Copyright
©The Author(s) 2024.
World J Gastrointest Pharmacol Ther. May 28, 2024; 15(3): 90757
Published online May 28, 2024. doi: 10.4292/wjgpt.v15.i3.90757
Published online May 28, 2024. doi: 10.4292/wjgpt.v15.i3.90757
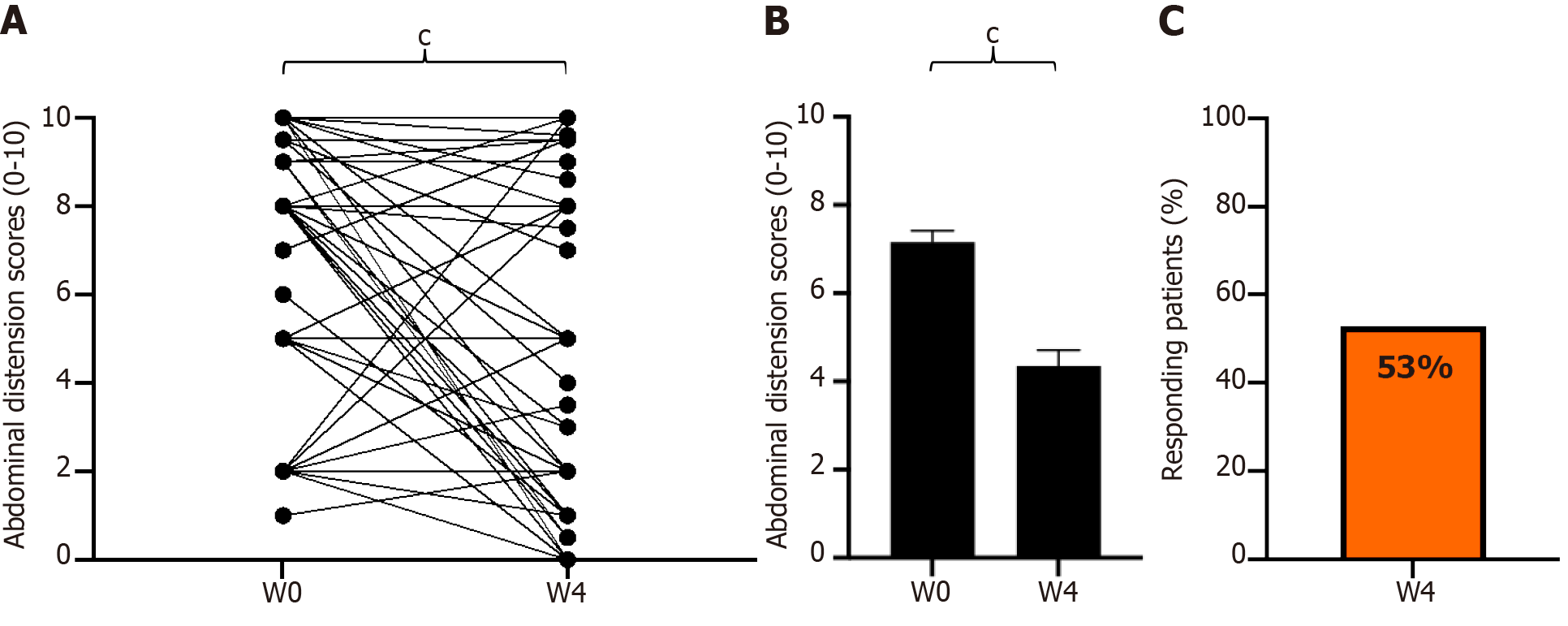
Figure 5 Change in abdominal distension scores from baseline to week 4 of GASTRAP® DIRECT treatment (visit 1 to visit 3).
A: Change in paired abdominal distension scores; B: Abdominal distension scores (0–10); C: Patient responders (delta > 30%) with respect to abdominal distension. cP < 0.001. W: Weeks.
- Citation: Talbodec N, Le Roy P, Fournier P, Lesage B, Lepoutre E, Castex F, Godchaux JM, Vandeville L, Bismuth B, Lesage X, Bayart P, Genin M, Rousseaux C, Maquet V, Modica S, Desreumaux P, Valibouze C. Efficacy and tolerability of chitin-glucan combined with simethicone (GASTRAP® DIRECT) in irritable bowel syndrome: A prospective, open-label, multicenter study. World J Gastrointest Pharmacol Ther 2024; 15(3): 90757
- URL: https://www.wjgnet.com/2150-5349/full/v15/i3/90757.htm
- DOI: https://dx.doi.org/10.4292/wjgpt.v15.i3.90757