Copyright
©The Author(s) 2025.
World J Gastroenterol. Mar 21, 2025; 31(11): 102848
Published online Mar 21, 2025. doi: 10.3748/wjg.v31.i11.102848
Published online Mar 21, 2025. doi: 10.3748/wjg.v31.i11.102848
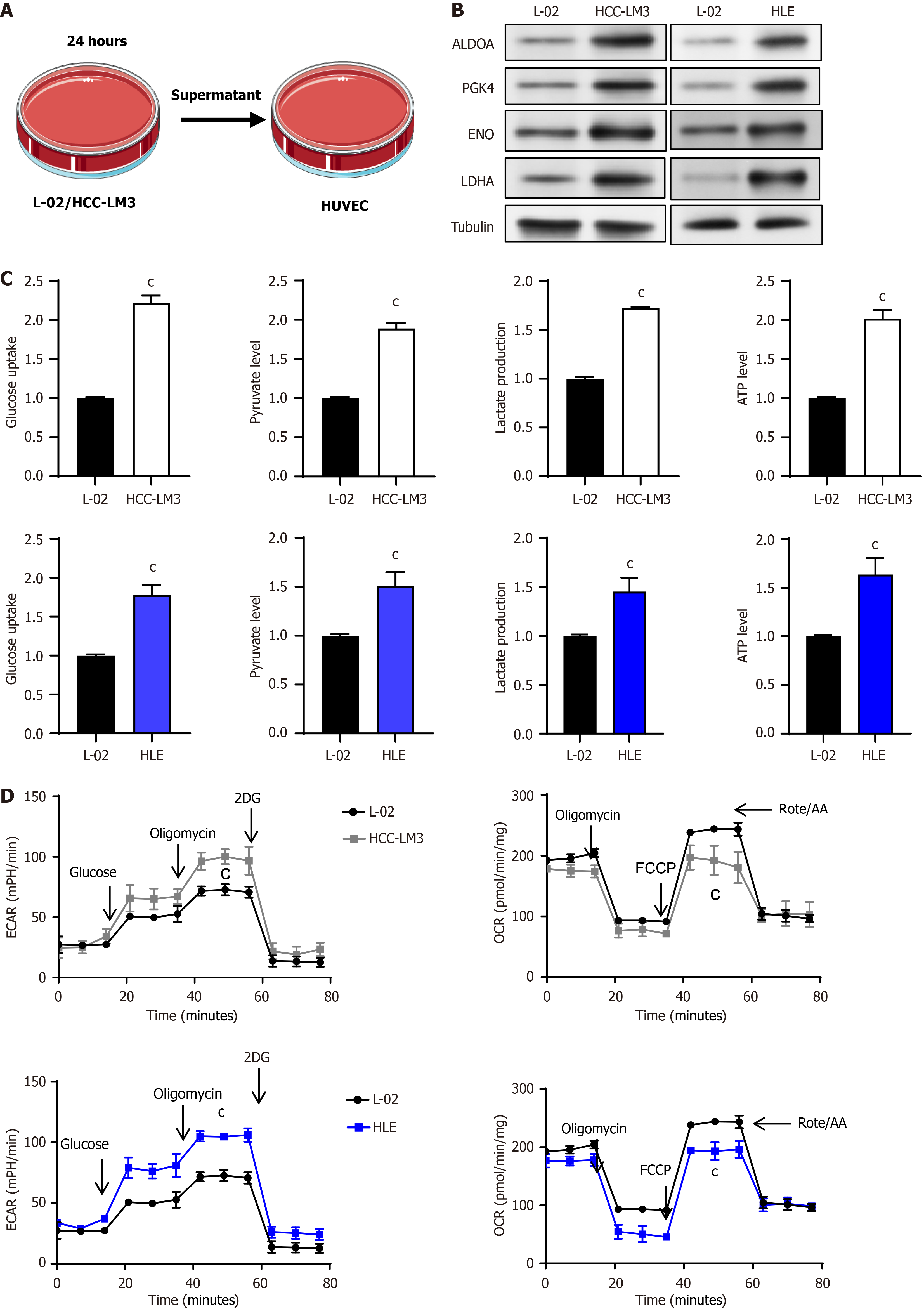
Figure 1 Enhanced glycolysis activity of vascular endothelial cells after co-culture with liver cancer cells.
A: Schematic diagram of co-culture of liver cells or liver cancer cells with vascular endothelial cells (VECs); B: Expression analysis of key glycolytic enzymes in VECs co-cultured with liver cells or liver cancer cells using Western blotting; C: Quantification of glucose consumption, pyruvate synthesis, lactate release, and adenosine triphosphate production during glycolysis in VECs; D: Evaluation of the glycolytic potential and oxidative phosphorylation efficiency of VECs when co-cultured with either liver cells or liver cancer cells, utilizing extracellular acidification rate and oxygen consumption rate. cP < 0.001. HUVEC: Human umbilical vein endothelial cells; ALDOA: Aldolase A, PGK: Phosphoglycerate kinase, ENO: Enolase, LDHA: Lactate dehydrogenase A; HCC: Hepatocellular carcinoma; ATP: Adenosine triphosphate; ECAR: Extracellular acidification rate; 2DG: 2-deoxy-D-glucose; OCR: Oxygen consumption rate; FCCP: Carbonylcyanide-p-trifluoromethoxyphenylhydrazone.
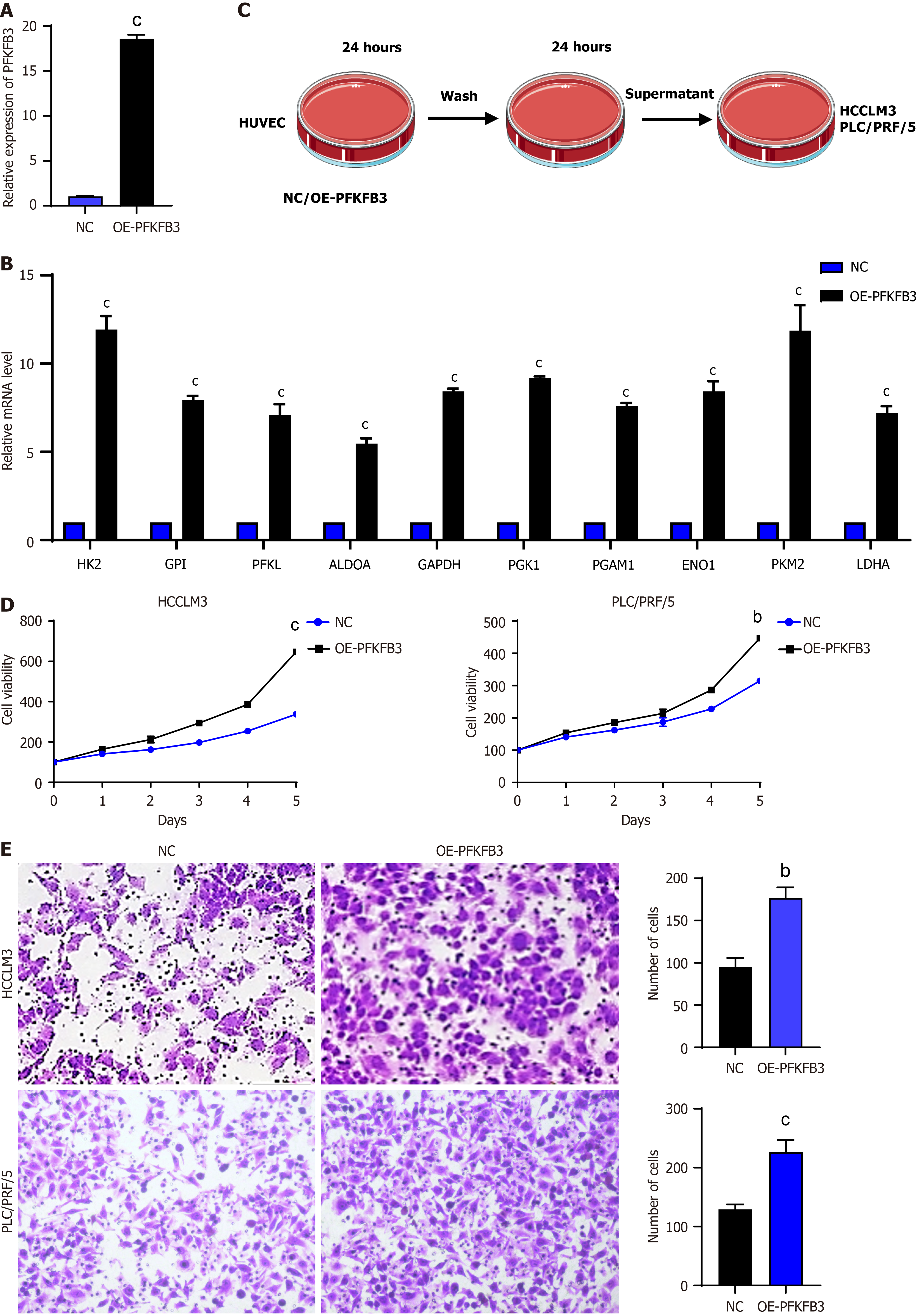
Figure 2 Increased proliferation and migration of liver cancer cells due to high glycolysis vascular endothelial cells.
A: Expression levels of 6-phosphofructo-2-kinase/fructose-2,6-biphosphatase 3 in human umbilical vein endothelial cells; B: Expression levels of key glycolytic enzymes in vascular endothelial cells (VECs) detected by quantitative real-time polymerase chain reaction; C: Schematic diagram of co-culture of VECs with liver cancer cells; D: Cell Counting Kit-8 proliferation assay of HCCLM3 and PLC/PRF/5 liver cancer cells; E: Transwell experiment of HCCLM3 and PLC/PRF/5. bP < 0.01, cP < 0.001. PFKFB3: 6-phosphofructo-2-kinase/fructose-2,6-biphosphatase 3; OE: Overexpression; HK2: Hexokinase 2; CPI: Cyclopropyl isomerase; PFKL: Phosphofructokinase liver type; ALDOA: Aldolase A; GAPDH: Glyceraldehyde-3-phosphate dehydrogenase; PGK1: Phosphoglycerate kinase 1; PGAM: Phosphoglycerate mutase; ENO: Enolase; PKM2: Pyruvate kinase M2; LDHA: Lactate dehydrogenase A.
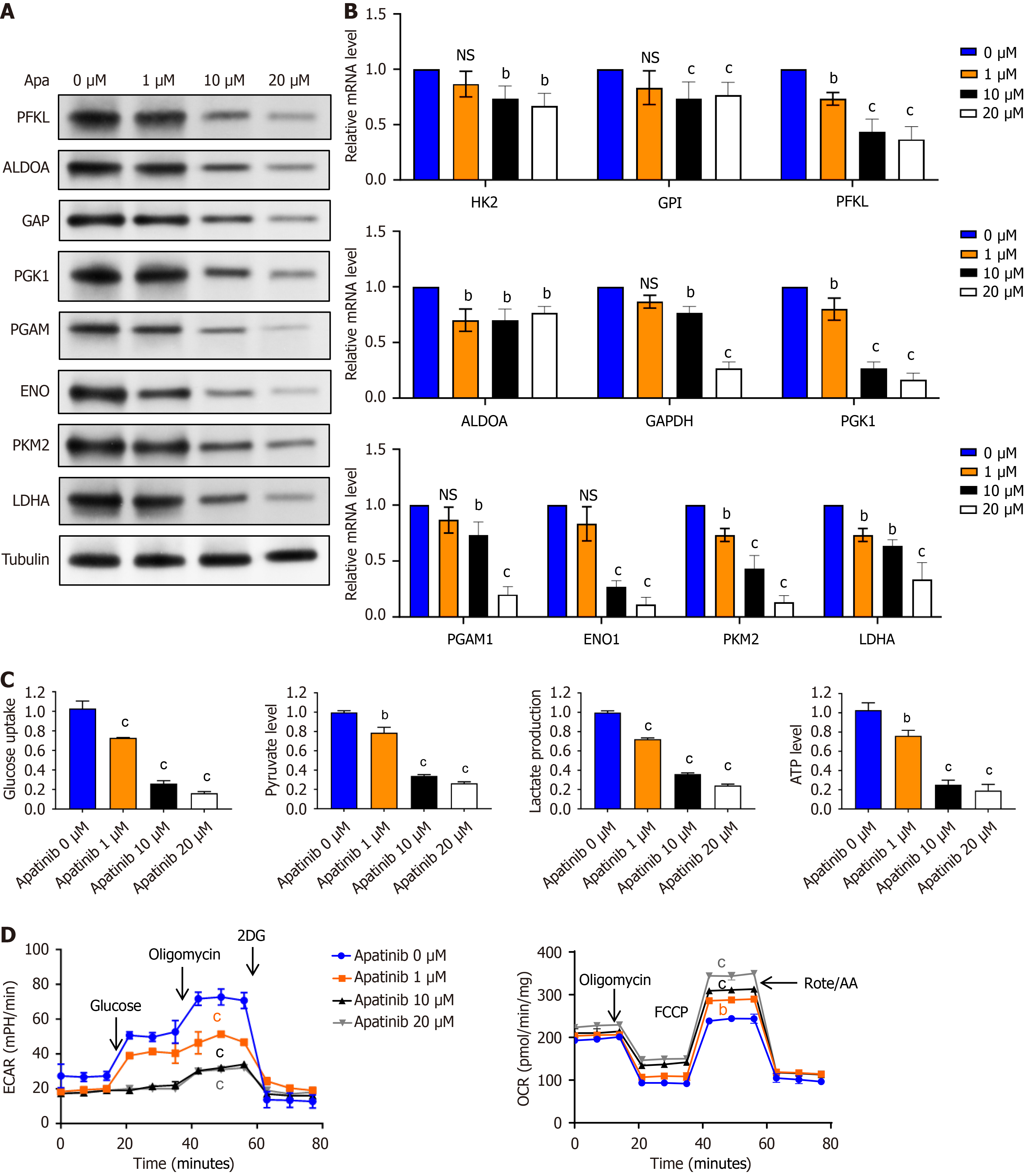
Figure 3 Apatinib suppresses glycolytic activity in vascular endothelial cells.
A: Investigation of the effects of varying concentrations of apatinib on the expression levels of key glycolytic enzymes in vascular endothelial cells (VECs) using Western blotting; B: Evaluation of the impact of different concentrations of apatinib on the expression levels of key glycolytic enzymes in VECs through quantitative real-time polymerase chain reaction; C: Assessment of the impact of different concentrations of apatinib on glucose uptake, pyruvate generation, lactate production, and adenosine triphosphate generation during glycolysis in VECs; D: Examination of the impact of varying apatinib concentrations on extracellular acidification rate and oxygen consumption rate in VECs. bP < 0.01, cP < 0.001. Apa: Apatinib; PFKL: Phosphofructokinase liver type; ALDOA: Aldolase A; GAPDH: Glyceraldehyde-3-phosphate dehydrogenase; PGK1: Phosphoglycerate kinase 1; PGAM: Phosphoglycerate mutase; ENO: Enolase; PKM2: Pyruvate kinase M2; LDHA: Lactate dehydrogenase A; NS: Not significant; ATP: Adenosine triphosphate; ECAR: Extracellular acidification rate; OCR: Oxygen consumption rate.
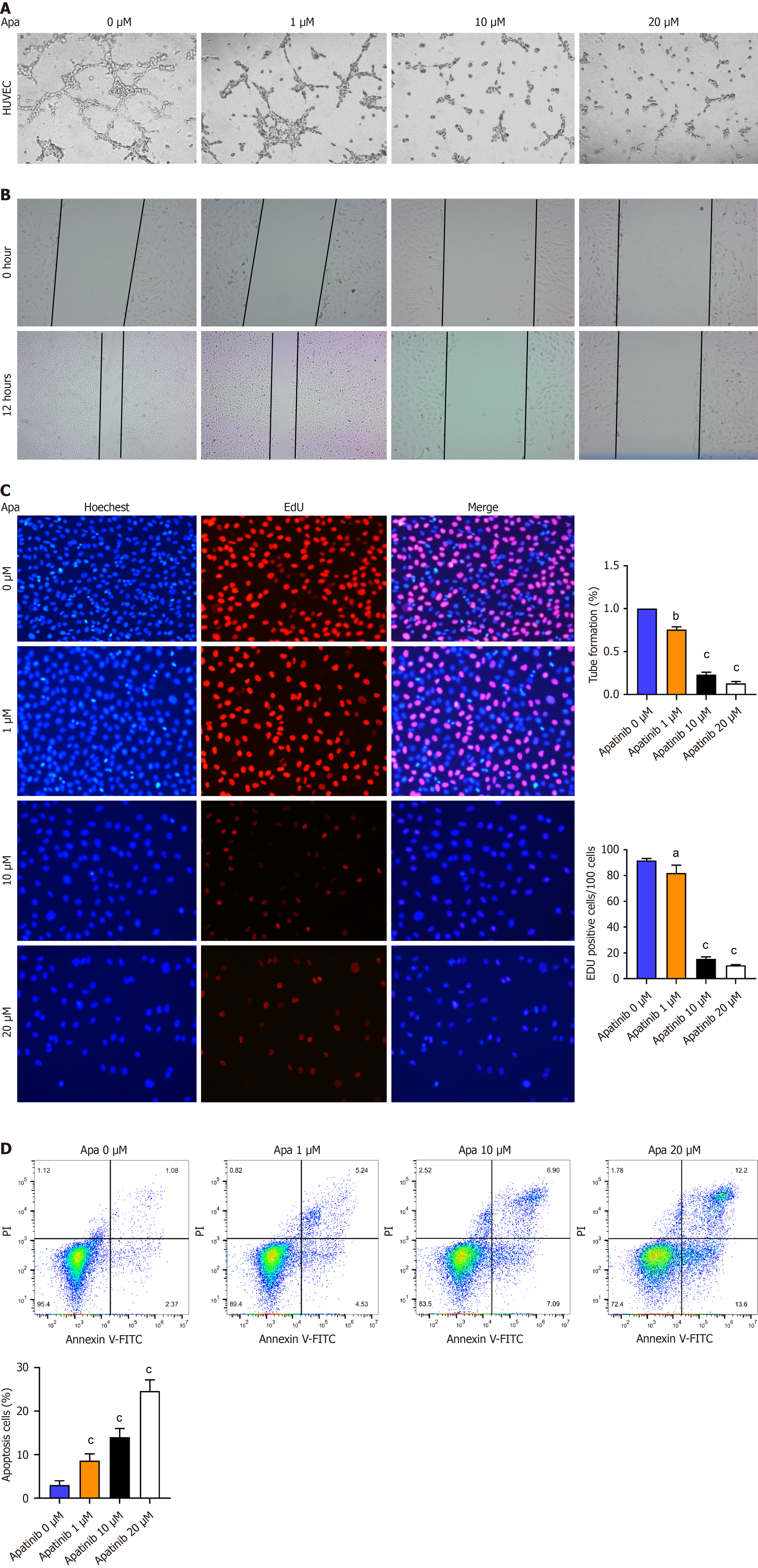
Figure 4 Apatinib inhibits tube formation, proliferation, and migration but induces apoptosis of vascular endothelial cells.
A: Influence of diverse apatinib levels on the capability of vascular endothelial cells (VECs) to form tubes; B: How varying concentrations of apatinib influence the migration patterns of VECs; C: The role of different apatinib doses in altering the proliferation capacity of VECs; D: Annexin V/ propidium iodide staining indicates that apatinib treatment triggers apoptosis in human umbilical vein endothelial cells. aP< 0.05, bP < 0.01, cP < 0.001. Apa: Apatinib; HUVEC: Human umbilical vein endothelial cells; PI: propidium iodide; FITC: Fluorescein isothiocyanate.
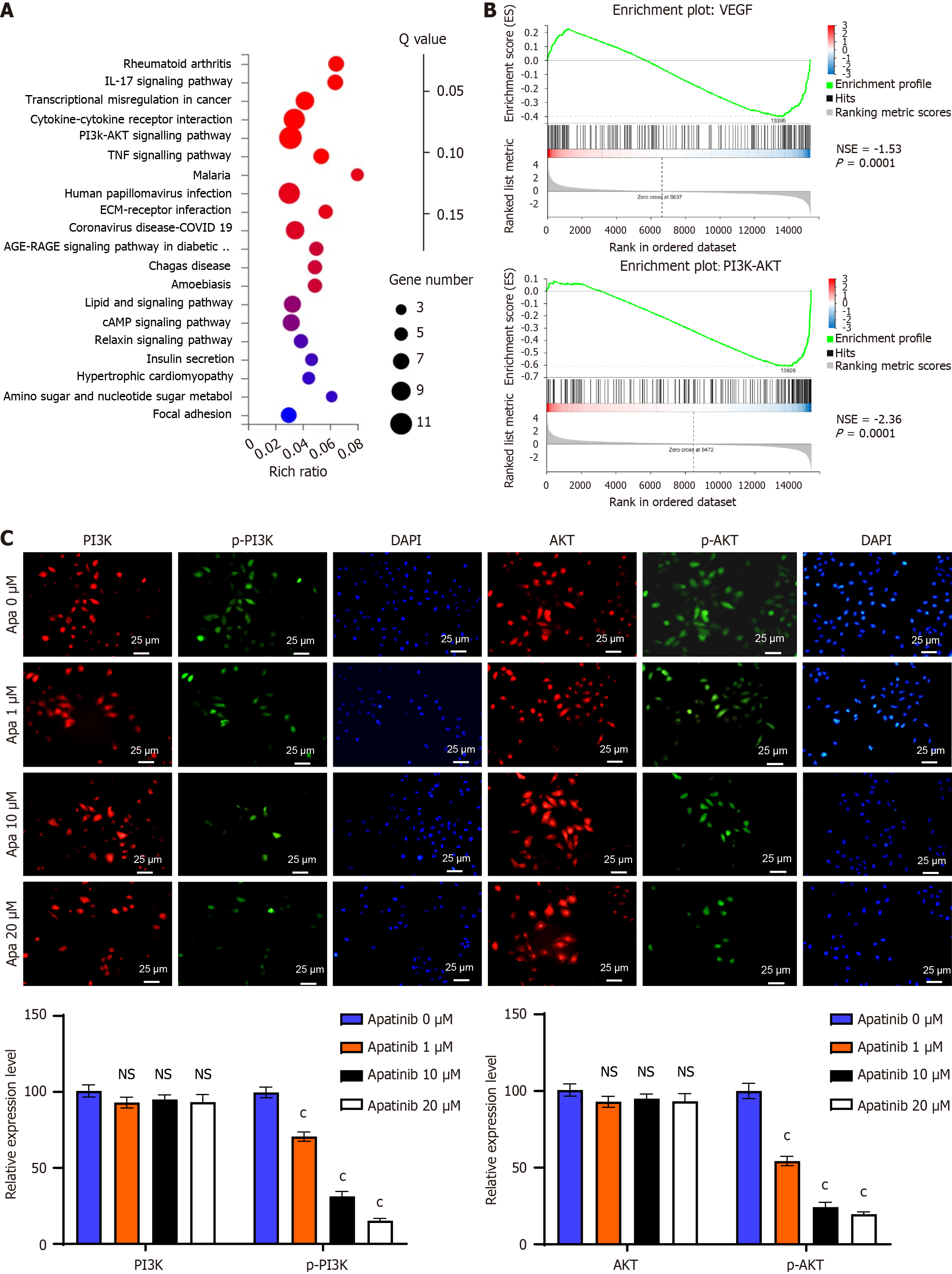
Figure 5 Apatinib suppresses the phosphatidylinositol 3-kinase/AKT signaling pathway.
A: Kyoto Encyclopedia of genes and genomes pathway analysis showed notable alterations in the phosphatidylinositol 3-kinase (PI3K)/AKT pathway following apatinib administration; B: Results from gene set enrichment analysis indicated a significant inhibition of the vascular endothelial growth factor and PI3K/AKT pathways by apatinib; C: Immunofluorescence studies showed a marked reduction in PI3K and AKT protein expression after apatinib administration, as well as a diminished translocation of these proteins from the cytoplasm to the nucleus. cP < 0.001. IL: Interleukin; PI3K: Phosphatidylinositol 3-kinase; TNF: Tumor necrosis factor; ECM: Extracellular matrix; VEGF: Vascular endothelial growth factor; NSE: Normalized enrichment score; Apa: Apatinib; NS: Not significant.
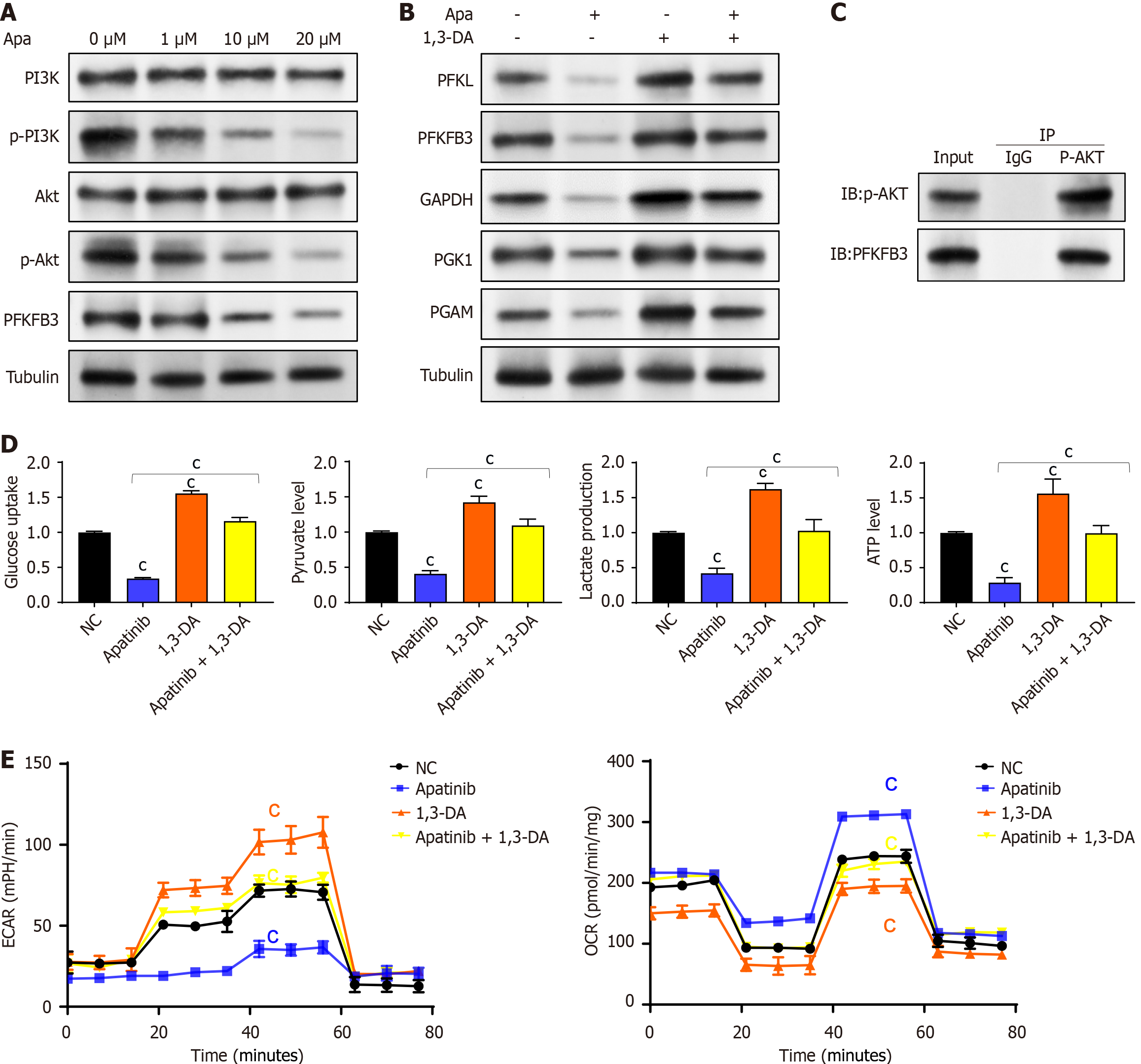
Figure 6 Apatinib inhibits glycolysis in vascular endothelial cells through regulation of the phosphatidylinositol 3-kinase/AKT/PFKFB3 pathway.
A: Apatinib decreases the expression of 6-phosphofructo-2-kinase/fructose-2,6-biphosphatase 3 and the phosphorylation of phosphatidylinositol 3-kinase/AKT, without affecting the total protein levels; B: The inhibition of the glycolytic pathway in vascular endothelial cells (VECs) by apatinib can be counteracted by the phosphatidylinositol 3-kinase activator 1,3-dicaffeoylquinic acid; C: Coimmunoprecipitation results showed that zero acidified AKT could interact with 6-phosphofructo-2-kinase/fructose-2,6-biphosphatase 3; D: The effects of apatinib on glucose uptake, pyruvate generation, lactate production, and adenosine triphosphate generation during glycolysis in VECs can be reversed by 1,3-dicaffeoylquinic acid; E: The effects of apatinib on extracellular acidification rate and oxygen consumption rate in VECs can be reversed by 1,3-dicaffeoylquinic acid. cP < 0.001. Apa: Apatinib; PI3K: Phosphatidylinositol 3-kinase; PFKFB3: 6-phosphofructo-2-kinase/fructose-2,6-biphosphatase 3; DA: Dicaffeoylquinic acid; PFKL: Phosphofructokinase liver type; GAPDH: Glyceraldehyde-3-phosphate dehydrogenase; PGK1: Phosphoglycerate kinase 1; PGAM: Phosphoglycerate mutase; IP: Immunoprecipitation; IgG: Immunoglobulin G.
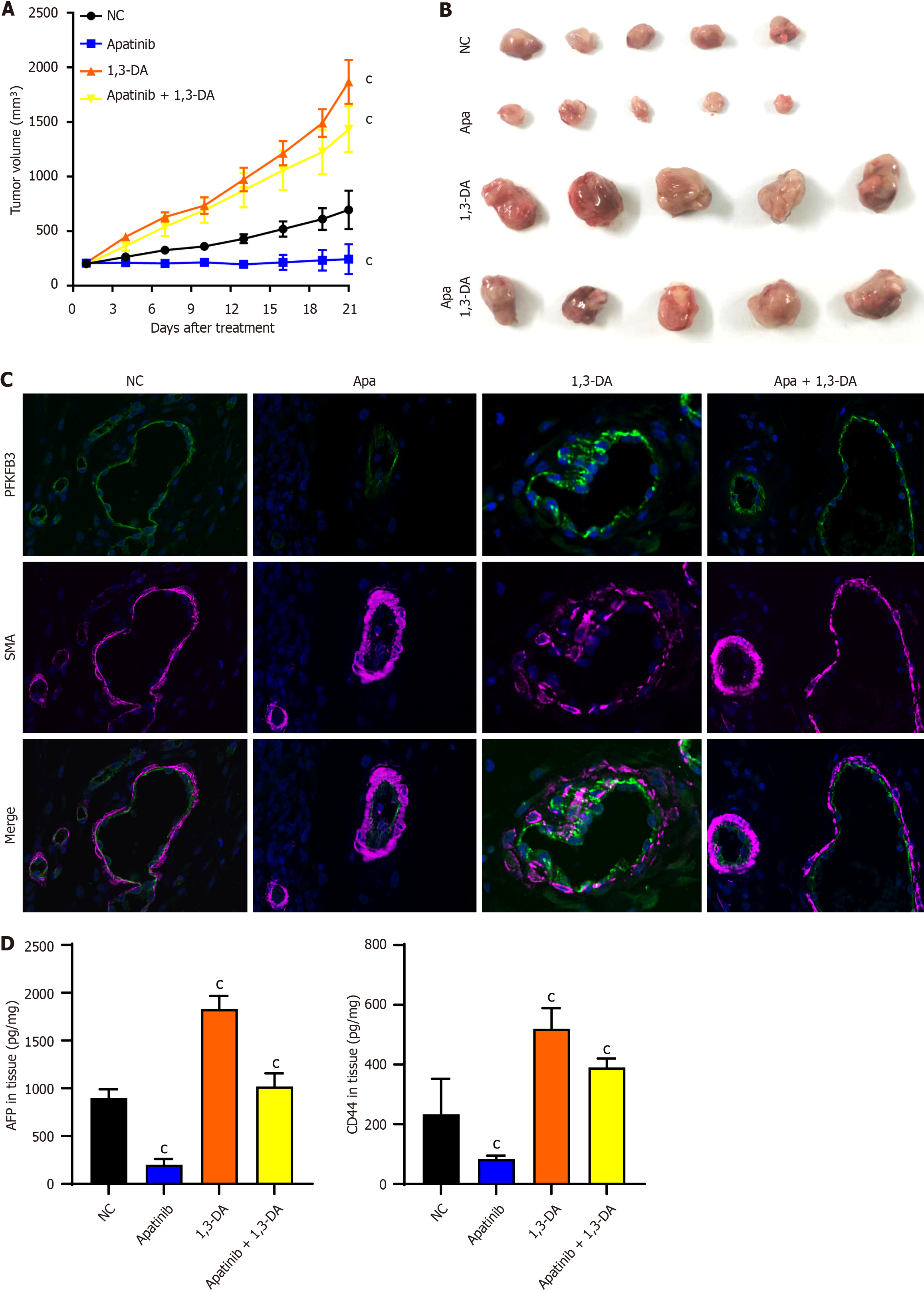
Figure 7 Apatinib inhibits liver cancer angiogenesis and suppresses glycolysis in vascular endothelial cells in vivo.
A: Tumor growth curve of mice treated with apatinib and 1,3-dicaffeoylquinic acid; B: Tumor size after 21 days of treatment with apatinib and 1,3-dicaffeoylquinic acid in mice; C: Immunofluorescence staining of mouse tissues treated with apatinib and 1,3-dicaffeoylquinic acid; D: The contents of alpha-fetoprotein and CD44 in animal tissues were detected by enzyme linked immunosorbent assay. cP < 0.001. Apa: Apatinib; DA: Dicaffeoylquinic acid; SMA: Smooth muscle actin; PFKFB3: 6-phosphofructo-2-kinase/fructose-2,6-biphosphatase 3; AFP: Alpha-fetoprotein.
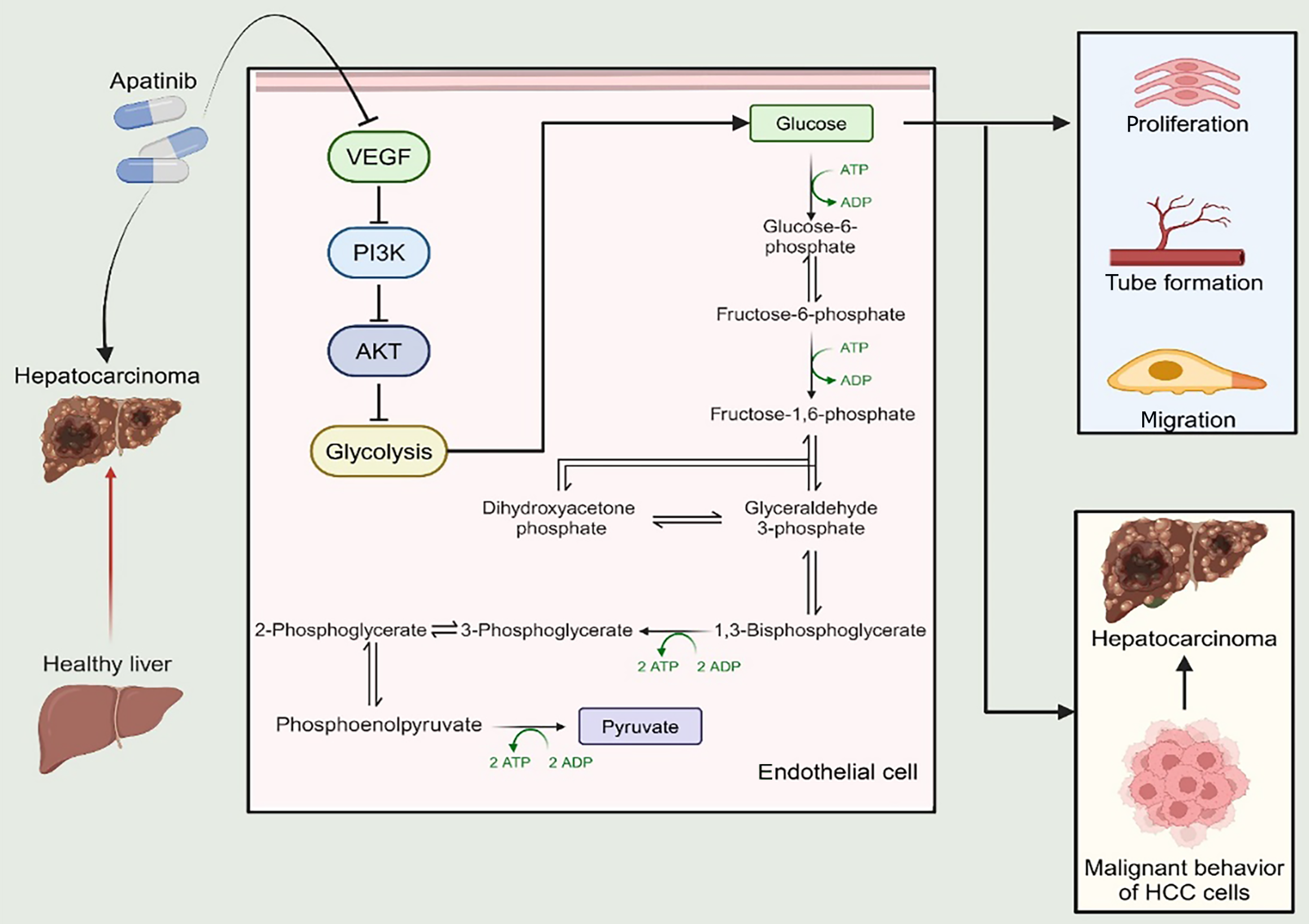
Figure 8 A schematic illustrating the impact of apatinib on glycolysis in vascular endothelial cells.
VEGF: Vascular endothelial growth factor; PI3K: Phosphatidylinositol 3-kinase; ATP: Adenosine triphosphate; ADP: Adenosine diphosphate; HCC: Hepatocellular carcinoma.
- Citation: Wu Y, Xie BB, Zhang BL, Zhuang QX, Liu SW, Pan HM. Apatinib regulates the glycolysis of vascular endothelial cells through PI3K/AKT/PFKFB3 pathway in hepatocellular carcinoma. World J Gastroenterol 2025; 31(11): 102848
- URL: https://www.wjgnet.com/1007-9327/full/v31/i11/102848.htm
- DOI: https://dx.doi.org/10.3748/wjg.v31.i11.102848