft models in vivo compared with bCDwt.
Human colon cancer cell line LoVo (CCL-229; ATCC, Manassas, VA, USA) was cultured in RPMI-1640 medium (Gibco, Invitrogen, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum (FBS; Gibco). HEK 293T (human embryonic kidney 293T cell line containing SV40 large T antigen) (CRL-11268; ATCC) was cultured in Dulbecco’s Modified Eagle’s Medium (Gibco) supplemented with 10% cosmic calf serum (Hyclone, Logan, UT, USA), 2 mmol/L L-glutamine (Sigma, St. Louis, MO, USA), 100 U/mL penicillin (Sigma) and 0.1 mg/mL streptomycin (Sigma). LoVo and HEK 293T cells were in a 5% CO2-humidified atmosphere at 37°C.
5-FC was obtained from Sigma. Restriction enzymes HindIII, KpnI, NheI, EcoRI and DpnI, T4 DNA ligase, pfu DNA polymerase, DNA marker DL2000 and PCR reagents were obtained from Takara (Otsu, Shiga, Japan). Primers were chemically synthesized by Shanghai Generay Co. Ltd. (Shanghai, China). The plasmid DNA extraction (Mini) kit was provided by Qiagen (Crawley, West Sussex, UK). TRIzol and Lipofectamine 2000™ transfection reagent were obtained from Invitrogen.
Female athymic Balb/c (nu/nu) nude mice were purchased from Shanghai SLAC Laboratory Animal Co. Ltd. (Shanghai, China) and were housed under aseptic conditions in micro-isolator cages, which were approved by the local Institutional Animal Care and Use Committee.
The plasmid pcDNA3.1/bCDwt that contained whole-length wild-type bCD gene, and pcDNA3.1/bCD-D314A that contained the mutant D314A gene, were prepared and stored in our department. The pLJM1-GFP lentivirus vector with green fluorescence protein (GFP) gene was a generous gift from Prof. J Li (Nanjing Medical University).
The pcDNA3.1-bCDwt and pcDNA3.1-bCD-D314A plasmids were all double digested with HindIII and KpnI. The products of enzyme digestion were connected to the lentiviral vector pLJM1-GFP, which was double digested with NheI and EcoRI, to produce pLJM1-bCDwt-GFP and pLJM1-bCD-D314A-GFP. These plasmids were then transformed into E. coli XL1-Blue. The colonies were selected for PCR identification. The sense sequence of bCD primers was 5'-CGCAAATGGGCGGTAGGCGTG-3', whereas the antisense sequence was 5'AATTCTCAACGTTTGTAATCGATGG-3'. These recombinant plasmids were extracted and sent to BGI Sequencing Company (Shanghai, China) for sequencing.
To produce recombinant lentiviruses that encoded bCDwt, bCD-D314A or GFP gene, three types of plasmids (pLJM1-bCDwt-GFP, pLJM1-bCD-D314A-GFP and pLJM1-GFP) were transfected to 293T cells according to the instructions for Lipofectamine 2000™ (Invitrogen). The virus-containing supernatant was collected 48 h after transfection, concentrated by centrifugation (4000 r/mim, 4°C for 5 min), and filtered with a 0.45-μm membrane filter. The virus titers were determined in 293T cells.
For LoVo cell infection, there were three groups: bCDwt-GFP, bCD-D314A-GFP, and GFP (null). LoVo cells were seeded at a density of 1 × 105 cells in a 60-mm plate and infected with different lentiviral vectors in the presence of 10 μg/mL polybrene (Millipore, Billerica, MA, USA). At 10-12 h post-infection, the growth medium was replaced. Forty-eight hours later, the GFP expression of transduced cells was observed under fluorescence microscopy. LoVo cells were infected twice in the same way. At 3 d after transfection, the FACSCalibur flow cytometer (BD, Franklin Lakes, NJ, USA) was used for fluorescence-activated cell sorting and then stable transfected cells, LoVo/null, LoVo/bCDwt and LoVo/bCD-D314A were cultured in a 5% CO2-humidified incubator at 37°C.
Total RNA was extracted from transfected LoVo cells harvested from the different groups with TRIzol reagent. First-strand cDNA was synthesized by reverse transcription according to the instructions for M2MLV (Promega, Madison, WI, USA). The sense sequence of bCD primers used in RT-PCR was 5'TTATGTCGAATAACGCTTTACAAAC-3', whereas the antisense sequence was 5'TACCTCCACGTTTGTAATCGATGGC-3'. PCR was performed for 35 cycles (94°C for 1 min, 60°C for 1.5 min, 72°C for 1.5 min) in an automated DNA Thermal Cycler (Perkin-Elmer Cetus, Norwalk, CT, USA).
To compare antitumor effects of bCDwt- and bCD-D314A-mediated molecular chemotherapy in vivo, combination of 5-FC, pools of LoVo, LoVo/null, LoVo/bCDwt or LoVo/bCD-D314A cells [5 × 106 cells in 100 μL PBS (pH 7.3)] were injected subcutaneously (s.c.) into the right flanks of 5-6-wk-old female Balb/c nude mice (n = 20, respectively). Two weeks post-LoVo cell inoculation, PBS or 5-FC (500 mg/kg) was administered by i.p. injection once daily for 14 d. Starting at day 1, the tumor volume was monitored daily using caliper measurement, calculated using the formula: π/6 × (width × length2). On the day after LoVo cell injection, mice were monitored daily for survival.
The treatment groups were compared with respect to tumor size. To test for significant differences in tumor volume among treatment groups, one-way ANOVA was conducted. When ANOVA indicated that a significant difference existed (P < 0.05), multiple comparison procedures were used to determine where the differences lay. Kaplan-Meier survival curves were analyzed by the log-rank test, and specific pairwise multiple comparisons were made using the Holm-Sidak method. All comparisons were made using the 0.05 level of significance.
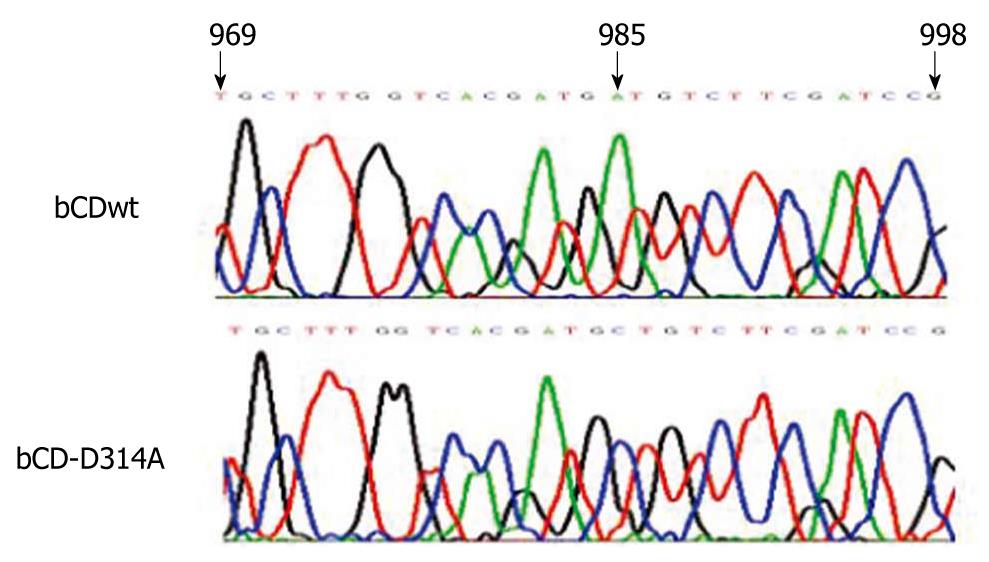
Sequencing results of recombinant lentiviral plasmids showed that pLJM1-bCDwt-GFP contained the wild-type bCD gene, and pLJM1-bCD-D314A-GFP contained the mutant D314A (Figure 1), which indicated that the two recombinant lentiviral plasmids were constructed successfully.
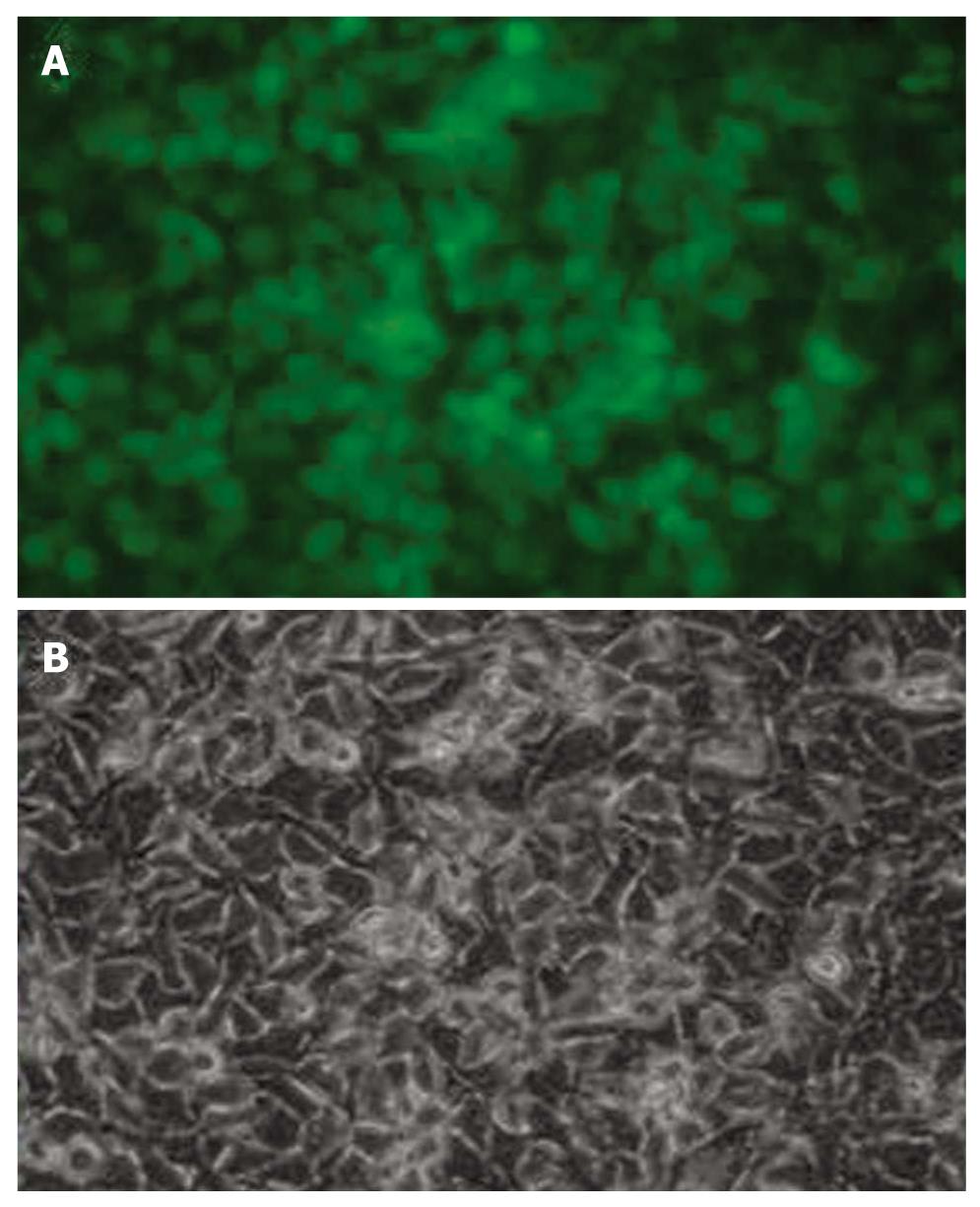
Lentiviral vectors have improved efficiency to deliver genes. In this study, GFP was used as a reporter gene. Figure 2 shows that the efficacy of gene delivery by lentiviral vectors was satisfactory.
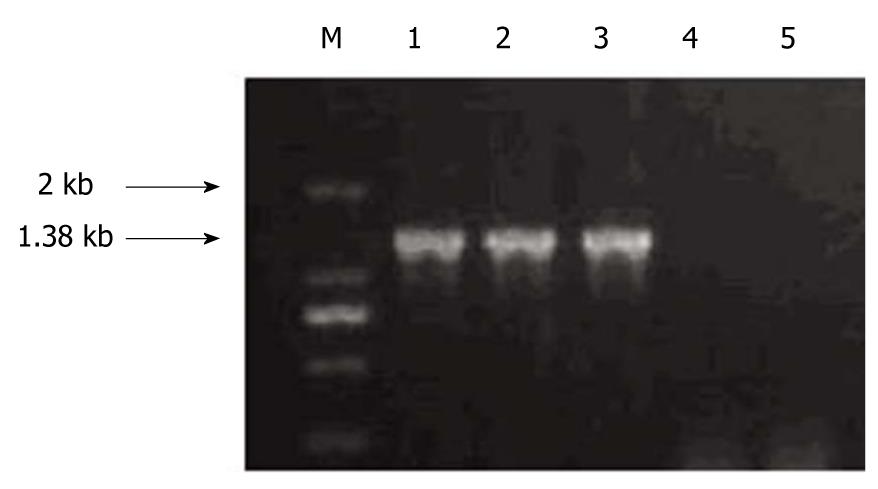
LoVo cells transfected with different lentiviral vectors, LoVo/null, LoVo/bCDwt and LoVo/bCD-D314A, were subjected to RT-PCR to identify bCD gene expression. As shown in Figure 3, LoVo/bCDwt and LoVo/bCD-D314A cells had bCD gene expression, while bCD gene was not detected in LoVo/null cells.
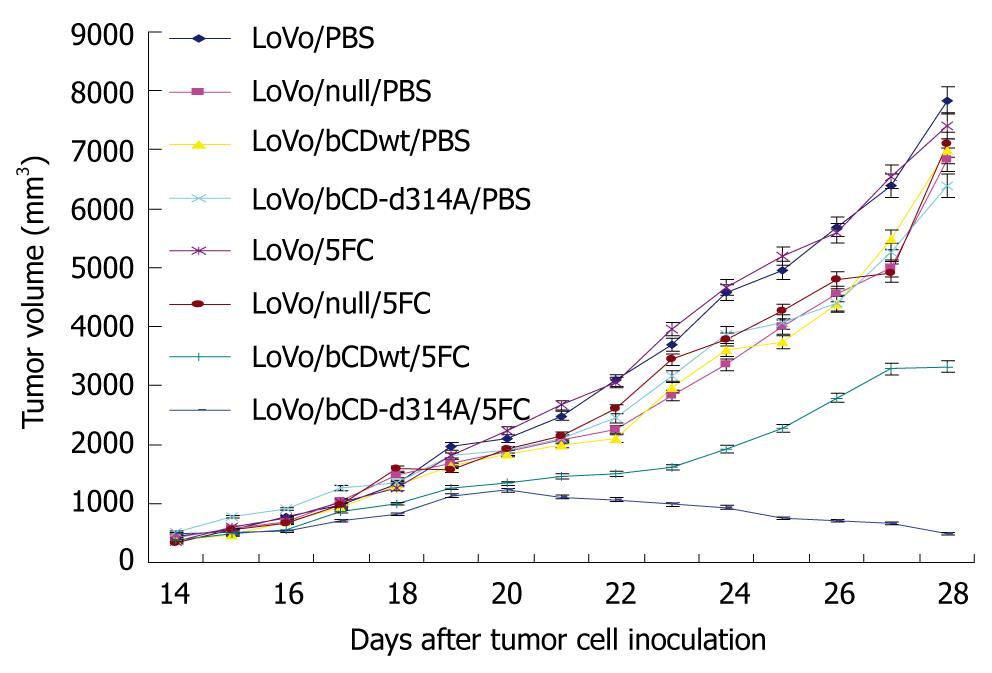
To evaluate the potential of bCD-D314A gene therapy with 5-FC in vivo, LoVo cells with different stably gene delivery were injected s.c. into the right flank of athymic nude mice. Two weeks after cell inoculation, before treatment, the tumors in each group were palpable and the mean volumes in each group did not differ significantly among the treatment groups (P > 0.05), and within treatment variances (PBS vs 5-FC) were not significantly different (P > 0.05). The baseline mean tumor volume at 14 d after tumor cell injection was 412.63 ± 36.79 mm3.
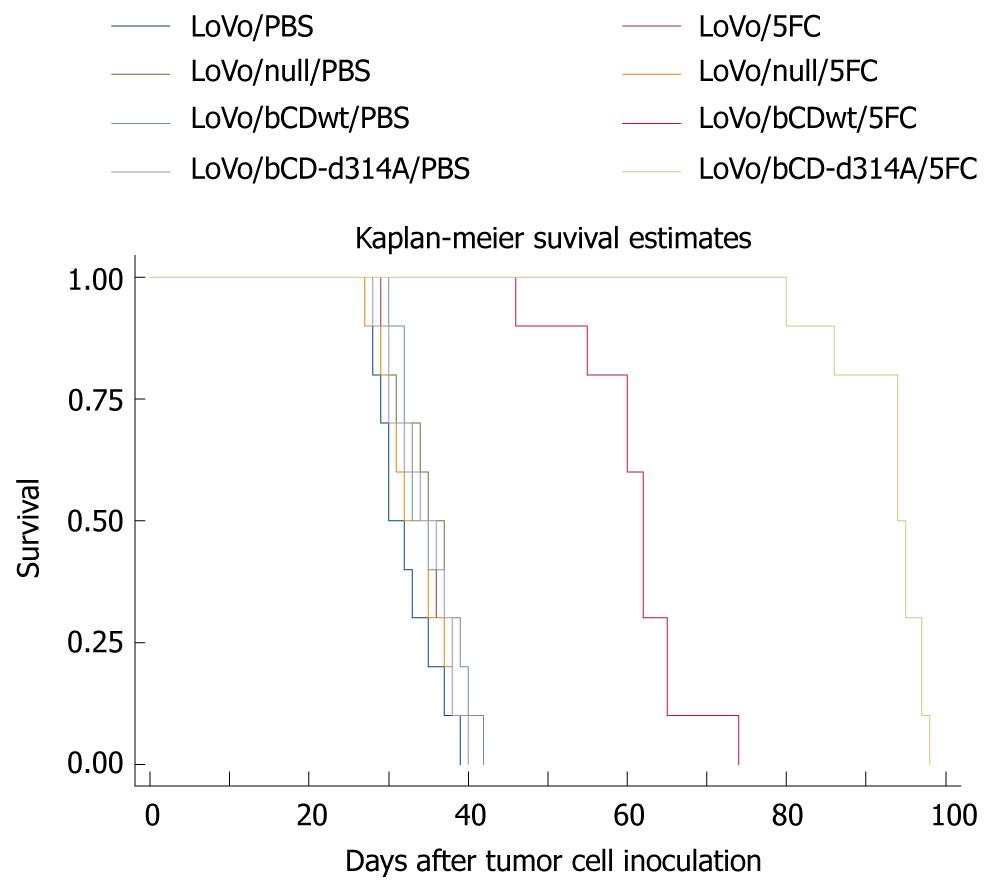
PBS or 5-FC (500 mg/kg) was administered i.p. once daily for 2 wk. Starting at day 1, mice were monitored for tumor volume and survival. Inhibition of tumor growth was initially noted in mice treated with LoVo/bCDwt or LoVo/bCD-D314A in combination with 5-FC compared with the other groups on day 20 (P < 0.05) (Figure 4). There were no significant differences in tumor growth between the other groups (P > 0.05), which indicated that gene delivery or 5-FC administration alone had no influence on tumor growth. From day 20 onwards, tumors in the mice treated with LoVo/bCD-D314A and 5-FC shrunk daily, whereas the tumors in mice treated with LoVo/bCDwt and 5-FC increased gradually. The difference in tumor volume between these two groups became increasingly marked (P < 0.05). At the same time, the tumors in the other groups kept growing (Figure 4).
As to the influence of bCD-D314A and bCDwt on survival, we showed that bCD-D314A/5-FC or bCDwt/5-FC treatment significantly prolonged survival of mice in comparison with the other groups. As shown in Figure 5, the median survival time of other groups was about 35 d and there was no difference among them (P > 0.05), whereas it was prolonged to 62 or 94 d in the bCDwt/5-FC or bCD-D314A/5-FC group, respectively (P < 0.05). Furthermore, bCD-D314A plus 5-FC significantly prolonged survival of mice in comparison with bCDwt plus 5-FC (P < 0.05).
Worldwide, more than one million individuals will develop colorectal cancer annually, and the disease-specific mortality rate is nearly 33% in the developed world[1]. Substantial progress has been made in understanding the molecular pathogenesis, diagnosis (hereditary and sporadic), and treatment of colorectal cancer during recent decades. Despite the use of 5-FU-based combination chemotherapy and active targeted drugs for treatment of metastatic colorectal cancer in the past decade, and improvement in overall survival for non-resectable disease, cure rates remain low[25]. There is a need for the development of new alternative therapeutic strategies. Gene therapy is a novel approach that might lead to improved treatments for colorectal cancer. Among these approaches, GDEPT using the bCD/5-FC system has been developed. The gene that encodes the CD that converts the prodrug 5-FC to 5-FU is delivered to the target tumor cells, which results in their death.
The most important characteristic of suicide gene therapy is its bystander effect. Although the viral or non-viral gene delivery systems currently available have poor efficacy for in vivo gene transfer, complete eradication of tumors has been seen in some experimental animal models, which is thought to depend on the bystander killing effect. In the bCD/5-FC system, the bystander effect is caused by the passive diffusion of 5-FU into the extracellular milieu and its diffusion into the adjacent cells, which requires no gap junctions[11]. The immune-related response also contributes to the bystander effect[26], which has been confirmed by our experimental results from the immunocompetent and immunodeficient mice[27]. Although this approach has been in development for several decades, new combinations with cancer therapies, such as selective conventional chemotherapy[28] and radiotherapy[29], are being tested.
Unlike conventional chemotherapy, suicide gene therapy renders specific killing of the tumor cells that express the suicide gene, but it may lead to systemic toxicity if these genes are delivered to normal cells. Thus, target specificity is of great importance to suicide gene therapy. The rationale behind suicide gene therapy is that, after targeted transfer of these genes into tumor cells, only tumor and neighboring cells will be rendered sensitive to their cytotoxic action. Specifically, targeted expression of the prodrug-activating enzyme avoids systemic toxicity, and results in high drug concentrations in the tumor mass and an improved therapeutic index compared with non-targeted gene delivery. To kill carcinoembryonic antigen (CEA)-positive colorectal carcinoma cells specifically using the bCD/5-FC system, we have constructed a new replication-deficient recombinant adenoviral vector that contains the bCD gene controlled by the CEA promoter, AdCEACD, and have evaluated its in vitro cytotoxic effects. We have shown that this vector can transfer bCD to CEA-positive tumor cells specifically by comparing the vector with cytomegalovirus (CMV) promoter, AdCMVCD[7]. However, the cytotoxic effects of bCD/5-FC decreased to some extent[22,23]. Although this loss of activity may be due to differences in transcriptional activation between the CEA and CMV promoters, the low affinity displayed by wild-type bCD towards 5-FC in comparison with cytosine is thought to be the principal factor that leads to the relatively poor turnover of 5-FC of wild-type bCD and limits the overall therapeutic response.
It has been shown previously that the bCD mutant, D314A, decreased efficiency for endogenous cytosine, which can compete with the prodrug for the active enzyme site, in combination with increased efficiency for 5-FC that resulted in a 19-fold relative substrate preference for 5-FC in comparison with bCDwt[16,19]. The bCD mutant D314A has been demonstrated to be an excellent candidate for subsequent preclinical comparisons with wild-type bCD and yCD.
Recently, we have constructed the bCD mutant D314A using site-directed mutagenesis. The in vitro results have indicated that its killing and bystander effects on human colon cancer LoVo cells are enhanced significantly as compared with wild-type bCD[24]. Thus, the rationale for using the mutant bCD gene for colon carcinoma in vivo is that the bCD mutant D314A can more effectively convert 5-FC to 5-FU, and increase the antitumor activity and prolong survival.
In the present study, we investigated mutant bCD gene transfer with lentiviral vector for treatment of human colon cancer in xenograft models. Lentivirus-based vectors (lentivectors) have been developed with improved efficiency, specificity, and safety, and are being increasingly used in basic and applied research. Clinical trials of human gene therapy are currently underway using lentivectors in a wide range of human diseases[30]. In the present study, lentiviral vector was used to transfer suicide genes. These preliminary results confirmed the efficacy of lentiviral vector for suicide gene delivery.
After the LoVo cells stably transfected with bCDwt gene or mutant bCD-D314A gene were established, they were inoculated into athymic nude mice to produce xenograft tumor models. Afterwards, 5-FC was administered. As expected, a more potent cytotoxicity effect for colon cancer was obtained using bCD-D314A/5-FC treatment in comparison with bCDwt/5-FC. During 5-FC administration, the tumors treated with bCDwt/5-FC or bCD-D314A/5-FC grew slower than those in other treatment groups, which indicated that 5-FC or suicide gene transfer alone had no effect on colon cancer. The comparative study of bCD-D314A/5-FC and bCDwt/5-FC showed an increased antitumor effect, and decreased tumor growth was observed following bCD-D314A/5-FC gene therapy. Furthermore, survival analysis showed that bCD-D314A/5-FC therapy prolonged life significantly, which confirmed the enhanced antitumor activity of bCD mutant D314A.
Although the intratumoral or blood 5-FU concentration was not estimated after 5-FC administration in this study, the enhanced antitumor effect of bCD mutant D314A in combination with 5-FC was thought to be due to its ability to convert 5-FC to 5-FU more effectively, which is consistent with recently published data in other tumor models[20,21]. The blood and tumor levels of 5-FC and 5-FU are a subject for future studies that will enable a rational dosing strategy.
In summary, our studies provide preliminary evidence that treatment using bCD mutant D314A for suicide gene/5-FC prodrug therapy is a promising approach for treatment of human colon carcinoma. Further studies on delivery systems, doses and protocols would be worthwhile to optimize this approach.
Suicide gene therapy is an appealing alternative to conventional therapies for colorectal cancer. Bacterial cytosine deaminase (bCD) can convert the non-toxic prodrug 5-fluorocytosine (5-FC) to its toxic form, 5-fluorouracil (5-FU). However, 5-FC is a poor substrate for bCD compared with its native substrate, cytosine, and its antitumor effect is limited.
The bCD mutant, D314A, has been shown to prefer 5-FC over cytosine by nearly 20-fold, and has been demonstrated to enhance therapeutic efficacy against human glioma and human pancreatic tumors. However, there have been only a few studies of bCD mutants in colorectal cancer.
Recently, the authors have constructed the bCD mutant, D314A, using site-directed mutagenesis, and have demonstrated that D314A has significantly increased cytotoxicity on human colon cancer cell line LoVo compared with wild-type bCD (bCDwt). The present study indicated that mutant D314A was able to significantly enhance antitumor efficacy in human colon cancer xenograft models compared with bCDwt.
Collectively, these studies provide preliminary evidence that treatment using bCD mutant D314A for suicide gene/5-FC prodrug therapy provides a promising approach for human colon carcinoma.
Suicide gene therapy is a form of gene-directed enzyme-prodrug therapy. When the suicide gene is delivered to cancer cells and a non-toxic prodrug is administered, the enzyme converts the non-toxic prodrug into its active and lethal form, which results in cancer cell death.
The authors investigated whether bCD mutant D314A suicide gene and 5-FC prodrug therapy increased therapeutic efficacy in a nude mouse model of human colon cancer, using lentiviral vectors. It revealed for the first time that D314A significantly enhanced antitumor activity in human colon cancer xenograft models. The results are useful and may provide a new strategy to treat colorectal cancer.
Peer reviewer: Dr. Dinesh Vyas, Department of Minimally and Endoscopic Surgery, St John Mercy Hospital, 851 E Fifth Street, Washington, MO 63090, United States
S- Editor Sun H L- Editor Kerr C E- Editor Ma WH
| 1. | Jemal A, Siegel R, Xu J, Ward E. Cancer statistics, 2010. CA Cancer J Clin. 2010;60:277-300. |
| 2. | Weiss J, Moghanaki D, Plastaras JP, Haller DG. Improved patient and regimen selection in locally advanced rectal cancer: who, how, and what next? Clin Colorectal Cancer. 2009;8:194-199. |
| 3. | Sharma A, Tandon M, Bangari DS, Mittal SK. Adenoviral vector-based strategies for cancer therapy. Curr Drug ther. 2009;4:117-138. |
| 4. | Wu C, Lin J, Hong M, Choudhury Y, Balani P, Leung D, Dang LH, Zhao Y, Zeng J, Wang S. Combinatorial control of suicide gene expression by tissue-specific promoter and microRNA regulation for cancer therapy. Mol Ther. 2009;17:2058-2066. |
| 5. | Figueiredo ML, Kao C, Wu L. Advances in preclinical investigation of prostate cancer gene therapy. Mol Ther. 2007;15:1053-1064. |
| 6. | Chaszczewska-Markowska M, Stebelska K, Sikorski A, Madej J, Opolski A, Ugorski M. Liposomal formulation of 5-fluorocytosine in suicide gene therapy with cytosine deaminase-for colorectal cancer. Cancer Lett. 2008;262:164-172. |
| 7. | Shen LZ, Wu WX, Xu DH, Zheng ZC, Liu XY, Ding Q, Hua YB, Yao K. Specific CEA-producing colorectal carcinoma cell killing with recombinant adenoviral vector containing cytosine deaminase gene. World J Gastroenterol. 2002;8:270-275. |
| 8. | Yazawa K, Fisher WE, Brunicardi FC. Current progress in suicide gene therapy for cancer. World J Surg. 2002;26:783-789. |
| 9. | Huang Q, Liu XZ, Kang CS, Wang GX, Zhong Y, Pu PY. The anti-glioma effect of suicide gene therapy using BMSC expressing HSV/TK combined with overexpression of Cx43 in glioma cells. Cancer Gene Ther. 2010;17:192-202. |
| 10. | Garcia-Rodríguez L, Abate-Daga D, Rojas A, González JR, Fillat C. E-cadherin contributes to the bystander effect of TK/GCV suicide therapy and enhances its antitumoral activity in pancreatic cancer models. Gene Ther. 2011;18:73-81. |
| 11. | Spasojević I, Maksimović V, Zakrzewska J, Bacić G. Effects of 5-fluorouracil on erythrocytes in relation to its cardiotoxicity: membrane structure and functioning. J Chem Inf Model. 2005;45:1680-1685. |
| 12. | Brown NL, Lemoine NR. Clinical trials with GDEPT: cytosine deaminase and 5-fluorocytosine. Methods Mol Med. 2004;90:451-457. |
| 13. | Freytag SO, Stricker H, Pegg J, Paielli D, Pradhan DG, Peabody J, DePeralta-Venturina M, Xia X, Brown S, Lu M. Phase I study of replication-competent adenovirus-mediated double-suicide gene therapy in combination with conventional-dose three-dimensional conformal radiation therapy for the treatment of newly diagnosed, intermediate- to high-risk prostate cancer. Cancer Res. 2003;63:7497-7506. |
| 14. | Breton E, Goetz C, Kintz J, Accart N, Aubertin G, Grellier B, Erbs P, Rooke R, Constantinesco A, Choquet P. In vivo preclinical low-field MRI monitoring of tumor growth following a suicide-gene therapy in an orthotopic mice model of human glioblastoma. C R Biol. 2010;333:220-225. |
| 15. | Shi DZ, Hu WX, Li LX, Chen G, Wei D, Gu PY. Pharmacokinetics and the bystander effect in CD::UPRT/5-FC bi-gene therapy of glioma. Chin Med J (Engl). 2009;122:1267-1272. |
| 16. | Mahan SD, Ireton GC, Stoddard BL, Black ME. Alanine-scanning mutagenesis reveals a cytosine deaminase mutant with altered substrate preference. Biochemistry. 2004;43:8957-8964. |
| 17. | Diasio RB, Lakings DE, Bennett JE. Evidence for conversion of 5-fluorocytosine to 5-fluorouracil in humans: possible factor in 5-fluorocytosine clinical toxicity. Antimicrob Agents Chemother. 1978;14:903-908. |
| 18. | Stolworthy TS, Korkegian AM, Willmon CL, Ardiani A, Cundiff J, Stoddard BL, Black ME. Yeast cytosine deaminase mutants with increased thermostability impart sensitivity to 5-fluorocytosine. J Mol Biol. 2008;377:854-869. |
| 19. | Mahan SD, Ireton GC, Knoeber C, Stoddard BL, Black ME. Random mutagenesis and selection of Escherichia coli cytosine deaminase for cancer gene therapy. Protein Eng Des Sel. 2004;17:625-633. |
| 20. | Kaliberov SA, Market JM, Gillespie GY, Krendelchtchikova V, Della Manna D, Sellers JC, Kaliberova LN, Black ME, Buchsbaum DJ. Mutation of Escherichia coli cytosine deaminase significantly enhances molecular chemotherapy of human glioma. Gene Ther. 2007;14:1111-1119. |
| 21. | Kaliberova LN, Della Manna DL, Krendelchtchikova V, Black ME, Buchsbaum DJ, Kaliberov SA. Molecular chemotherapy of pancreatic cancer using novel mutant bacterial cytosine deaminase gene. Mol Cancer Ther. 2008;7:2845-2854. |
| 22. | Shen LZ, Hua YB, Wu WX, Xu DH, Ding Q, Liu XY, Wang GL. IL-2 gene therapy enhances cytotoxic effect of E. Coli. Cytosine deaminase gene for colon cancer. Zhonghua Weichang Waike Zazhi. 2004;7:411-413. |
| 23. | Shen LZ, Hua YB, Wu WX, Xu DH, Ding Q, Chen GY, Zheng ZC, Liu XY. Enhancement of tumor killing using a combination of E. Coli. Cytosine deaminase gene and INF- gene therapy. Acta Univ Med Nanjing. 2004;24:618-620. |
| 24. | Sun MC, Huang YM, Zhu ZC, Wang JP, Shen LZ, Wu WX. Inhubitory effect of mutant cytosine deaminase D314A against huaman colon cancer cells. Zhongguo Zhongliu Shengwu Zhiliao Zazhi. 2009;16:595-599. |
| 25. | Cunningham D, Atkin W, Lenz HJ, Lynch HT, Minsky B, Nordlinger B, Starling N. Colorectal cancer. Lancet. 2010;375:1030-1047. |
| 26. | Agard C, Ligeza C, Dupas B, Izembart A, El Kouri C, Moullier P, Ferry N. Immune-dependent distant bystander effect after adenovirus-mediated suicide gene transfer in a rat model of liver colorectal metastasis. Cancer Gene Ther. 2001;8:128-136. |
| 27. | Shen LZ, Wu WX, Hua YB, Ding Q, Chen T. The relationship between the bystander effect of E. coli cytosine deaminase gene depends on the immune status of host. Linchuang Zhongliuxue Zazhi. 2004;9:237-240. |
| 28. | Shen LZ, Ding Q, Wu WX, Xu DH, Liu XY, Zheng ZC, Wu ZY. In vitro effect of cytosine deaminase gene therapy and chemical reagents on colon cancer cell line. Zhonghua Weichang Waike Zazhi. 2002;17:404-406. |
| 29. | Xing L, Sun X, Deng X, Kotedia K, Urano M, Koutcher JA, Ling CC, Li GC. Expression of the bifunctional suicide gene CDUPRT increases radiosensitization and bystander effect of 5-FC in prostate cancer cells. Radiother Oncol. 2009;92:345-352. |
| 30. | Escors D, Breckpot K. Lentiviral vectors in gene therapy: their current status and future potential. Arch Immunol Ther Exp (Warsz). 2010;58:107-119. |