Published online Feb 21, 2009. doi: 10.3748/wjg.15.817
Revised: January 10, 2009
Accepted: January 17, 2009
Published online: February 21, 2009
In adults, the hepatobiliary system, together with the kidney, constitute the main routes for the elimination of several endogenous and xenobiotic compounds into bile and urine, respectively. However, during intrauterine life the biliary route of excretion for cholephilic compounds, such as bile acids and biliary pigments, is very poor. Although very early in pregnancy the fetal liver produces bile acids, bilirubin and biliverdin, these compounds cannot be efficiently eliminated by the fetal hepatobiliary system, owing to the immaturity of the excretory machinery in the fetal liver. Therefore, the potentially harmful accumulation of cholephilic compounds in the fetus is prevented by their elimination across the placenta. Owing to the presence of detoxifying enzymes and specific transport systems at different locations of the placental barrier, such as the endothelial cells of chorionic vessels and trophoblast cells, this organ plays an important role in the hepatobiliary-like function during intrauterine life. The relevance of this excretory function in normal fetal physiology is evident in situations where high concentrations of biliary compounds are accumulated in the mother. This may result in oxidative stress and apoptosis, mainly in the placenta and fetal liver, which might affect normal fetal development and challenge the fate of the pregnancy. The present article reviews current knowledge of the mechanisms underlying the hepatobiliary function of the fetal-placental unit and the repercussions of several pathological conditions on this tandem.
- Citation: Macias RI, Marin JJ, Serrano MA. Excretion of biliary compounds during intrauterine life. World J Gastroenterol 2009; 15(7): 817-828
- URL: https://www.wjgnet.com/1007-9327/full/v15/i7/817.htm
- DOI: https://dx.doi.org/10.3748/wjg.15.817
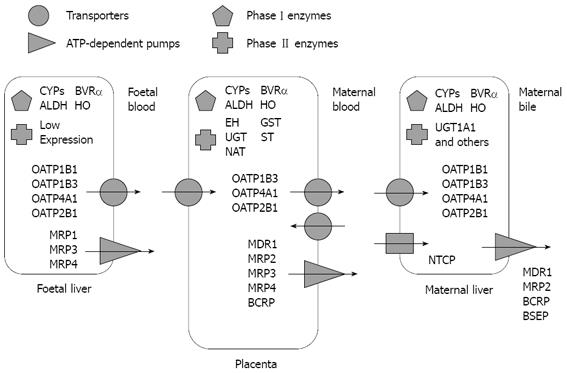
In the adult liver, most cholephilic organic anions are taken up from the portal blood by hepatocytes across the basolateral plasma membrane by sodium-dependent and -independent carriers (Figure 1). These are members of two groups of proteins: (1) the organic anion-transporting polypeptides family (OATP, gene symbol SLCO), whose isoforms OATP1B1, OATP1B3 and to a lesser extent OATP1A2[1], play a major role in the uptake of cholephilic compounds by human hepatocytes; (2) the Na+-taurocholate-cotransporting polypeptide (NTCP, SLC10A1)[2]. Several members of the organic anion transporter (OAT) and organic cation transporter (OCT) family (gene symbol SLC22A) collaborate in the uptake of a large variety of organic molecules by the liver.
The secretion of cholephilic compounds into bile is accounted for by export pumps located at the canalicular plasma membrane. These are proteins belonging to the ATP-binding cassette (ABC) superfamily, which, in this region of the hepatocyte include the P-glycoprotein or multidrug resistance protein (MDR1; ABCB1), able to transport organic and inorganic cations[3]; the sister of P-glycoprotein or bile salt export pump (BSEP; ABCB11), which constitutes the main secretory system for bile acids[4]; the isoform 2 of the multidrug resistance-associated protein (MRP2; ABCC2), which exports conjugated forms of bilirubin, bile acids and xenobiotics[56], and the breast cancer resistance protein (BCRP; ABCG2), able to export sulfated steroids, which probably include bile acids[7].
In normally functioning healthy adult livers, at least as far as the excretion of cholephilic compounds into bile is concerned, the expression levels of MRP1 (ABCC1) and MRP3 (ABCC3) at the basolateral membrane of hepatocytes is low[89]. However when the biliary excretory route is impaired, such as in cholestasis or endotoxemia, cholephilic compounds accumulate in hepatocytes, inducing an up-regulation of basolateral export pumps[10–12]. This acts as an adaptative response to reduce the cytotoxic effects of cholephilic compounds by pumping them back to the systemic circulation and accounts for an increased elimination of these substances into urine[13].
During pregnancy, owing to the immaturity of the fetal hepatobiliary excretory function, the existence of an alternative mechanism for the detoxification of cholephilic compounds produced by the fetus is required. The placenta, in collaboration with the maternal liver, carries out this function, which is very important for maintaining low bile acid and bilirubin levels in the fetal compartment. Moreover, the placenta also protects the fetal compartment, at least to a certain extent, from potentially toxic compounds coming from the maternal blood[14]. When the fetal-maternal homeostasis is altered, as happens during intrahepatic cholestasis of pregnancy, and these molecules accumulate in the conceptus, the consequences can be as serious as stillbirth and fetal death[15].
Fetal bile acid synthesis and maturation of the enzyme equipment required for bile acid and bile pigment metabolism precede the development of an efficient biliary-secretory system. Thus, although during intrauterine life bile acids are not required for digestive purposes, the fetal liver is able, from very early on during gestation, to synthesize primary bile acids, mainly cholic acid and chenodeoxycholic acid from cholesterol. Indeed, these two molecules are the major components of the human fetal bile acid pool[1617]. The fetal bile acid pool is also characterized by the presence of molecular species with hydroxyl groups in positions that are unusual in bile acids found in adults. These are C-1, C-4 and C-6[18], which convert the molecule into a more hydrophilic one. This is believed to protect the fetal liver against the cytotoxic effect of less polar bile acid species when detoxification pathways are poorly developed. Another important characteristic of the fetal bile acid pool is the existence of bile acids with “flat” structures, accounted for by the presence of Δ4 or Δ5 insaturations or the alpha configuration of a hydroxyl in C-5[18]. Although the fetal gut is germ-free, the bile acid pool contains small amounts of secondary bile acids, such as deoxycholic acid and lithocholic acid, together with tertiary bile acids, such as ursodeoxycholic acid. This is probably due to placental transfer of these compounds from the maternal circulation[19].
Data collected from both rats[20] and humans[161921] have revealed that serum bile acid concentrations are higher in fetuses than in their mothers, and that the composition of the bile acid species in both compartments is different. This has been explained in terms of the selective transplacental transfer of these cholephilic compounds[19], together with a different degree of maturation of the enzymatic machinery involved in bile acid metabolism[22]. The recently described role of bile acids as signaling molecules with several endocrine and paracrine functions[23] might account for the yet unknown physiological meaning of the early synthesis and special composition of the bile acid pool in fetuses.
From early gestation, the fetus also produces biliary pigments. The green pigment biliverdin, mainly the IXα isomer, is generated by cleavage of protoporphyrin IX by heme oxygenase[2425] and is reduced to the golden pigment bilirubin IXα by the enzyme biliverdin reductase. The high production of bilirubin by the fetal liver, together with the still low activity in this organ of glucuronosyl transferase, the enzyme that produces more polar glucuronide conjugates to facilitate biliary excretion in adults, account for the higher concentrations of the unconjugated pigment in fetal serum than in maternal serum[1926].
For many years, these bile pigments were considered mere waste products from heme metabolism, and the biological advantage of the conversion of the water-soluble and non-toxic compound biliverdin into the poorly water-soluble and neurotoxic compound bilirubin was not understood. Since the efficacy of biliverdin and bilirubin glucuronide transfer across the placenta is very poor, it was suggested that the formation of bilirubin from biliverdin may play a role in facilitating the elimination of heme-derived pigments in utero[27]. During the last decade, however, different studies have demonstrated the ability of bilirubin to protect cells against free radical damage both in vitro and in vivo in several tissues[242829]. A recent work carried out by our group[30] revealed that, up to a certain degree of accumulation of bilirubin (below toxic levels), this pigment may help to protect the placental-fetal unit from maternal cholestasis-induced oxidative stress. Together with its direct antioxidant properties, bilirubin is also able to induce the expression of antioxidant systems. Thus, the current concept is that, when maintained in the physiological non-toxic range, bilirubin must be considered a beneficial compound[31].
In spite of the immaturity of fetal bile secretion, small amounts of bile acids have been detected in the gallbladder bile collected from human fetuses obtained from abortions older than 12 wk of age[32]. Regarding bile pigments, although the IXβ isomer of bilirubin constitutes only a small fraction of the total amount produced in the fetus[33], this more water-soluble isomer is the most abundant isomer found in fetal gallbladder bile and meconium[3435]. The reason for this is two-fold: (1) bilirubin IXβ cannot easily cross the placenta; and (2) it can be excreted into bile without previous conjugation with glucuronic acid[36].
The fact that the expression of export pumps, such as Mrp2 and Bsep, only appears in rat fetal liver in the last third of gestation[3738] is probably the cause of the low efficiency of this route of excretion during pregnancy. As previously commented, the serum levels of bile acids and bile pigments are higher in the fetus than in the mother. It is not known how cholephilic organic anions generated by the fetal liver reach the sinusoidal blood, but because some OATPs may act as bi-directional transporters[39–41], they are good candidates for carrying out this function. Moreover, the expression of several OATP isoforms has been detected in rat fetal liver[3842]. However, the abundance of these transporters in fetal liver is much lower than in adult liver, except for Oatp4a1[43] and its human orthologue OATP4A1[44]. Another possibility is that cholephilic compounds may exit the fetal liver via ATP-dependent pumps of the ABC family located in the basolateral plasma membrane. Supporting this concept is the higher expression of Mrp1 and Mrp3 in fetal than in maternal rat liver[3842].
The accumulation of bile acids in fetal serum can have serious consequences, depending on the magnitude of the hypercholanemia[45]; in the most severe cases there is an increased risk of stillbirth and perinatal mortality[46], while in less severe conditions, maternal hypercholanemia can affect normal fetal development, the liver being one of the tissues most affected[47]. In fact, in a laboratory animal model of maternal hypercholanemia, the repercussions on fetal hepatobiliary function, although reversible, are maintained in young animals[20], and are characterized by a partial impairment in the ability of the liver to secrete organic anions, whereas the bile acid-induced biliary secretion of phospholipids, but not cholesterol, is increased[2048].
The fetal kidney is able to secrete small amounts of organic anions into the amniotic fluid[17]. This, together with the detection of ABC proteins in the apical membrane of the yolk sac, has led to the suggestion that fetal membranes provide an additional route to protect the fetus against endogenous and xenobiotic compounds[49]. However, owing to the immaturity of the fetal renal system, the importance of this route in excreting cholephilic compounds during gestation is low[50–52].
Based on the foregoing it is clear that in contrast to what happens in the adult where the hepatobiliary system with the collaboration of the kidney are responsible for the biotransformation and elimination of bile acids, biliary pigments, drugs and food components, the main route for the elimination of these compounds during intrauterine life is their transfer to the mother across the placenta. Later on, the biotransformation and elimination into feces and urine is carried out by the maternal liver and, to a lesser extent, by the maternal kidney, respectively.
As mentioned above, there is a transplacental gradient for bile acids in the fetal-to-mother direction, except for secondary and tertiary bile acids, which are more abundant in maternal serum[19]. Several experimental lines of evidence suggest that simple diffusion is not the main mechanism by which these organic anions cross the human placenta[53]. In fact, ATP-dependent mechanisms account for the vectorial transfer of these compounds in the fetus-to-mother direction[54]. This has important implications, because in situations of maternal hypercholanemia there is only a moderate increase in bile acid concentrations in fetal serum[47].
The human placenta is of the haemochorial type, i.e. only the endothelium of chorionic vessels, the stroma of chorionic villi and the trophoblast layer separate the fetal and maternal blood. This means that in order to eliminate fetal metabolic by-products across the placenta, they must cross these three components of the placental barrier. Once in the maternal blood, most foetal bile acids are eliminated in bile by the maternal liver and excreted into feces. Regarding this task, the maternal kidney only contributes slightly to the excretion of sulphated and glucuronidated species[55].
For several years there has been functional evidence for a mediated transport of cholephilic organic anions at both poles of human and rat trophoblasts[56]. Functional studies carried out on isolated human trophoblast membrane vesicles have suggested the presence of an anion exchanger transport system for the uptake of bile acids across the basal membrane of trophoblasts[53]. The trans-activation of this transport system with bicarbonate[57], the fact that substrate specificity is not restricted to bile acids[58] and the different affinities found for bile acid species depending on the number of hydroxyl groups and amidation[59] have led to the speculation that in the fetal-side membrane of the trophoblast there are proteins, probably belonging to the OATP family, that could be responsible for the uptake of organic anions by the trophoblast from fetal blood.
With respect to the opposite pole of the polarized epithelial trophoblastic cells, functional studies using plasma membrane vesicles have demonstrated that the transfer of bile acids toward the maternal circulation is dependent on ATP hydrolysis, in both human[54] and rat[60] placentas. However, it has been suggested that in the absence of ATP, bile acids could also cross this membrane by electrogenic-facilitated diffusion[61] and/or anion exchange[62]. The ATP-dependent system has higher substrate affinity, while the ATP-independent system has greater capacity[54]. These data suggest that in the apical membrane of trophoblasts, ABC proteins may be involved in pumping out bile acids towards the mother and, that proteins of the OATP family may participate in the ATP-independent component of this transport.
Concerning human OATPs, the mRNA of OATP1A2, OATP1B1 and OATP1B3 was detected in human placenta using real-time quantitative PCR[6364]. The expression levels of OATP1A2 and OATP1B1 were shown to be very low at term, although they were detected at higher levels early during gestation[65]. Both OATP2B1 and OATP4A1 were also highly expressed in human placenta[4466]. However, the former is not believed to be involved in the transport of bile acids[67], while the latter, which is considered to be a thyroid hormone carrier, is able to transport several bile acids[44].
Regarding the expression of Oatps in rat placenta, several isoforms have been detected. Under normal physiological circumstances, mRNA expression levels at term were low for Oatp1a1, Oatp1b4 and Oatp1b2, but high for Oatp4a1[42]. However, maternal cholestasis induces an up-regulation of these transporters, which is further enhanced when pregnant rats are treated with UDCA[68]. In addition, Oatp2b1 is also present in rat placenta and its ability to transport taurocholate has been described[69].
Little is known about the sub-tissue and sub-cellular localization of OATPs in the placenta. It has been suggested that OATP2B1 would be localized in the basal plasma membrane of the trophoblast[66], whereas OATP4A1 has been detected in the apical plasma membrane[44].
Regarding ABC proteins, several members of this superfamily are expressed in the placenta. The main system accounting for bile acid excretion into bile, BSEP, has been detected at very low levels in human and rat placentas at term[426470], but at higher levels during the first trimester of pregnancy in humans[65].
Some members of the MRP family with the ability to transport biliary compounds - MRP1, MPR2, MRP3 and MRP4 - have been identified in human placenta[6471]. The mRNA expression levels of MRP1 are higher in placenta than in liver; the abundance of MRP2 in the placenta is low compared to liver, and the expression levels of MRP3 and MRP4 are similar and low in both tissues, respectively[64].
The available data for the rat orthologues Mrp1, Mrp2, Mrp3 and Mrp4 suggest that they have similar expression patterns as those described for human isoforms[68]. Moreover, it has been observed that, at least in rats, there is a strong up-regulation of these transporters during maternal cholestasis, which can contribute to the protection of the fetus against the high concentrations of bile acids and biliary pigments existing on the maternal side of the placenta under these circumstances[68].
The cellular localization of some of these proteins in the placenta is controversial. MRP1, MRP2 and MRP3 have been detected by immunofluorescence and Western blotting in the apical membrane of the syncytiotrophoblast[71], and MRP1 is also expressed in fetal blood vessels[71], and in the basal membrane of the syncytiotrophoblast[72].
Another important member of the ABC family is the breast cancer resistance protein (BCRP, ABCG2), also known as ABC placental protein (ABCP) due to its high expression in this organ[73]. This protein is able to export a broad range of substrates, which have been reported to include bile acids[7]. BCRP has been detected in the apical membrane of trophoblasts and in fetal vessels[6474]. Both the mRNA and protein levels of Bcrp are higher in rodent placenta during mid-gestation but decrease at term[75].
The endothelium of chorionic vessels and the syncytiotrophoblast are exposed to high concentrations of hemoglobin through their direct contact with fetal and maternal blood, respectively. Hemoglobin and free heme can undergo auto-oxidation to produce superoxide (O2-) and H2O2, which in turn promote the formation of other highly reactive and damaging radical species. These include lipid peroxides and the very reactive hydroxyl radical if trace amounts of free iron are available[76]. Heme can be degraded either enzymatically or chemically. Both mechanisms utilize molecular oxygen (O2) and require a reducing agent. In the reaction catalyzed by heme oxygenase (HO), NADPH is the source of the reducing equivalent[77].
HO is a microsomal enzyme that induces the cleavage of heme, a pro-oxidant, to produce the biliary pigment biliverdin, iron and carbon monoxide (CO)[77]. There are three HO isoenzymes: HO-1 is a 32 kDa protein also known as heat-shock protein (HSP) 32, which is expressed at high levels in spleen and liver. HO-1 can be induced by several stimuli including hypoxia and hyperoxia[7879]. The induction of HO-1 was coupled to the synthesis of the iron-sequestering protein, ferritin[80]. Ferritin avidly binds iron and interrupts the redox cycling of iron, thereby preventing iron from being useful as a catalyst for oxidant stress[81]. Subsequent studies have demonstrated that the induction of HO-1 is also coupled to the synthesis of iron-exporting proteins and hence a critical role of HO-1 in maintaining iron homeostasis in vivo has been suggested[82].
HO-2 is a 36 kDa protein that is widely distributed in tissues throughout the body, where it is constitutively expressed and not readily inducible[83]. HO-2 appears to have an additional function as a “heme/oxygen cell sensor”, accounted for by the presence of an oxygen-sensing consensus region in the sequence of the gene[84].
As compared with the other two isoforms, HO-3 has low catalytic activity[85]. The differences between the HO isoforms also include the control of their expression, which is probably due to differences in the regulatory elements present in their promoter regions[77].
The expression of HO in human placenta has been studied extensively. The contribution of this enzyme to normal placental function is based on its sub-tissue localization, its enzymatic activity and the ability of the HO-related by-products to exert physiological effects on placental and fetal tissues[86]. Using RT-PCR, the amounts of mRNA encoding both the HO-1 and HO-2 isoforms have been measured in placental tissue. These studies demonstrated an elevated expression of HO-2 as compared to that of HO-1[87–89]. Moreover, the placental expression of both HO-1 and HO-2 increased as gestation advanced[88].
The cytoprotective properties of HO are partly due to the products of its activity, such as CO and biliary pigments. Notably, while the clinical toxicity of CO is clearly recognized, much smaller quantities of CO are remarkably cytoprotective, antiapoptotic, vasorelaxant, and anti-inflammatory[90]. As has already been mentioned, biliary pigments, long regarded to have adverse consequences in hyperbilirubinemic states, are now recognized as anti-inflammatory and antioxidant when present in low concentrations.
Biliverdin-IXα and subsequently bilirubin-IXα are the major biliary pigments in humans. However, small amounts of the other three isomers are also generated depending on the position of the protoporphyrin IX that is cleaved. These include biliverdin-IXγ, biliverdin-IXδ and biliverdin-IXβ, which is the most abundant of these three pigments in humans and other mammals[35].
Biliverdin, which is produced by HO-1 and HO-2 activity, is further biotransformed to bilirubin by biliverdin reductase: mainly biliverdin-IXα reductase (BVRα). This enzyme also functions as a kinase and as a transcription factor in the MAPK signalling cascade[91]. BVRα is expressed in many tissues[92], including the placenta[27]. Bilirubin is conjugated in the liver with glucuronic acid by bilirubin uridine diphosphate-glucuronosyl transferase-1A1 (UGT1A1)[93] prior to being secreted into bile. Owing to the immaturity of the fetal liver, no hepatobiliary elimination of bilirubin occurs, at least at a physiologically relevant rate.
Unconjugated bilirubin concentrations are higher in fetal than in maternal serum[1926]. Several factors contribute to the existence of this gradient. In the fetus, there is a very active heme catabolism, and hence a high rate of bilirubin production, together with a very low expression of bilirubin uridine diphosphate-glucuronosyl transferase in the liver[94]. Moreover, simple diffusion is not the major route for the placental transfer of biliary pigments[56].
In the presence of reactive oxygen species, bilirubin is oxidized to biliverdin and then converted back into bilirubin by BVRα[95]. Thus the biliverdin-bilirubin tandem acts as an efficient scavenger of reactive oxygen species and inhibits lipid oxidation both in vitro and in vivo[9697]. Bilirubin is also an effective antioxidant of peroxynitrite-mediated protein oxidation and inhibits the production of superoxide by blocking the activation of NADPH oxidase[9899]. Sub-toxic bilirubin concentrations have direct anti-oxidant properties and indirect beneficial effects against cholestasis-induced toxicity during pregnancy, such as the enhanced expression of several elements of the anti-oxidant defence system, i.e. BVRα, SVCT1 and SVCT2, as well as several nuclear receptors sensitive to activation by biliary compounds[100]. This function is mainly dependent on the expression of BVRα, which has been found to be moderately up-regulated in the maternal liver-placenta-fetal liver trio in pregnant rats with surgically induced obstructive cholestasis during the last week of gestation[100]. However, beneficial antioxidant properties are limited to low bilirubin concentrations because at higher levels this pigment can also cause irreversible damage or even death when it is accumulated in the nervous system[101].
It has been suggested that the reduction of biliverdin to bilirubin could have the evolutionary advantage of facilitating the placental excretion of bile pigments by simple diffusion[102]. However, in vitro[103] and in vivo[104] studies have suggested that under normal physiological circumstances the major pathway for bilirubin placental transfer involves carrier-mediated transport across both poles of the plasma membrane of the human trophoblast[103]. Moreover, at least in rodents, bilirubin does not undergo any major biotransformation during its residence in the placenta[104]. The existence of vectorial properties for transplacental bilirubin transfer are consistent with the moderate increases in serum bilirubin concentrations observed in the fetuses of pregnant rats with marked hyperbilirubinemia due to common bile duct ligation[47]. The mechanism for the placental uptake of fetal biliary pigments is not completely understood. Proteins of the OATP family, in particular human OATP1B1 and OATP1B3, have been reported to confer the ability to take up unconjugated bilirubin when expressed in Xenopus laevis oocytes[63]. However, the mRNA of OATP1B1 is almost absent in isolated human trophoblast cells, whereas OATP1B3 is clearly expressed in this epithelium, although at low levels[63].
Inside trophoblast cells, bilirubin is probably partly bound to lipids and proteins such as glutathione-S-transferase[105]. Functional studies have suggested that bilirubin might be exported across the apical pole of the trophoblast via an ATP-dependent mechanism[103]. Whether one or several isoforms of MRPs expressed in human[71] and rat[4268] placenta are involved in this process is not known.
MRP2, and probably MRP1, are also able to perform ATP-dependent transport of bilirubin glucuronides[5]. However, owing to the low UDP-glucuronosyl transferase activity of the fetal liver and the absence of placental biotransformation of unconjugated bilirubin during transplacental transfer[106], MRP2 and MRP1 are not expected to play an important role in bilirubin transfer across the placenta.
It has been shown that biliverdin itself is poorly transferred-without prior reduction to bilirubin - across the guinea pig[26] and rat[106] placenta. However, biliverdin is able to inhibit bilirubin transfer in rat placenta when co-administered through the umbilical artery of in situ perfused rat placentas[106]. The transport of biliverdin from the trophoblast toward the mother is very poor and/or that placental biotransformation of biliverdin into bilirubin is very efficient. Part of the endogenous biliverdin produced by the fetus could be transformed into bilirubin by the fetal liver prior to being taken up by the placenta, because the expression of BVRα in fetal liver is even higher than in rat placenta[100].
Among the transporters involved in fetal biliverdin uptake by rat placenta, several OATPs, in particular Oatp1a1, may be involved[106]. Once in the placenta, and prior to being transferred to the mother, biliverdin is extensively converted into bilirubin by BVRα, which is highly expressed in this organ[106]. The small amount of biliverdin that reaches the maternal blood is efficiently taken up, probably in part by Oatp1a1, Oatp1a4 and Oatp1b2, and biotransformed into bilirubin, which joins the fetal bilirubin transferred by the placenta, to be eliminated mainly through secretion into the bile by the maternal liver[106].
Fetal exposure to foreign molecules is partly dependent on the maternal capacity to eliminate such compounds and on the ability of the xenobiotics to cross the placenta. One important characteristic of the placenta is that this organ undergoes continuous development. This implies the existence of changes that must be compatible with the maintenance of a partially permeable epithelial barrier required to provide protection against exposure to potentially harmful substances present in the maternal blood[56107108]. Therefore, before any pharmacological interventions, the different stages of pregnancy should be considered, because these will determine both the permeability of the placental barrier and the vulnerability of the conceptus to xenobiotics[109].
Although most drugs administered during pregnancy may cross the placenta to some extent, the magnitude of this depends on the size and structure of the molecule. Diffusional transfer across the placenta for drugs with a molecular weight higher than 500 Da is usually very restricted[110]. Liposolubility and ionization are strong determinants for drug diffusion across the placenta. For instance, several penicillins, in spite of being strong acids, can be efficiently transferred across the human placenta, probably by simple diffusion[111]. Among weak-base drugs, acetaminophen, phenobarbital, phenytoin and clonidine are able to cross the placenta at a high rate, probably by simple diffusion[112]. Nucleoside analogue reverse transcriptase inhibitors (NRTIs) are molecules with low molecular weight and low protein binding, so most of them are also able to cross the placenta by simple diffusion and are concentrated in the amniotic fluid[113].
Although most transporters localized at the plasma membrane of cells and forming part of the placental barrier have specific physiological substrates, some of them also transport structurally similar compounds. In some cases, however, there are no known physiological substrates and only certain xenobiotics have been reported to be transported by them. Moreover, some of the xenobiotics able to cross the placental barrier may have the ability to affect gene expression. This may result in a decrease in the expression of placental transporters, which may affect their ability to accomplish their physiological roles and eventually lead to an enhanced entry of drugs into placental tissue[114].
The placenta expresses some isoforms of monocarboxylate transporters (MCTs)[115]. The primary substrate of MCTs in placenta is lactate, although pyruvate and β-hydroxybutyrate are also transported. Placental MCTs exert a significant influence on the transfer across the maternal-fetal interface of drugs such as valproate, benzoate, salicylates, statins, nateglinide, and foscarnet[114115].
Equilibrative nucleoside transporters (ENTs) are widely distributed and have broad substrate specificity. There is evidence of the presence of two ENT isoforms in the human placenta: ENT1 (SLC29A1) and ENT2 (SLC29A2)[116117]. Moreover, concentrative nucleoside transporters (CNTs), CNT2 (SLC28A2) and CNT3 (SLC28A3) are also expressed in human placenta. Both ENT1 and ENT2 are able to transport a wide variety of therapeutic agents such as the anticancer drugs cytarabine and gemcitabine and the antiviral drugs zalcitabine (ddC) and zidovudine[117118], and they therefore probably play a role in fetal exposure to these types of drugs.
Regarding amino acids and monoamines, 17 mammalian transport systems for amino acids have been functionally identified in the human placenta[119120]. The interaction of xenobiotics with amino acid transport systems in the syncytiotrophoblast may result in a deficit in the transport of amino acids across the placenta. This seems to be the case for cocaine, which readily crosses the placental barrier and enters the fetal circulation. This constitutes a potential cause of adverse effects on the developing fetus in pregnant women consuming this drug[121]. Maternal smoking during pregnancy also decreases the ability of the placenta to efficiently take up amino acids and hence affects the overall transfer of these important metabolites from the maternal to the fetal circulation[121].
Additionally, cocaine may also interact with other placental carriers, such as those involved in monoamine transport[121]. This may affect serotonin and noradrenaline transport across the apical (maternal-facing) plasma membrane of the trophoblast[122]. Moreover, antidepressants (fluoxetine, paroxetine, sertraline, citalopram, and desipramine) as well as cocaine are inhibitors of monoamine transporters but are not transportable substrates. In contrast, amphetamines are transportable substrates for monoamine transporters, thereby gaining access into the placenta and fetus[122123].
Many xenobiotics are substrates of OATP isoforms[124], which are expressed in the human placenta[6366]. These transporters have partially different and overlapping substrate preferences for a wide range of exogenous organic solutes, including gadodexate, ouabain, iloprost, Gd-B 20790, methotrexate, rifampicin, the endothelin receptor antagonist BQ-123, the thrombin inhibitor CRC-220, the opioid receptor agonists D-penicillamine-(2,5)-enkephalin (DPDPE) and deltorphin II, the angiotensin-converting enzyme inhibitors enalapril and temocaprilat, the HMG-CoA reductase inhibitor pravastatin, and the antihistamine fexofenadine, in addition to several cytostatic derivatives obtained by coupling bile acid moieties to chlorambucil or cisplatin[125–127]. Some OATP isoforms have also been shown to transport bulky organic cations[124128]. This suggests that isoforms detected in placenta could serve as a route for the transfer of anions and relatively hydrophilic cationic organic drugs.
Organic cations can be transferred across the placenta using a different route. At least one member of the subfamily of carriers for organic cations (OCTs), namely OCT3, is very abundantly expressed in the human placenta[129130]. Examples of OCT3 substrates include cimetidine, MPP+, agmatine, tetraethylammonium, and prazosin[131]. The sodium-dependent carnitine transporter (OCTN2) also belongs to the SLC22A family and is expressed in human placenta[132]. OCTN2 transports a variety of organic cations including tetraethylammonium, nicotine, MPP+, pyrilamine, cimetidine, clonidine, procainamide, quinidine, quinine, and verapamil[133] and certain β-lactam antibiotics of zwitterionic nature[134].
During the first trimester of pregnancy, a broader variety of xenobiotic-metabolizing enzymes are expressed in the placenta as compared to at term[135136]. However, placental expression of phaseIand II metabolizing enzymes is moderate and probably more closely involved in the endocrine functions of this organ than in the metabolism of xenobiotics[135]. The placenta expresses several cytochrome P450 enzymes (CYPs) at mRNA levels that increase throughout pregnancy. Although placental CYPs are capable of metabolizing several xenobiotic compounds at term[135136], only a few of these enzymes are actually functionally active[137138]. Moreover, the abundance of some of these CYPs has been shown to be affected by exposure to xenobiotics, as occurs in tobacco-smoking pregnant women[138139]. Other phaseImetabolizing enzymes such as aldehyde dehydrogenases (ALDHs) participate in the detoxification of endogenous and exogenous compounds, including ethanol. The presence of this activity in human placenta may be relevant in the toxicity of a number of substances and for the gestational consequences of alcohol consumption[140].
Among phase II enzymes, glutathione-S-transferases, epoxide hydrolase, N-acetyltransferases, sulfotransferases, and UDP-glucuronosyl transferases are expressed at moderate levels in the placenta and have been shown to be involved in the detoxification of several xenobiotics[141]. In contrast, drug- and toxin-induced up-regulation of biotransforming enzymes can lead to an enhanced production of reactive metabolites able to interact with DNA, resulting in the formation of DNA adducts[142]. This may challenge the normal development of the conceptus. Indeed the levels of smoking-related adducts in the placenta have been inversely correlated with offspring birth weight[143].
ATP-dependent efflux transporters expressed in the apical membrane of placental syncytiotrophoblasts are very important in limiting the magnitude of drug penetration across the placental barrier, hence reducing fetal drug exposure. The superfamily of ABC proteins includes a large number of members with the ability to translocate a broad variety of substrates across extra- and intra-cellular membranes. These proteins are involved in many physiological processes, such as sterol homeostasis, immune mechanisms, and the transport of endogenous and xenobiotic substances such as sugars, amino acids, metal ions, peptides and proteins, and a large number of hydrophobic compounds and metabolites. Several members of three families of ABC transporters, ABCB, ABCC and ABCG, known to be involved in multidrug resistance are major candidates for involvement in the placental barrier for drugs[144145].
The first ABC transporter recognized to play a significant role in the placental barrier was MDR1[145]. MDR1 is abundantly expressed during pregnancy, and in particular in the syncytiotrophoblast[146]. The substrates of MDR1 are usually organic molecules ranging in size from about 200 Da to almost 1900 Da. Most of them are uncharged or weakly basic in nature, but some acidic compounds can also be transported. As a consequence, a large number of drugs from several pharmacotherapeutic groups are recognized as MDR1 substrates. Thus, placental MDR1 may contribute to the protection of the foetus from a wide variety of drugs, including antivirals and anticancer agents[147].
Other major efflux transporters involved in the protection of the developing fetus from exposure to these drugs are members of the MRP subfamily, involved in the transport of conjugates of several drugs and endogenous compounds, have been found in the human placenta. MRP2 is expressed in the syncytiotrophoblast, whereas MRP1 and MRP3 are expressed both in blood vessel endothelia and in the syncytiotrophoblast[71], and MRP5 is expressed in the basal membrane of syncytiotrophoblasts and around fetal vessels[148], where aside from its potential role in drug disposition this transporter may mediate the cellular efflux of 3′,5′-cyclic nucleotides, cAMP, and cGMP, thus playing an important role in paracrine signal transduction.
BCRP expression in the placenta is possibly tightly controlled during pregnancy by pregnancy-related steroid hormones, growth factors, and cytokines[149]. BCRP transports a broad variety of conjugated or non-conjugated organic anions, but from a physiological point of view it is probably involved in the elimination of endogenous sulphate conjugates[150]. Substantial variations in BCRP expression have been observed in human placenta[151], suggesting that considerable variability could exist in the ability of the placenta to protect the fetus from exposure to drugs, xenobiotics and metabolites. Such variable expression and/or activity has been suggested to be due to genetic polymorphisms in the BCRP gene[151].
The immaturity of the fetal hepatobiliary system precludes the use of this mechanism of defence against endogenous and xenobiotic compounds during intrauterine life. Consequently, this function is carried out by a complex and efficient combined action of the placenta and the maternal liver. However, when one of these two members of the defensive tandem is impaired the overall function may be compromised, resulting in deleterious effects in the fetus. A better understanding of the molecular mechanisms involved in hepatobiliary excretory function during intrauterine life is needed to recognize the danger the fetus may face, to develop novel pharmacological tools to manipulate the placental transfer of xenobiotics, and to generate new drugs with enhanced or reduced ability to cross the placental barrier.
| 1. | Meier PJ, Stieger B. Bile salt transporters. Annu Rev Physiol. 2002;64:635-661. |
| 2. | Hagenbuch B, Stieger B, Foguet M, Lubbert H, Meier PJ. Functional expression cloning and characterization of the hepatocyte Na+/bile acid cotransport system. Proc Natl Acad Sci USA. 1991;88:10629-10633. |
| 3. | Muller M, Mayer R, Hero U, Keppler D. ATP-dependent transport of amphiphilic cations across the hepatocyte canalicular membrane mediated by mdr1 P-glycoprotein. FEBS Lett. 1994;343:168-172. |
| 4. | Gerloff T, Stieger B, Hagenbuch B, Madon J, Landmann L, Roth J, Hofmann AF, Meier PJ. The sister of P-glycoprotein represents the canalicular bile salt export pump of mammalian liver. J Biol Chem. 1998;273:10046-10050. |
| 5. | Jedlitschky G, Leier I, Buchholz U, Hummel-Eisenbeiss J, Burchell B, Keppler D. ATP-dependent transport of bilirubin glucuronides by the multidrug resistance protein MRP1 and its hepatocyte canalicular isoform MRP2. Biochem J. 1997;327:305-310. |
| 6. | Konig J, Nies AT, Cui Y, Leier I, Keppler D. Conjugate export pumps of the multidrug resistance protein (MRP) family: localization, substrate specificity, and MRP2-mediated drug resistance. Biochim Biophys Acta. 1999;1461:377-394. |
| 7. | Janvilisri T, Shahi S, Venter H, Balakrishnan L, van Veen HW. Arginine-482 is not essential for transport of antibiotics, primary bile acids and unconjugated sterols by the human breast cancer resistance protein (ABCG2). Biochem J. 2005;385:419-426. |
| 8. | Roelofsen H, Muller M, Jansen PL. Regulation of organic anion transport in the liver. Yale J Biol Med. 1997;70:435-445. |
| 9. | Ogawa K, Suzuki H, Hirohashi T, Ishikawa T, Meier PJ, Hirose K, Akizawa T, Yoshioka M, Sugiyama Y. Characterization of inducible nature of MRP3 in rat liver. Am J Physiol Gastrointest Liver Physiol. 2000;278:G438-G446. |
| 10. | Soroka CJ, Lee JM, Azzaroli F, Boyer JL. Cellular localization and up-regulation of multidrug resistance-associated protein 3 in hepatocytes and cholangiocytes during obstructive cholestasis in rat liver. Hepatology. 2001;33:783-791. |
| 11. | Donner MG, Keppler D. Up-regulation of basolateral multidrug resistance protein 3 (Mrp3) in cholestatic rat liver. Hepatology. 2001;34:351-359. |
| 12. | Vos TA, Hooiveld GJ, Koning H, Childs S, Meijer DK, Moshage H, Jansen PL, Muller M. Up-regulation of the multidrug resistance genes, Mrp1 and Mdr1b, and down-regulation of the organic anion transporter, Mrp2, and the bile salt transporter, Spgp, in endotoxemic rat liver. Hepatology. 1998;28:1637-1644. |
| 13. | Tanaka Y, Kobayashi Y, Gabazza EC, Higuchi K, Kamisako T, Kuroda M, Takeuchi K, Iwasa M, Kaito M, Adachi Y. Increased renal expression of bilirubin glucuronide transporters in a rat model of obstructive jaundice. Am J Physiol Gastrointest Liver Physiol. 2002;282:G656-G662. |
| 14. | Marin JJ, Macias RI, Briz O, Perez MJ, Blazquez AG, Arrese M, Serrano MA. Molecular bases of the fetal liver-placenta-maternal liver excretory pathway for cholephilic compounds. Liver Int. 2008;28:435-454. |
| 15. | Arrese M, Macias RI, Briz O, Perez MJ, Marin JJ. Molecular pathogenesis of intrahepatic cholestasis of pregnancy. Expert Rev Mol Med. 2008;10:e9. |
| 16. | Colombo C, Roda A, Roda E, Buscaglia M, dell’Agnola CA, Filippetti P, Ronchi M, Sereni F. Correlation between fetal and maternal serum bile acid concentrations. Pediatr Res. 1985;19:227-231. |
| 17. | Nakagawa M, Setchell KD. Bile acid metabolism in early life: studies of amniotic fluid. J Lipid Res. 1990;31:1089-1098. |
| 18. | Setchell KD, Dumaswala R, Colombo C, Ronchi M. Hepatic bile acid metabolism during early development revealed from the analysis of human fetal gallbladder bile. J Biol Chem. 1988;263:16637-16644. |
| 19. | Monte MJ, Rodriguez-Bravo T, Macias RI, Bravo P, el-Mir MY, Serrano MA, Lopez-Salva A, Marin JJ. Relationship between bile acid transplacental gradients and transport across the fetal-facing plasma membrane of the human trophoblast. Pediatr Res. 1995;38:156-163. |
| 20. | Monte MJ, Morales AI, Arevalo M, Alvaro I, Macias RI, Marin JJ. Reversible impairment of neonatal hepatobiliary function by maternal cholestasis. Hepatology. 1996;23:1208-1217. |
| 21. | Balistreri WF, A-Kader HH, Setchell KD, Gremse D, Ryckman FC, Schroeder TJ. New methods for assessing liver function in infants and children. Ann Clin Lab Sci. 1992;22:162-174. |
| 22. | Massimi M, Lear SR, Huling SL, Jones AL, Erickson SK. Cholesterol 7alpha-hydroxylase (CYP7A): patterns of messenger RNA expression during rat liver development. Hepatology. 1998;28:1064-1072. |
| 23. | Keitel V, Kubitz R, Haussinger D. Endocrine and paracrine role of bile acids. World J Gastroenterol. 2008;14:5620-5629. |
| 24. | Galbraith R. Heme oxygenase: who needs it? Proc Soc Exp Biol Med. 1999;222:299-305. |
| 25. | Ryter SW, Tyrrell RM. The heme synthesis and degradation pathways: role in oxidant sensitivity. Heme oxygenase has both pro- and antioxidant properties. Free Radic Biol Med. 2000;28:289-309. |
| 26. | Knudsen A, Lebech M. Maternal bilirubin, cord bilirubin, and placenta function at delivery and the development of jaundice in mature newborns. Acta Obstet Gynecol Scand. 1989;68:719-724. |
| 27. | McDonagh AF, Palma LA, Schmid R. Reduction of biliverdin and placental transfer of bilirubin and biliverdin in the pregnant guinea pig. Biochem J. 1981;194:273-282. |
| 28. | Dore S, Snyder SH. Neuroprotective action of bilirubin against oxidative stress in primary hippocampal cultures. Ann N Y Acad Sci. 1999;890:167-172. |
| 29. | Dore S, Sampei K, Goto S, Alkayed NJ, Guastella D, Blackshaw S, Gallagher M, Traystman RJ, Hurn PD, Koehler RC. Heme oxygenase-2 is neuroprotective in cerebral ischemia. Mol Med. 1999;5:656-663. |
| 30. | Perez MJ, Castano B, Jimenez S, Serrano MA, Gonzalez-Buitrago JM, Marin JJ. Role of vitamin C transporters and biliverdin reductase in the dual pro-oxidant and anti-oxidant effect of biliary compounds on the placental-fetal unit in cholestasis during pregnancy. Toxicol Appl Pharmacol. 2008;232:327-336. |
| 31. | Sedlak TW, Snyder SH. Bilirubin benefits: cellular protection by a biliverdin reductase antioxidant cycle. Pediatrics. 2004;113:1776-1782. |
| 32. | Colombo C, Zuliani G, Ronchi M, Breidenstein J, Setchell KD. Biliary bile acid composition of the human fetus in early gestation. Pediatr Res. 1987;21:197-200. |
| 33. | McDonagh AF. Turning green to gold. Nat Struct Biol. 2001;8:198-200. |
| 34. | Aziz S, Kotal P, Leroy P, Servaes R, Eggermont E, Fevery J. Bilirubin-IXalpha and -IXbeta pigments, coproporphyrins and bile acids in meconium and stools from full-term and preterm neonates during the first month of life. Acta Paediatr. 2001;90:81-87. |
| 35. | Yamaguchi T, Nakajima H. Changes in the composition of bilirubin-IX isomers during human prenatal development. Eur J Biochem. 1995;233:467-472. |
| 36. | Blanckaert N, Heirwegh KP, Zaman Z. Comparison of the biliary excretion of the four isomers of bilirubin-IX in Wistar and homozygous Gunn rats. Biochem J. 1977;164:229-236. |
| 37. | Zinchuk VS, Okada T, Akimaru K, Seguchi H. Asynchronous expression and colocalization of Bsep and Mrp2 during development of rat liver. Am J Physiol Gastrointest Liver Physiol. 2002;282:G540-G548. |
| 38. | Macias RI, Jimenez S, Serrano MA, Monte MJ, Marin JJ. Effect of maternal cholestasis and treatment with ursodeoxycholic acid on the expression of genes involved in the secretion of biliary lipids by the neonatal rat liver. Life Sci. 2006;79:1014-1019. |
| 39. | Li L, Meier PJ, Ballatori N. Oatp2 mediates bidirectional organic solute transport: a role for intracellular glutathione. Mol Pharmacol. 2000;58:335-340. |
| 40. | Briz O, Romero MR, Martinez-Becerra P, Macias RI, Perez MJ, Jimenez F, San Martin FG, Marin JJ. OATP8/1B3-mediated cotransport of bile acids and glutathione: an export pathway for organic anions from hepatocytes? J Biol Chem. 2006;281:30326-30335. |
| 41. | Mahagita C, Grassl SM, Piyachaturawat P, Ballatori N. Human organic anion transporter 1B1 and 1B3 function as bidirectional carriers and do not mediate GSH-bile acid cotransport. Am J Physiol Gastrointest Liver Physiol. 2007;293:G271-G278. |
| 42. | St-Pierre MV, Stallmach T, Freimoser Grundschober A, Dufour JF, Serrano MA, Marin JJ, Sugiyama Y, Meier PJ. Temporal expression profiles of organic anion transport proteins in placenta and fetal liver of the rat. Am J Physiol Regul Integr Comp Physiol. 2004;287:R1505-R1516. |
| 43. | Fujiwara K, Adachi H, Nishio T, Unno M, Tokui T, Okabe M, Onogawa T, Suzuki T, Asano N, Tanemoto M. Identification of thyroid hormone transporters in humans: different molecules are involved in a tissue-specific manner. Endocrinology. 2001;142:2005-2012. |
| 44. | Sato K, Sugawara J, Sato T, Mizutamari H, Suzuki T, Ito A, Mikkaichi T, Onogawa T, Tanemoto M, Unno M. Expression of organic anion transporting polypeptide E (OATP-E) in human placenta. Placenta. 2003;24:144-148. |
| 45. | Glantz A, Marschall HU, Mattsson LA. Intrahepatic cholestasis of pregnancy: Relationships between bile acid levels and fetal complication rates. Hepatology. 2004;40:467-474. |
| 46. | Lammert F, Marschall HU, Matern S. Intrahepatic Cholestasis of Pregnancy. Curr Treat Options Gastroenterol. 2003;6:123-132. |
| 47. | Macias RI, Pascual MJ, Bravo A, Alcalde MP, Larena MG, St-Pierre MV, Serrano MA, Marin JJ. Effect of maternal cholestasis on bile acid transfer across the rat placenta-maternal liver tandem. Hepatology. 2000;31:975-983. |
| 48. | El-Mir MY, Monte MJ, Morales AI, Arevalo M, Serrano MA, Marin JJ. Effect of maternal cholestasis on biliary lipid and bile acid secretion in the infant rat. Hepatology. 1997;26:527-536. |
| 49. | Aleksunes LM, Cui Y, Klaassen CD. Prominent expression of xenobiotic efflux transporters in mouse extraembryonic fetal membranes compared with placenta. Drug Metab Dispos. 2008;36:1960-1970. |
| 50. | Jackson BT, Smallwood RA, Piasecki GJ, Brown AS, Rauschecker HF, Lester R. Fetal bile salt metabolism. I. The metabolism of sodium cholate-14C in the fetal dog. J Clin Invest. 1971;50:1286-1294. |
| 51. | Little JM, Smallwood RA, Lester R, Piasecki GJ, Jackson BT. Bile-salt metabolism in the primate fetus. Gastroenterology. 1975;69:1315-1320. |
| 52. | Watkins JB. Placental transport: bile acid conjugation and sulfation in the fetus. J Pediatr Gastroenterol Nutr. 1983;2:365-373. |
| 53. | Marin JJ, Serrano MA, el-Mir MY, Eleno N, Boyd CA. Bile acid transport by basal membrane vesicles of human term placental trophoblast. Gastroenterology. 1990;99:1431-1438. |
| 54. | Marin JJ, Bravo P, el-Mir MY, Serrano MA. ATP-dependent bile acid transport across microvillous membrane of human term trophoblast. Am J Physiol. 1995;268:G685-G694. |
| 55. | Frohling W, Stiehl A. Bile salt glucuronides: identification and quantitative analysis in the urine of patients with cholestasis. Eur J Clin Invest. 1976;6:67-74. |
| 56. | Marin JJ, Macias RI, Serrano MA. The hepatobiliary-like excretory function of the placenta. A review. Placenta. 2003;24:431-438. |
| 57. | el-Mir MY, Eleno N, Serrano MA, Bravo P, Marin JJ. Bicarbonate-induced activation of taurocholate transport across the basal plasma membrane of human term trophoblast. Am J Physiol. 1991;260:G887-G894. |
| 58. | Bravo P, el-Mir MY, Serrano MA, Boyd R, Marin JJ. Interaction between cholephilic anions and bile acid transport across basal membrane of human trophoblast. Am J Physiol. 1993;265:G242-G250. |
| 59. | Serrano MA, Bravo P, el-Mir MY, Marin JJ. Influence of hydroxylation and conjugation in cross-inhibition of bile acid transport across the human trophoblast basal membrane. Biochim Biophys Acta. 1993;1151:28-34. |
| 60. | Bravo P, Marin JJ, Beveridge MJ, Novak DA. Reconstitution and characterization of ATP-dependent bile acid transport in human and rat placenta. Biochem J. 1995;311:479-485. |
| 61. | Iioka H, Hisanaga H, Akada S, Shimamoto T, Yamada Y, Sakamoto Y, Moriyama IS, Ichijo M. Characterization of human placental activity for transport of taurocholate, using brush border (microvillous) membrane vesicles. Placenta. 1993;14:93-102. |
| 62. | Dumaswala R, Setchell KD, Moyer MS, Suchy FJ. An anion exchanger mediates bile acid transport across the placental microvillous membrane. Am J Physiol. 1993;264:G1016-G1023. |
| 63. | Briz O, Serrano MA, MacIas RI, Gonzalez-Gallego J, Marin JJ. Role of organic anion-transporting polypeptides, OATP-A, OATP-C and OATP-8, in the human placenta-maternal liver tandem excretory pathway for foetal bilirubin. Biochem J. 2003;371:897-905. |
| 64. | Serrano MA, Macias RI, Briz O, Monte MJ, Blazquez AG, Williamson C, Kubitz R, Marin JJ. Expression in human trophoblast and choriocarcinoma cell lines, BeWo, Jeg-3 and JAr of genes involved in the hepatobiliary-like excretory function of the placenta. Placenta. 2007;28:107-117. |
| 65. | Patel P, Weerasekera N, Hitchins M, Boyd CA, Johnston DG, Williamson C. Semi quantitative expression analysis of MDR3, FIC1, BSEP, OATP-A, OATP-C,OATP-D, OATP-E and NTCP gene transcripts in 1st and 3rd trimester human placenta. Placenta. 2003;24:39-44. |
| 66. | St-Pierre MV, Hagenbuch B, Ugele B, Meier PJ, Stallmach T. Characterization of an organic anion-transporting polypeptide (OATP-B) in human placenta. J Clin Endocrinol Metab. 2002;87:1856-1863. |
| 67. | Kullak-Ublick GA, Ismair MG, Stieger B, Landmann L, Huber R, Pizzagalli F, Fattinger K, Meier PJ, Hagenbuch B. Organic anion-transporting polypeptide B (OATP-B) and its functional comparison with three other OATPs of human liver. Gastroenterology. 2001;120:525-533. |
| 68. | Serrano MA, Macias RI, Vallejo M, Briz O, Bravo A, Pascual MJ, St-Pierre MV, Stieger B, Meier PJ, Marin JJ. Effect of ursodeoxycholic acid on the impairment induced by maternal cholestasis in the rat placenta-maternal liver tandem excretory pathway. J Pharmacol Exp Ther. 2003;305:515-524. |
| 69. | Nishio T, Adachi H, Nakagomi R, Tokui T, Sato E, Tanemoto M, Fujiwara K, Okabe M, Onogawa T, Suzuki T. Molecular identification of a rat novel organic anion transporter moat1, which transports prostaglandin D(2), leukotriene C(4), and taurocholate. Biochem Biophys Res Commun. 2000;275:831-838. |
| 70. | St-Pierre MV, Serrano MA, Lauper U, Stieger B, Marin JJG, Meier-Abt PJ. Identification of bile salt transporters in rat and human placenta. J Hepatol. 30:141-141. |
| 71. | St-Pierre MV, Serrano MA, Macias RI, Dubs U, Hoechli M, Lauper U, Meier PJ, Marin JJ. Expression of members of the multidrug resistance protein family in human term placenta. Am J Physiol Regul Integr Comp Physiol. 2000;279:R1495-R1503. |
| 72. | Nagashige M, Ushigome F, Koyabu N, Hirata K, Kawabuchi M, Hirakawa T, Satoh S, Tsukimori K, Nakano H, Uchiumi T. Basal membrane localization of MRP1 in human placental trophoblast. Placenta. 2003;24:951-958. |
| 73. | Allikmets R, Schriml LM, Hutchinson A, Romano-Spica V, Dean M. A human placenta-specific ATP-binding cassette gene (ABCP) on chromosome 4q22 that is involved in multidrug resistance. Cancer Res. 1998;58:5337-5339. |
| 74. | Ceckova M, Libra A, Pavek P, Nachtigal P, Brabec M, Fuchs R, Staud F. Expression and functional activity of breast cancer resistance protein (BCRP, ABCG2) transporter in the human choriocarcinoma cell line BeWo. Clin Exp Pharmacol Physiol. 2006;33:58-65. |
| 75. | Kalabis GM, Petropoulos S, Gibb W, Matthews SG. Breast cancer resistance protein (Bcrp1/Abcg2) in mouse placenta and yolk sac: ontogeny and its regulation by progesterone. Placenta. 2007;28:1073-1081. |
| 76. | Mancuso C, Preziosi P, Grossman AB, Navarra P. The role of carbon monoxide in the regulation of neuroendocrine function. Neuroimmunomodulation. 1997;4:225-229. |
| 77. | Maines MD. The heme oxygenase system: a regulator of second messenger gases. Annu Rev Pharmacol Toxicol. 1997;37:517-554. |
| 78. | Keyse SM, Tyrrell RM. Heme oxygenase is the major 32-kDa stress protein induced in human skin fibroblasts by UVA radiation, hydrogen peroxide, and sodium arsenite. Proc Natl Acad Sci USA. 1989;86:99-103. |
| 79. | Maines MD, Mayer RD, Ewing JF, McCoubrey WK Jr. Induction of kidney heme oxygenase-1 (HSP32) mRNA and protein by ischemia/reperfusion: possible role of heme as both promotor of tissue damage and regulator of HSP32. J Pharmacol Exp Ther. 1993;264:457-462. |
| 80. | Nath KA, Balla G, Vercellotti GM, Balla J, Jacob HS, Levitt MD, Rosenberg ME. Induction of heme oxygenase is a rapid, protective response in rhabdomyolysis in the rat. J Clin Invest. 1992;90:267-270. |
| 81. | Balla G, Jacob HS, Balla J, Rosenberg M, Nath K, Apple F, Eaton JW, Vercellotti GM. Ferritin: a cytoprotective antioxidant strategem of endothelium. J Biol Chem. 1992;267:18148-18153. |
| 82. | Baranano DE, Wolosker H, Bae BI, Barrow RK, Snyder SH, Ferris CD. A mammalian iron ATPase induced by iron. J Biol Chem. 2000;275:15166-15173. |
| 83. | Maines MD. Heme oxygenase: function, multiplicity, regulatory mechanisms, and clinical applications. FASEB J. 1988;2:2557-2568. |
| 84. | Lee-Huang S, Lin JJ, Kung HF, Huan PL, Lee L, Huang PL. The 3’ flanking region of the human erythropoietin-encoding gene contains nitrogen-regulatory/oxygen-sensing consensus sequences and tissue-specific transcriptional regulatory elements. Gene. 1993;137:203-210. |
| 85. | McCoubrey WK Jr, Huang TJ, Maines MD. Isolation and characterization of a cDNA from the rat brain that encodes hemoprotein heme oxygenase-3. Eur J Biochem. 1997;247:725-732. |
| 86. | Bainbridge SA, Smith GN. HO in pregnancy. Free Radic Biol Med. 2005;38:979-988. |
| 87. | McLean M, Bowman M, Clifton V, Smith R, Grossman AB. Expression of the heme oxygenase-carbon monoxide signalling system in human placenta. J Clin Endocrinol Metab. 2000;85:2345-2349. |
| 88. | Yoshiki N, Kubota T, Aso T. Expression and localization of heme oxygenase in human placental villi. Biochem Biophys Res Commun. 2000;276:1136-1142. |
| 89. | McLaughlin BE, Lash GE, Smith GN, Marks GS, Nakatsu K, Graham CH, Brien JF. Heme oxygenase expression in selected regions of term human placenta. Exp Biol Med (Maywood). 2003;228:564-567. |
| 91. | Tudor C, Lerner-Marmarosh N, Engelborghs Y, Gibbs PE, Maines MD. Biliverdin reductase is a transporter of haem into the nucleus and is essential for regulation of HO-1 gene expression by haematin. Biochem J. 2008;413:405-416. |
| 92. | McCoubrey WK Jr, Cooklis MA, Maines MD. The structure, organization and differential expression of the rat gene encoding biliverdin reductase. Gene. 1995;160:235-240. |
| 93. | Bosma PJ, Seppen J, Goldhoorn B, Bakker C, Oude Elferink RP, Chowdhury JR, Chowdhury NR, Jansen PL. Bilirubin UDP-glucuronosyltransferase 1 is the only relevant bilirubin glucuronidating isoform in man. J Biol Chem. 1994;269:17960-17964. |
| 94. | Kawade N, Onishi S. The prenatal and postnatal develo-pment of UDP-glucuronyltransferase activity towards bilirubin and the effect of premature birth on this activity in the human liver. Biochem J. 1981;196:257-260. |
| 95. | Baranano DE, Rao M, Ferris CD, Snyder SH. Biliverdin reductase: a major physiologic cytoprotectant. Proc Natl Acad Sci USA. 2002;99:16093-16098. |
| 96. | Stocker R, Yamamoto Y, McDonagh AF, Glazer AN, Ames BN. Bilirubin is an antioxidant of possible physiological importance. Science. 1987;235:1043-1046. |
| 97. | Neuzil J, Stocker R. Free and albumin-bound bilirubin are efficient co-antioxidants for alpha-tocopherol, inhibiting plasma and low density lipoprotein lipid peroxidation. J Biol Chem. 1994;269:16712-16719. |
| 98. | Foresti R, Sarathchandra P, Clark JE, Green CJ, Motterlini R. Peroxynitrite induces haem oxygenase-1 in vascular endothelial cells: a link to apoptosis. Biochem J. 1999;339:729-736. |
| 99. | Kwak JY, Takeshige K, Cheung BS, Minakami S. Bilirubin inhibits the activation of superoxide-producing NADPH oxidase in a neutrophil cell-free system. Biochim Biophys Acta. 1991;1076:369-373. |
| 100. | Perez MJ, Castano B, Gonzalez-Buitrago JM, Marin JJ. Multiple protective effects of melatonin against maternal cholestasis-induced oxidative stress and apoptosis in the rat fetal liver-placenta-maternal liver trio. J Pineal Res. 2007;43:130-139. |
| 101. | Gourley GR. Bilirubin metabolism and kernicterus. Adv Pediatr. 1997;44:173-229. |
| 102. | Schmid R. The distinguished lecture: Pyrrolic victories. Trans Assoc Am Physicians. 1976;89:64-76. |
| 103. | Serrano MA, Bayon JE, Pascolo L, Tiribelli C, Ostrow JD, Gonzalez-Gallego J, Marin JJ. Evidence for carrier-mediated transport of unconjugated bilirubin across plasma membrane vesicles from human placental trophoblast. Placenta. 2002;23:527-535. |
| 104. | Briz O, Macias RI, Serrano MA, Gonzalez-Gallego J, Bayon JE, Marin JJ. Excretion of foetal bilirubin by the rat placenta-maternal liver tandem. Placenta. 2003;24:462-472. |
| 105. | Vander Jagt DL, Wilson SP, Heidrich JE. Purification and bilirubin binding properties of glutathione S-transferase from human placenta. FEBS Lett. 1981;136:319-321. |
| 106. | Briz O, Macias RI, Perez MJ, Serrano MA, Marin JJ. Excretion of fetal biliverdin by the rat placenta-maternal liver tandem. Am J Physiol Regul Integr Comp Physiol. 2006;290:R749-R756. |
| 107. | Ganapathy V, Prasad PD, Ganapathy ME, Leibach FH. Placental transporters relevant to drug distribution across the maternal-fetal interface. J Pharmacol Exp Ther. 2000;294:413-420. |
| 108. | van der Aa EM, Peereboom-Stegeman JH, Noordhoek J, Gribnau FW, Russel FG. Mechanisms of drug transfer across the human placenta. Pharm World Sci. 1998;20:139-148. |
| 109. | Redmond GP. Physiological changes during pregnancy and their implications for pharmacological treatment. Clin Invest Med. 1985;8:317-322. |
| 110. | Stulc J. Placental transfer of inorganic ions and water. Physiol Rev. 1997;77:805-836. |
| 111. | Pacifici GM. Placental transfer of antibiotics administered to the mother: a review. Int J Clin Pharmacol Ther. 2006;44:57-63. |
| 112. | Pacifici GM, Nottoli R. Placental transfer of drugs admini-stered to the mother. Clin Pharmacokinet. 1995;28:235-269. |
| 113. | Chappuy H, Treluyer JM, Jullien V, Dimet J, Rey E, Fouche M, Firtion G, Pons G, Mandelbrot L. Maternal-fetal transfer and amniotic fluid accumulation of nucleoside analogue reverse transcriptase inhibitors in human immunodeficiency virus-infected pregnant women. Antimicrob Agents Chemother. 2004;48:4332-4336. |
| 114. | Ganapathy V, Prasad PD. Role of transporters in placental transfer of drugs. Toxicol Appl Pharmacol. 2005;207:381-387. |
| 115. | Halestrap AP, Meredith D. The SLC16 gene family-from monocarboxylate transporters (MCTs) to aromatic amino acid transporters and beyond. Pflugers Arch. 2004;447:619-628. |
| 116. | Barros LF, Yudilevich DL, Jarvis SM, Beaumont N, Young JD, Baldwin SA. Immunolocalisation of nucleoside transporters in human placental trophoblast and endothelial cells: evidence for multiple transporter isoforms. Pflugers Arch. 1995;429:394-399. |
| 117. | Griffiths M, Yao SY, Abidi F, Phillips SE, Cass CE, Young JD, Baldwin SA. Molecular cloning and characterization of a nitrobenzylthioinosine-insensitive (ei) equilibrative nucleoside transporter from human placenta. Biochem J. 1997;328:739-743. |
| 118. | Griffiths M, Beaumont N, Yao SY, Sundaram M, Boumah CE, Davies A, Kwong FY, Coe I, Cass CE, Young JD. Cloning of a human nucleoside transporter implicated in the cellular uptake of adenosine and chemotherapeutic drugs. Nat Med. 1997;3:89-93. |
| 119. | Grillo MA, Lanza A, Colombatto S. Transport of amino acids through the placenta and their role. Amino Acids. 2008;34:517-523. |
| 120. | Cleal JK, Lewis RM. The mechanisms and regulation of placental amino acid transport to the human foetus. J Neuroendocrinol. 2008;20:419-426. |
| 121. | Pastrakuljic A, Derewlany LO, Koren G. Maternal cocaine use and cigarette smoking in pregnancy in relation to amino acid transport and fetal growth. Placenta. 1999;20:499-512. |
| 122. | Ganapathy VV, Prasad PD, Ganapathy ME, Leibach FH. Drugs of abuse and placental transport. Adv Drug Deliv Rev. 1999;38:99-110. |
| 123. | Ganapathy V, Leibach FH 1995. Placental biogenic amines and their transporters. editor. Placental Toxicology. CRC Press: Boca Raton, FL 1995; 161-174. |
| 124. | Hagenbuch B, Meier PJ. Organic anion transporting polypeptides of the OATP/ SLC21 family: phylogenetic classification as OATP/ SLCO superfamily, new nomenclature and molecular/functional properties. Pflugers Arch. 2004;447:653-665. |
| 125. | Hagenbuch B, Gui C. Xenobiotic transporters of the human organic anion transporting polypeptides (OATP) family. Xenobiotica. 2008;38:778-801. |
| 126. | Meier-Abt F, Mokrab Y, Mizuguchi K. Organic anion transporting polypeptides of the OATP/SLCO superfamily: identification of new members in nonmammalian species, comparative modeling and a potential transport mode. J Membr Biol. 2005;208:213-227. |
| 127. | Briz O, Macias RI, Vallejo M, Silva A, Serrano MA, Marin JJ. Usefulness of liposomes loaded with cytostatic bile acid derivatives to circumvent chemotherapy resistance of enterohepatic tumors. Mol Pharmacol. 2003;63:742-750. |
| 128. | Hagenbuch B, Meier PJ. The superfamily of organic anion transporting polypeptides. Biochim Biophys Acta. 2003;1609:1-18. |
| 129. | Kekuda R, Prasad PD, Wu X, Wang H, Fei YJ, Leibach FH, Ganapathy V. Cloning and functional characterization of a potential-sensitive, polyspecific organic cation transporter (OCT3) most abundantly expressed in placenta. J Biol Chem. 1998;273:15971-15979. |
| 130. | Sata R, Ohtani H, Tsujimoto M, Murakami H, Koyabu N, Nakamura T, Uchiumi T, Kuwano M, Nagata H, Tsukimori K. Functional analysis of organic cation transporter 3 expressed in human placenta. J Pharmacol Exp Ther. 2005;315:888-895. |
| 131. | Zwart R, Verhaagh S, Buitelaar M, Popp-Snijders C, Barlow DP. Impaired activity of the extraneuronal monoamine transporter system known as uptake-2 in Orct3/Slc22a3-deficient mice. Mol Cell Biol. 2001;21:4188-4196. |
| 132. | Grube M, Schwabedissen HM, Draber K, Prager D, Moritz KU, Linnemann K, Fusch C, Jedlitschky G, Kroemer HK. Expression, localization, and function of the carnitine transporter octn2 (slc22a5) in human placenta. Drug Metab Dispos. 2005;33:31-37. |
| 133. | Ohashi R, Tamai I, Yabuuchi H, Nezu JI, Oku A, Sai Y, Shimane M, Tsuji A. Na(+)-dependent carnitine transport by organic cation transporter (OCTN2): its pharmacological and toxicological relevance. J Pharmacol Exp Ther. 1999;291:778-784. |
| 134. | Ganapathy ME, Huang W, Rajan DP, Carter AL, Sugawara M, Iseki K, Leibach FH, Ganapathy V. beta-lactam antibiotics as substrates for OCTN2, an organic cation/carnitine transporter. J Biol Chem. 2000;275:1699-1707. |
| 135. | Hakkola J, Raunio H, Purkunen R, Pelkonen O, Saarikoski S, Cresteil T, Pasanen M. Detection of cytochrome P450 gene expression in human placenta in first trimester of pregnancy. Biochem Pharmacol. 1996;52:379-383. |
| 136. | Hakkola J, Pasanen M, Hukkanen J, Pelkonen O, Maenpaa J, Edwards RJ, Boobis AR, Raunio H. Expression of xenobiotic-metabolizing cytochrome P450 forms in human full-term placenta. Biochem Pharmacol. 1996;51:403-411. |
| 137. | Pasanen M. The expression and regulation of drug metabolism in human placenta. Adv Drug Deliv Rev. 1999;38:81-97. |
| 138. | Myllynen P, Pasanen M, Pelkonen O. Human placenta: a human organ for developmental toxicology research and biomonitoring. Placenta. 2005;26:361-371. |
| 139. | Barnea ER. Modulatory effect of maternal serum on xenobiotic metabolizing activity of placental explants: modification by cigarette smoking. Hum Reprod. 1994;9:1017-1021. |
| 140. | Karl PI, Gordon BH, Lieber CS, Fisher SE. Acetaldehyde production and transfer by the perfused human placental cotyledon. Science. 1988;242:273-275. |
| 141. | Weier N, He SM, Li XT, Wang LL, Zhou SF. Placental drug disposition and its clinical implications. Curr Drug Metab. 2008;9:106-121. |
| 142. | Everson RB, Randerath E, Santella RM, Cefalo RC, Avitts TA, Randerath K. Detection of smoking-related covalent DNA adducts in human placenta. Science. 1986;231:54-57. |
| 143. | Everson RB, Randerath E, Santella RM, Avitts TA, Weinstein IB, Randerath K. Quantitative associations between DNA damage in human placenta and maternal smoking and birth weight. J Natl Cancer Inst. 1988;80:567-576. |
| 144. | Utoguchi N, Chandorkar GA, Avery M, Audus KL. Functional expression of P-glycoprotein in primary cultures of human cytotrophoblasts and BeWo cells. Reprod Toxicol. 2000;14:217-224. |
| 145. | Young AM, Allen CE, Audus KL. Efflux transporters of the human placenta. Adv Drug Deliv Rev. 2003;55:125-132. |
| 146. | Fromm MF. Importance of P-glycoprotein at blood-tissue barriers. Trends Pharmacol Sci. 2004;25:423-429. |
| 147. | Ceckova-Novotna M, Pavek P, Staud F. P-glycoprotein in the placenta: expression, localization, regulation and function. Reprod Toxicol. 2006;22:400-410. |
| 148. | Meyer Zu Schwabedissen HE, Grube M, Heydrich B, Linnemann K, Fusch C, Kroemer HK, Jedlitschky G. Expression, localization, and function of MRP5 (ABCC5), a transporter for cyclic nucleotides, in human placenta and cultured human trophoblasts: effects of gestational age and cellular differentiation. Am J Pathol. 2005;166:39-48. |
| 149. | Mao Q. BCRP/ABCG2 in the placenta: expression, function and regulation. Pharm Res. 2008;25:1244-1255. |
| 150. | Grube M, Reuther S, Meyer Zu Schwabedissen H, Kock K, Draber K, Ritter CA, Fusch C, Jedlitschky G, Kroemer HK. Organic anion transporting polypeptide 2B1 and breast cancer resistance protein interact in the transepithelial transport of steroid sulfates in human placenta. Drug Metab Dispos. 2007;35:30-35. |
| 151. | Kobayashi D, Ieiri I, Hirota T, Takane H, Maegawa S, Kigawa J, Suzuki H, Nanba E, Oshimura M, Terakawa N. Functional assessment of ABCG2 (BCRP) gene polymorphisms to protein expression in human placenta. Drug Metab Dispos. 2005;33:94-101. |