Published online Apr 1, 2004. doi: 10.3748/wjg.v10.i7.985
Revised: November 3, 2003
Accepted: December 6, 2003
Published online: April 1, 2004
AIM: To construct a recombinant prokaryotic expression vector inserted with Helicobacter pylori vacA gene and identify the immunity of the expressed recombinant protein, and to determine prevalence of vacA-carrying/VacA expressing H pylori isolates and seroprevalence of specific ant-VacA antibody in H pylori infected patients.
METHODS: Polymerase chain reaction technique was used to amplify complete vacA gene of H pylori strain NCTC11637 and to detect vacA gene in 109 H pylori isolates. The amplification product of the complete vacA gene was sequenced after T-A cloning. A recombinant expression vector inserted with a complete vacA gene fragment, named as pET32a-vacA, was constructed. Expression of the target recombinant protein VacA (rVacA) was examined by SDS-PAGE. Western blot using commercial antibodies against whole cell of H pylori and an immunodiffusion assay using self-prepared rabbit anti-rVacA antibody were applied to determine immunoreaction and antigenicity of rVacA. Two ELISA methods were established to detect VacA expression in H pylori isolates and the specific anti-VacA antibody in sera from 125 patients infected with H pylori.
RESULTS: In comparison with the reported corresponding sequences, homologies of nucleotide and putative amino acid sequences of the cloned vacA gene were 99.82% and 100%, respectively. The constructed recombinant prokaryotic expression system efficiently produced rVacA. rVacA was able to combine with the commercial antibodies against whole cell of H pylori and to induce the immunized rabbit to produce specific antibody with an immunodiffusion titer of 1:4. All tested H pylori isolates carried vacA gene, but only 66.1% expressed VacA protein. Of the serum samples tested, 42.4% were positive for specific anti-VacA antibody.
CONCLUSION: A prokaryotic expression system of H pylori vacA gene was successfully constructed. The expressed rVacA can be used to detect specific anti-VacA antibody in human and to prepare antiserum in animals. The high frequency of vacA gene in H pylori isolates, but with a low frequency of VacA expression and specific anti-VacA antibody in H pylori infected patients implies that VacA is not an ideal antigen for H pylori vaccine.
- Citation: Yan J, Mao YF. Construction of a prokaryotic expression system of vacA gene and detection of vacA gene, VacA protein in Helicobacter pylori isolates and ant-VacA antibody in patients’ sera. World J Gastroenterol 2004; 10(7): 985-990
- URL: https://www.wjgnet.com/1007-9327/full/v10/i7/985.htm
- DOI: https://dx.doi.org/10.3748/wjg.v10.i7.985
In China, gastritis and peptic ulcer are two most prevalent gastric diseases, and gastric cancer is one of the malignant tumors with high morbidities[1-34]. Helicobacter pylori is recognized as a human-specific gastric pathogen that colonizes the stomachs of at least half of the world’s populations[35]. Most infected individuals are asymptomatic. However, in some subjects, the infection causes acute/chronic gastritis or peptic ulceration, and plays an important role in the development of gastric adenocarcinoma, mucosa-associated lymphoid tissue (MALT) lymphoma and primary gastric non-Hodgkin’s lymphoma[36-43].
Vacuolating cytotoxin, an important pathogenic factor of H pylori, is able to cause vacuolar degeneration in epithelial cells such as HeLa and RK-13 cell lines[44-46]. The gene of vacuolating cytotoxin has 3 864-3 888 bp in size containing a region for a signal peptide at the amino-end, a fragment with approximate 2 241 bp (vacA) and a region for a peptide at the cariboxyl-end[47]. The signal and C-end peptides are left in inner and outer membranes of H pylori during secreting the cytotoxin[48]. VacA, responsible for the toxicity of vacuolating cytotoxin, is excreted out of the bacterium[49]. In some of the previous studies, VacA was demonstrated as a fine antigen for H pylori vaccine[50,51]. However, it has been reported that almost all H pylori strains carry vacA gene but VacA is detectable only in 50-60% of the strains[52]. For a fine practical strategy of genetic engineering vaccine development, an antigen candidate must satisfy the requirements including exposure on the surface of bacterial body, universal distribution in different strains and strong antigenicity to induce a specific antibody. So it is a critical subject to determine VacA expression in different H pylori strains and the prevalence of specific anti-VacA antibody in infected individuals from various geographical areas. In this study, a recombinant expression plasmid containing complete vacA gene was constructed. By using ELISA, VacA expression in different H pylori isolates and seroprevalence of anti-VacA specific antibody from H pylori infected patients were determined.
H pylori strain NCTC11637 was kept in our laboratory. Primers for polymerase chain reaction (PCR) amplification were synthezed by BioAsia (Shanghai, China). Taq-plus high fidelity PCR kit and restriction endonucleases were purchased from TaKaRa (Dalian, China). The T-A Cloning kit and sequencing service were provided by BBST (Shanghai, China). A plasmid pET32a as an expression vector and E. coli BL21 DE3 as a host cell were provided by Novagen (Novagen, Madison, USA). Rabbit antiserum against whole cell of H pylori, HRP-labeling sheep antisera against rabbit IgG and against human IgG were purchased from DAKO (Glostrup, Denmark) and Jackson ImmunoResearch (West Grove, USA), respectively. Overall, 156 patients who were referred for upper endoscopy at four hospitals in Hangzhou during November 2001 to February 2003 due to various gastroduodenal diseases and who had a positive urease test on gastric biopsy specimens and serum samples collected, were included in the study. Reagents used in isolation and identification of H pylori were purchased from bioMérieux (Marcy I’Etoile, France).
Each biopsy specimen was homogenized with a tissue grinder and then inoculated on Columbia agar plates supplemented with 80 mL/L sheep blood, 5 g/L cyclodextrin, 5 mg/L trimethoprim, 10 mg/L vancomycin, 2.5 mg/L amphotericin B and 2500 U/L cefsoludin. The plates were incubated at 37 °C under microaerobic conditions (50 mL/L O2, 100 mL/L CO2 and 850 mL/L N2) for 3 to 5 d. A bacterial isolate was identified as H pylori according to typical Gram staining morphology, biochemical tests positive for urease and oxidase, and slide agglutination with the commercial rabbit antibodies against whole cell of H pylori. A total of 126(80.1%) H pylori strains isolated from the 156 specimens were well-characterized.
Genomic DNA from each H pylori strain was extracted by the conventional phenol-chloroform method and DNase-free RNase treatment. The obtained DNA was dissolved in TE buffer. Concentration and purity of the DNA preparations were determined by ultraviolet spectrophotometry[53].
Primers were designed to amplify complete vacA gene from H pylori strain NCTC11637 based on the published data (GenBank accession No.: AF049653)[54]. The sequences of sense primer with an endonuclease site of EcoR I and antisense primer with an endonuclease site of Xho I were 5’-GAGGAATTCATGGAAATACAACAAACACACCGC-3’ and 5’-GGACTCGAGTTAATTGGTACCTGTAGAA ACATTACC-3’, respectively. The total volume per PCR was 100 µL containing 2.5 mol/L each dNTP, 250 nmol/L each of the 2 primers, 15 mol/L MgCl2, 3.0 U Taq-plus polymerase, 100 ng DNA template and 1 × PCR buffer (pH8.3). The parameters for PCR were 94 °C for 5 min, × 1; 94 °C for 30 s, 58 °C for 30 s, 72 °C for 120 s, × 10; 94 °C for 30 s, 58 °C for 30 s, 72 °C for 135 s (15 s addition for the each of the following cycles), × 15; and then 72 °C for 15 min, × 1.
The sequences of primers for detecting vacA gene in H pylori isolates were 5’-TCAATATCAACAAGCTC-3’ (sense), and 5’-CCGCATGCTTTAATGTC-3’ (antisense)[55]. The reagents and the concentrations, reaction volume and PCR parameters were the same as those for amplifying complete vacA gene, except primer difference, annealing temperature at 52 °C, extensive time for 60 s and 35 cycles.
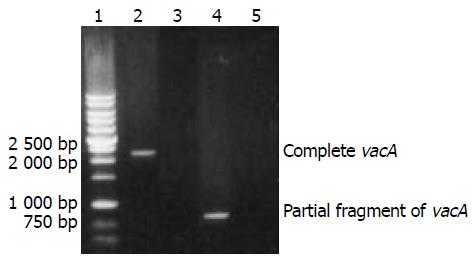
The results of PCR were visualized under UV light after electrophoresis in 10 g/L agarose pre-stained with ethidium bromide. The expected sizes of the target amplification fragments with whole length of vacA gene for cloning and partial length of vacA gene for detection were 2241 bp and 787 bp, respectively.
The target amplification fragment of complete vacA gene from H pylori strain NCTC11637 was cloned into plasmid vector pUCm-T (pUCm-T-vacA) by using the T-A Cloning kit according to the manufacturer’s instruction. The recombinant plasmid was amplified in E. coli DH5α and then extracted by Sambrook’s method[53]. A professional company (BBST) was responsible for nucleotide sequence analysis of the inserted fragment. Two plasmids, pUCm-T-vacA and pET32a, in 2 different strains of E. coli DH5α after amplified in LB medium were extracted and then digested with EcoRI and Xho I, respectively[53]. The target fragments of vacA gene and pET32a were recovered and then ligased. The recombinant expression vector pET32a-vacA was transformed into E. coli BL21DE3, and the expression system was named as pET32a-vacA-E. coliBL21DE3. vacA gene inserted in pET32a plasmid was sequenced again.
pET32a-vacA-E. coliBL21DE3 was rotatively cultured in LB medium at 37 °C and induced by isopropylthio-β-D-galactoside (IPTG) at different concentrations of 1.0, 0.5 and 0.1 mmol/L Supernatant and precipitate of the culture after incubation were separated by centrifugation and then the bacterial pallet was ultrasonically broken (300 V, 5 s × 3). SDS-PAGE was used to measure the molecular weight and output of rVacA, and Ni-NTA affinity chromatography was applied to collect the recombinant protein. The commercial rabbit antiserum against whole cell of H pylori and HRP-labeling sheep antiserum against rabbit IgG were respectively used as the first and second antibodies to determine the immunoreactivity of rVacA by Western blot. Rabbits were immunized with rVacA to prepare the antiserum. An immunodiffusion assay was performed to determine antigenicity of rVacA.
By using rVacA as the coated antigen at concentration of 20 µg/mL, a patient serum sample (1:200 dilution) as the first antibody and HRP-labeling sheep antibody against human IgG (1:4 000 dilution) as the second antibody, antibody against VacA in sera of the 126 H pylori infected patients were detected. The result of ELISA for a patient’s serum sample was considered as positive if the value of optical density at 490 nm (A490) was over the mean plus 3 SD of five different negative serum samples[56]. VacA expression in clinical isolates of H pylori was examined by using the ultrasonic supernatant of each isolate (50 µg/mL) as the coated antigen, the self-prepared rabbit anti-rVacA serum (1:2 000 dilution) as the first antibody and HRP-labeling sheep antibody against rabbit IgG (1:4 000 dilution) as the second antibody. The result of ELISA for a H pylori ultrasonic supernatant sample was considered as positive if its OD490 value was over the mean plus 3 SD of five separated E. coli ATCC 25922 ultrasonic supernatant samples at the same protein concentration[56].
The nucleotide and putative amino acid sequences of the cloned vacA gene were compared for homology with the published corresponding sequences (GenBank accession No. AF049653)[54].
All tested H pylori isolates (109/109) were positive for vacA gene. The target amplification products with whole length of vacA gene from H pylori strain NCTC11637 and with partial fragment of vacA gene from H pylori isolate were shown in Figure 1, respectively.
The nucleotide sequences of vacA gene in pUCm-T-vacA and pET32a-vacA were completely same. The nucleotide and putative amino acid sequences of the cloned vacA gene (Figure 2) showed 99.82% and 100% homologies, respectively, compared with the published sequence from H pylori strain NCTC11637 vacA gene (GenBank accession No.: AF049653)[54].
IPTG at concentrations of 1.0, 0.5 and 0.1 mmol/L efficiently induced the expression of rVacA in the pET32a-vacA-E. coli BL21DE3 system. The expressed rVacA was mainly presented in ultrasonic precipitates, and the output was approximate 15% of the total bacterial proteins (Figure 3).
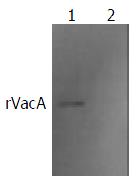
The commercial rabbit antibodies against whole cell of H pylori combined with rVacA as confirmed by Western blot (Figure 4). The titer demonstrated by immunodiffusion assay between rVacA and rabbit anti-rVacA serum was 1:4.
Since the mean ± SD of A490 values of the five negative serum samples was 0.275 ± 0.111 in the detection of specific antibodies in patients’ sera, the positive reference value was 0.625. According to the reference value, 42.4%(53/125, one serum sample was contaminated) of the tested patients’ serum samples were positive for antibody against rVacA with an A490 value range from 0.63-1.21. Since the mean ± SD of A490 values of the five negative bacterial controls was 0.098 ± 0.036 in the detection of VacA expression in H pylori isolates, the positive reference value was 0.205. According to the reference value, 66.1%(72/109, other 17 isolates could not be revived from -70 °C) of the tested H pylori isolates were positive for the epitope of rVacA with an A490 value range from 0.27-1.73.
Amoxciilin and metronidazole, used as routine therapeutic antibiotics in triple therapies, efficiently eradicate H pylori infection in vivo[57-59]. However, there are problems such as side effects, emergence of drug resistance and re-infection after withdrawal of the antibiotics, etc. It is generally considered that H pylori vaccination is the optimal strategy for the prevention and control of H pylori infection.
VacA, confirmed as a unique H pylori exotoxin with strong antigenicity[47,48], has been considered to be an antigen candidate in H pylori vaccine[50,51]. However, the low prevalence (50-60%) of VacA expression in H pylori strains and rare data on specific antibody inducement after H pylori infection in human make this consideration difficult to test. Therefore, we constructed a prokaryotic expression system of H pylori vacA gene, used rVacA to detect anti-VacA antibody in H pylori infected patients and prepared rabbit anti-rVacA serum to determine VacA expression in H pylori isolates, in order to determine the potential of VacA as an antigen in H pylori vaccine development.
The present study demonstrated high homologies (99.82% and 100%) of the nucleotide and putative amino acid sequences of the cloned vacA gene compared with the reported (GenBank accession No.: AF049653)[54] and efficient expression of pET32a-vacA-E. coliBL21DE3, indicating the successful establishment of a prokaryotic expression system of this target gene. The high specificity, immunoreactivity and strong antigenicity of rVacA exhibited in this study are beneficial to establish ELISAs to detect VacA-specific antibody in H pylori infected patients and VacA expression in H pylori isolates.
All tested H pylori isolates were positive for vacA gene by PCR. However, expression of VacA, not like those of UreB and HpaA in our previous studies[60], was detectable only in 66.1% of the tested H pylori isolates. The lower positive detection rate (42.4%) for VacA-specific antibody in sera of H pylori infected patients was also found. The fact that the prevalence of VacA expression is lower than that of vacA gene in H pylori strains suggests possible mutation of the gene[44], and the even lower seroprevalence of specific anti-VacA antibody in infected patients is probably due to the defect of signal peptide in the protein[61].
In conclusion, a prokaryotic expression system of H pylori vacA gene was successfully constructed. The expressed rVacA can be used to detect specific anti-VacA antibody in human and to prepare antiserum in animals. However, the evidence obtained from this study indicates a poor potential for VacA as an antigen in the development of H pylori vaccine. In addition, detection of specific anti-VacA antibody is not a reliable diagnostic indicator as to whether an individual is infected by H pylori.
We are grateful to the people’s Hospital of Zhejiang, the First Affiliated Hospital of Zhejiang University, the Second Affiliated Hospital of Zhejiang University and the Affiliated Run Run Shaw Hospital of Zhejiang University in Hangzhou for kindly providing gastric biopsies in this study.
Edited by Xia HHX and Xu FM
| 1. | Ye GA, Zhang WD, Liu LM, Shi L, Xu ZM, Chen Y, Zhou DY. Helicobacter pylori vacA gene polymorphism and chronic gastrosis. Shijie Huaren Xiaohua Zazhi. 2001;9:593-595. |
| 2. | Lu SY, Pan XZ, Peng XW, Shi ZL. Effect of Hp infection on gas-tric epithelial cell kinetics in stomach diseases. Shijie Huaren Xiaohua Zazhi. 1999;7:760-762. |
| 3. | Zhang Z, Yuan Y, Gao H, Dong M, Wang L, Gong YH. Apoptosis, proliferation and p53 gene expression of H. pylori associated gastric epithelial lesions. World J Gastroenterol. 2001;7:779-782. [PubMed] |
| 4. | Lu XL, Qian KD, Tang XQ, Zhu YL, Du Q. Detection of H.pylori DNA in gastric epithelial cells by in situ hybridization. World J Gastroenterol. 2002;8:305-307. [PubMed] |
| 5. | Yao YL, Xu B, Song YG, Zhang WD. Overexpression of cyclin E in Mongolian gerbil with Helicobacter pylori-induced gastric precancerosis. World J Gastroenterol. 2002;8:60-63. [PubMed] |
| 6. | Guo DL, Dong M, Wang L, Sun LP, Yuan Y. Expression of gastric cancer-associated MG7 antigen in gastric cancer, precancerous lesions and H. pylori -associated gastric diseases. World J Gastroenterol. 2002;8:1009-1013. [PubMed] |
| 7. | Peng ZS, Liang ZC, Liu MC, Ouyang NT. Studies on gastric epi-thelial cell proliferation and apoptosis in Hp associated gastric ulcer. Shijie Huaren Xiaohua Zazhi. 1999;7:218-219. |
| 8. | Hiyama T, Haruma K, Kitadai Y, Miyamoto M, Tanaka S, Yoshihara M, Sumii K, Shimamoto F, Kajiyama G. B-cell monoclonality in Helicobacter pylori-associated chronic atrophic gastritis. Virchows Arch. 2001;438:232-237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 9. | Harry XH. Association between Helicobacter pylori and gastric cancer: current knowledge and future research. World J Gastroenterol. 1998;4:93-96. [PubMed] |
| 10. | Quan J, Fan XG. Progress in experimental research of Helicobacter pylori infection and gastric carcinoma. Shijie Huaren Xiaohua Zazhi. 1999;7:1068-1069. |
| 11. | Liu HF, Liu WW, Fang DC. Study of the relationship between apoptosis and proliferation in gastric carcinoma and its precan-cerous lesion. Shijie Huaren Xiaohua Zazhi. 1999;7:649-651. |
| 12. | Zhu ZH, Xia ZS, He SG. The effects of ATRA and 5-Fu on telomerase activity and cell growth of gastric cancer cells in vitro. Shijie Huaren Xiaohua Zazhi. 2000;8:669-673. |
| 13. | Tu SP, Zhong J, Tan JH, Jiang XH, Qiao MM, Wu YX, Jiang SH. Induction of apoptosis by arsenic trioxide and hydroxy camptothecin in gastriccancer cells in vitro. World J Gastroenterol. 2000;6:532-539. [PubMed] |
| 14. | Cai L, Yu SZ, Zhang ZF. Helicobacter pylori infection and risk of gastric cancer in Changle County,Fujian Province,China. World J Gastroenterol. 2000;6:374-376. [PubMed] |
| 15. | Yao XX, Yin L, Zhang JY, Bai WY, Li YM, Sun ZC. HTERT ex-pression and cellular immunity in gastric cancer and precancerosis. Shijie Huaren Xiaohua Zazhi. 2001;9:508-512. |
| 16. | Xu AG, Li SG, Liu JH, Gan AH. Function of apoptosis and expression of the proteins Bcl-2, p53 and C-myc in the development of gastric cancer. World J Gastroenterol. 2001;7:403-406. [PubMed] |
| 17. | Wang X, Lan M, Shi YQ, Lu J, Zhong YX, Wu HP, Zai HH, Ding J, Wu KC, Pan BR. Differential display of vincristine-resistance-related genes in gastric cancer SGC7901 cell. World J Gastroenterol. 2002;8:54-59. [PubMed] |
| 18. | Liu JR, Li BX, Chen BQ, Han XH, Xue YB, Yang YM, Zheng YM, Liu RH. Effect of cis-9, trans-11-conjugated linoleic acid on cell cycle of gastric adenocarcinoma cell line (SGC-7901). World J Gastroenterol. 2002;8:224-229. [PubMed] |
| 19. | Cai L, Yu SZ. A molecular epidemiologic study on gastric cancer in Changle, Fujian Province. Shijie Huaren Xiaohua Zazhi. 1999;7:652-655. |
| 20. | Gao GL, Yang Y, Yang SF, Ren CW. Relationship between pro-liferation of vascular andothelial cells and gastric cancer. Shijie Huaren Xiaohua Zazhi. 2000;8:282-284. |
| 21. | Xue XC, Fang GE, Hua JD. Gastric cancer and apoptosis. Shijie Huaren Xiaohua Zazhi. 1999;7:359-361. |
| 22. | Niu WX, Qin XY, Liu H, Wang CP. Clinicopathological analysis of patients with gastric cancer in 1200 cases. World J Gastroenterol. 2001;7:281-284. [PubMed] |
| 23. | Li XY, Wei PK. Diagnosis of stomach cancer by serum tumor markers. Shijie Huaren Xiaohua Zazhi. 2001;9:568-570. |
| 24. | Fang DC, Yang SM, Zhou XD, Wang DX, Luo YH. Telomere erosion is independent of microsatellite instability but related to loss of heterozygosity in gastric cancer. World J Gastroenterol. 2001;7:522-526. [PubMed] |
| 25. | Morgner A, Miehlke S, Stolte M, Neubauer A, Alpen B, Thiede C, Klann H, Hierlmeier FX, Ell C, Ehninger G. Development of early gastric cancer 4 and 5 years after complete remission of Helicobacter pylori associated gastric low grade marginal zone B cell lymphoma of MALT type. World J Gastroenterol. 2001;7:248-253. [PubMed] |
| 26. | Deng DJ. progress of gastric cancer etiology: N-nitrosamides 1999s. World J Gastroenterol. 2000;6:613-618. [PubMed] |
| 27. | Liu ZM, Shou NH, Jiang XH. Expression of lung resistance protein in patients with gastric carcinoma and its clinical significance. World J Gastroenterol. 2000;6:433-434. [PubMed] |
| 28. | Guo CQ, Wang YP, Liu GY, Ma SW, Ding GY, Li JC. Study on Helicobacter pylori infection and p53, c-erbB-2 gene expression in carcinogenesis of gastric mucosa. Shijie Huaren Xiaohua Zazhi. 1999;7:313-315. |
| 29. | Cai L, Yu SZ, Ye WM, Yi YN. Fish sauce and gastric cancer: an ecological study in Fujian Province,China. World J Gastroenterol. 2000;6:671-675. [PubMed] |
| 30. | Xue FB, Xu YY, Wan Y, Pan BR, Ren J, Fan DM. Association of H. pylori infection with gastric carcinoma: a Meta analysis. World J Gastroenterol. 2001;7:801-804. [PubMed] |
| 31. | Wang RT, Wang T, Chen K, Wang JY, Zhang JP, Lin SR, Zhu YM, Zhang WM, Cao YX, Zhu CW. Helicobacter pylori infection and gastric cancer: evidence from a retrospective cohort study and nested case-control study in China. World J Gastroenterol. 2002;8:1103-1107. [PubMed] |
| 32. | Hua JS. Effect of Hp: cell proliferation and apoptosis on stomach cancer. Shijie Huaren Xiaohua Zazhi. 1999;7:647-648. |
| 33. | Liu DH, Zhang XY, Fan DM, Huang YX, Zhang JS, Huang WQ, Zhang YQ, Huang QS, Ma WY, Chai YB. Expression of vascular endothelial growth factor and its role in oncogenesis of human gastric carcinoma. World J Gastroenterol. 2001;7:500-505. [PubMed] |
| 34. | Cao WX, Ou JM, Fei XF, Zhu ZG, Yin HR, Yan M, Lin YZ. Methionine-dependence and combination chemotherapy on human gastric cancer cells in vitro. World J Gastroenterol. 2002;8:230-232. [PubMed] |
| 35. | Michetti P, Kreiss C, Kotloff KL, Porta N, Blanco JL, Bachmann D, Herranz M, Saldinger PF, Corthésy-Theulaz I, Losonsky G. Oral immunization with urease and Escherichia coli heat-labile enterotoxin is safe and immunogenic in Helicobacter pylori-infected adults. Gastroenterology. 1999;116:804-812. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 234] [Cited by in RCA: 231] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 36. | Suganuma M, Kurusu M, Okabe S, Sueoka N, Yoshida M, Wakatsuki Y, Fujiki H. Helicobacter pylori membrane protein 1: a new carcinogenic factor of Helicobacter pylori. Cancer Res. 2001;61:6356-6359. [PubMed] |
| 37. | Nakamura S, Matsumoto T, Suekane H, Takeshita M, Hizawa K, Kawasaki M, Yao T, Tsuneyoshi M, Iida M, Fujishima M. Pre-dictive value of endoscopic ultrasonography for regression of gastric low grade and high grade MALT lymphomas after eradi-cation of Helicobacter pylori. Gut. 2001;48:454-460. [RCA] [DOI] [Full Text] [Cited by in Crossref: 170] [Cited by in RCA: 153] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 38. | Uemura N, Okamoto S, Yamamoto S, Matsumura N, Yamaguchi S, Yamakido M, Taniyama K, Sasaki N, Schlemper RJ. Helicobacter pylori infection and the development of gastric cancer. N Engl J Med. 2001;345:784-789. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3126] [Cited by in RCA: 3187] [Article Influence: 132.8] [Reference Citation Analysis (0)] |
| 39. | Morgner A, Miehlke S, Fischbach W, Schmitt W, Müller-Hermelink H, Greiner A, Thiede C, Schetelig J, Neubauer A, Stolte M. Complete remission of primary high-grade B-cell gastric lymphoma after cure of Helicobacter pylori infection. J Clin Oncol. 2001;19:2041-2048. [PubMed] |
| 40. | Kate V, Ananthakrishnan N, Badrinath S. Effect of Helicobacter pylori eradication on the ulcer recurrence rate after simple closure of perforated duodenal ulcer: retrospective and prospective randomized controlled studies. Br J Surg. 2001;88:1054-1058. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 58] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 41. | Zhuang XQ, Lin SR. Progress in research on the relationship be-tween Hp and stomach cancer. Shijie Huaren Xiaohua Zazhi. 2000;8:206-207. |
| 42. | Gao HJ, Yu LZ, Bai JF, Peng YS, Sun G, Zhao HL, Miu K, L XZ, Zhang XY, Zhao ZQ. Multiple genetic alterations and behavior of cellular biology in gastric cancer and other gastric mucosal lesions: H.pylori infection, histological types and staging. World J Gastroenterol. 2000;6:848-854. [PubMed] |
| 43. | Yao YL, Zhang WD. Relation between Helicobacter pylori and gas-tric cancer. Shijie Huaren Xiaohua Zazhi. 2001;9:1045-1049. |
| 44. | Atherton JC, Cao P, Peek RM, Tummuru MK, Blaser MJ, Cover TL. Mosaicism in vacuolating cytotoxin alleles of Helicobacter pylori. Association of specific vacA types with cytotoxin production and peptic ulceration. J Biol Chem. 1995;270:17771-17777. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1078] [Cited by in RCA: 1108] [Article Influence: 36.9] [Reference Citation Analysis (0)] |
| 45. | Harris PR, Cover TL, Crowe DR, Orenstein JM, Graham MF, Blaser MJ, Smith PD. Helicobacter pylori cytotoxin induces vacuolation of primary human mucosal epithelial cells. Infect Immun. 1996;64:4867-4871. [PubMed] |
| 46. | Pagliaccia C, de Bernard M, Lupetti P, Ji X, Burroni D, Cover TL, Papini E, Rappuoli R, Telford JL, Reyrat JM. The m2 form of the Helicobacter pylori cytotoxin has cell type-specific vacuolating activity. Proc Natl Acad Sci U S A. 1998;95:10212-10217. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 146] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 47. | Phadnis SH, Ilver D, Janzon L, Normark S, Westblom TU. Pathological significance and molecular characterization of the vacuolating toxin gene of Helicobacter pylori. Infect Immun. 1994;62:1557-1565. [PubMed] |
| 48. | Montecucco C, Papini E, de Bernard M, Zoratti M. Molecular and cellular activities of Helicobacter pylori pathogenic factors. FEBS Lett. 1999;452:16-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 41] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 49. | Censini S, Lange C, Xiang Z, Crabtree JE, Ghiara P, Borodovsky M, Rappuoli R, Covacci A. cag, a pathogenicity island of Helicobacter pylori, encodes type I-specific and disease-associated virulence factors. Proc Natl Acad Sci U S A. 1996;93:14648-14653. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1375] [Cited by in RCA: 1393] [Article Influence: 48.0] [Reference Citation Analysis (0)] |
| 50. | Marchetti M, Aricò B, Burroni D, Figura N, Rappuoli R, Ghiara P. Development of a mouse model of Helicobacter pylori infection that mimics human disease. Science. 1995;267:1655-1658. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 441] [Cited by in RCA: 409] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 51. | Prinz C, Hafsi N, Voland P. Helicobacter pylori virulence factors and the host immune response: implications for therapeutic vaccination. Trends Microbiol. 2003;11:134-138. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 43] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 52. | Cover TL, Blaser MJ. Purification and characterization of the vacuolating toxin from Helicobacter pylori. J Biol Chem. 1992;267:10570-10575. [PubMed] |
| 53. | Sambrook J, Fritsch EF, Maniatis T. Molecular Cloning, A Labo-ratory Manual [M]. 2nd edition. New York: Cold Spring Harbor Laboratory Press. 1989, pp1.21-1.52, 2.60-2.80, 7.3-7.35, 9.14-9.22. . |
| 54. | Ito Y, Azuma T, Ito S, Suto H, Miyaji H, Yamazaki Y, Kohli Y, Kuriyama M. Full-length sequence analysis of the vacA gene from cytotoxic and noncytotoxic Helicobacter pylori. J Infect Dis. 1998;178:1391-1398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 47] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 55. | Strobel S, Bereswill S, Balig P, Allgaier P, Sonntag HG, Kist M. Identification and analysis of a new vacA genotype variant of Helicobacter pylori in different patient groups in Germany. J Clin Microbiol. 1998;36:1285-1289. [PubMed] |
| 56. | Chen Y, Wang J, Shi L. [In vitro study of the biological activities and immunogenicity of recombinant adhesin of Heliobacter pylori rHpaA]. Zhonghua Yi Xue Za Zhi. 2001;81:276-279. [PubMed] |
| 57. | McMahon BJ, Hennessy TW, Bensler JM, Bruden DL, Parkinson AJ, Morris JM, Reasonover AL, Hurlburt DA, Bruce MG, Sacco F. The relationship among previous antimicrobial use, antimicrobial resistance, and treatment outcomes for Helicobacter pylori infections. Ann Intern Med. 2003;139:463-469. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 141] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 58. | Wong WM, Gu Q, Wang WH, Fung FM, Berg DE, Lai KC, Xia HH, Hu WH, Chan CK, Chan AO. Effects of primary metronidazole and clarithromycin resistance to Helicobacter pylori on omeprazole, metronidazole, and clarithromycin triple-therapy regimen in a region with high rates of metronidazole resistance. Clin Infect Dis. 2003;37:882-889. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 28] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 59. | Chi CH, Lin CY, Sheu BS, Yang HB, Huang AH, Wu JJ. Qua-druple therapy containing amoxicillin and tetracycline is an ef-fective regimen to rescue failed triple therapy by overcoming the antimicrobial resistance of Helicobacter pylori. Aliment Pharmacol Ther. 2003;18:347-353. [RCA] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 46] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 60. | Mao YF, Yan J, Li LW, Li SP. Construction of hpaA gene from a clinical isolate of Helicobacter pylori and identification of fusion protein. World J Gastroenterol. 2003;9:1529-1536. [PubMed] |
| 61. | Kim N, Weeks DL, Shin JM, Scott DR, Young MK, Sachs G. Proteins released by Helicobacter pylori in vitro. J Bacteriol. 2002;184:6155-6162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 76] [Article Influence: 3.3] [Reference Citation Analysis (0)] |