Copyright
©2010 Baishideng Publishing Group Co.
World J Biol Chem. Aug 26, 2010; 1(8): 239-247
Published online Aug 26, 2010. doi: 10.4331/wjbc.v1.i8.239
Published online Aug 26, 2010. doi: 10.4331/wjbc.v1.i8.239
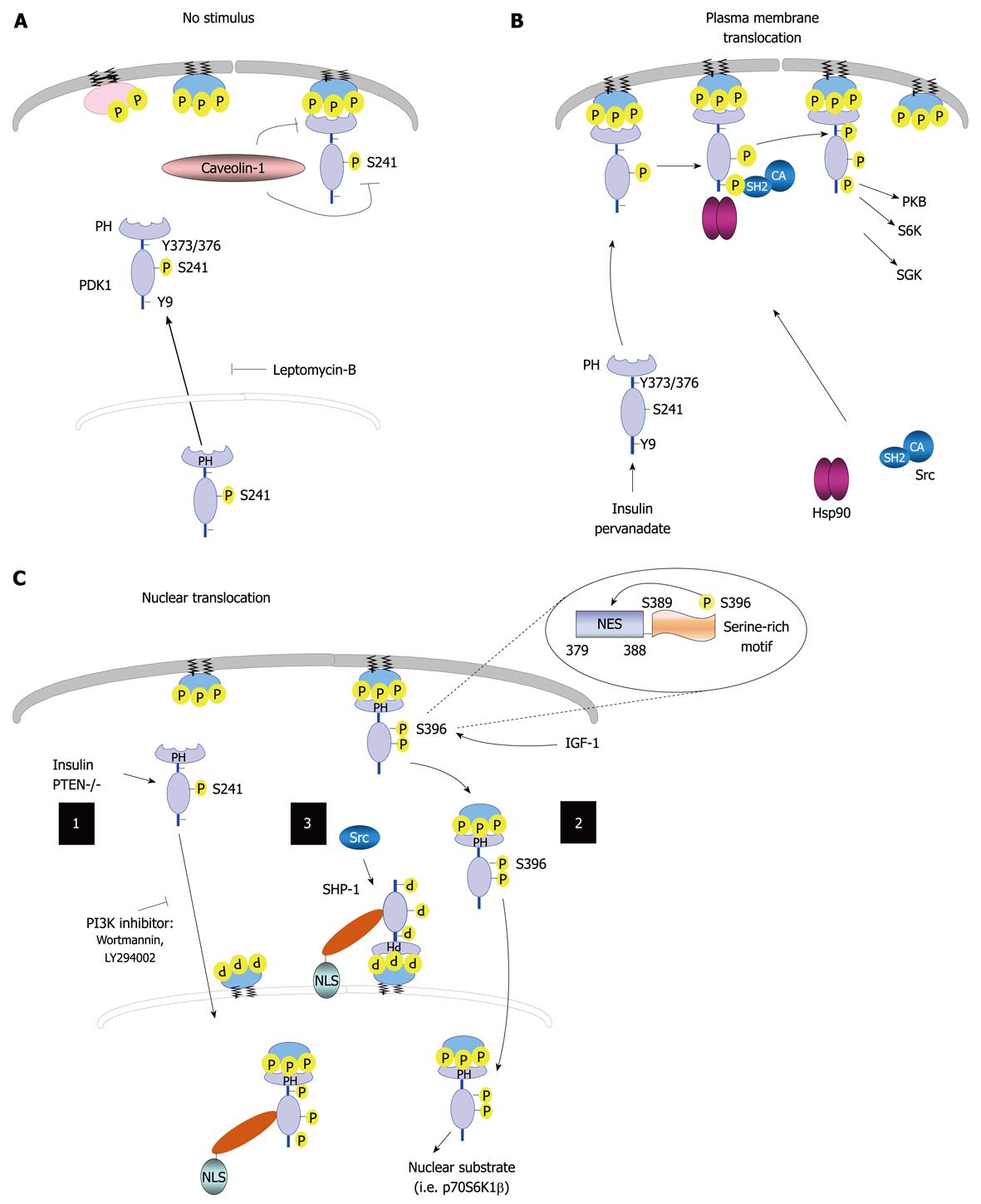
Figure 3 Regulation of 3-phosphoinositide-dependent protein kinase-1 cellular localization.
A: 3-phosphoinositide-dependent protein kinase-1 (PDK1) resides in the cytoplasm and membranes in unstimulated cells, and can shuttle between these two compartments. The treatment of cells with leptomycin-B results in PDK1 accumulation in the nucleus. The localization of this kinase to the plasma membrane is disrupted by caveolin-1; B: PDK1 can translocate to the plasma membrane following treatment with pervanadate or insulin, which leads to PDK1 phosphorylation on Tyr-9 and Tyr-373/376; C: Nucleus-cytoplasm shuttle. (1) Phosphoinositide 3-kinase (PI3K) signaling regulates PDK1 nuclear translocation; (2) Insulin growth factor-1 (IGF-1) induces Ser-396 phosphorylation, thereby placing the serine-rich motif (amino acids Ser-389-396) close to its nuclear export sequence (NES) region, which leads to PDK1 nuclear translocation; and (3) Tyrosine phosphatase SH2 domain-1 (SHP-1) binds to PDK1 following tyrosine phosphorylation. The SHP-1/PDK1 complex is recruited to the nuclear membrane by binding to perinuclear phosphatidylinositol 3,4,5 trisphosphate, where SHP-1 and its nuclear localization signal (NLS) facilitates active import. In addition, Src promotes the association of SHP-1 to PDK1. Nuclear PDK1 regulates its nuclear substrates and activates corresponding signaling pathways. PTEN: Phosphatase and tensin homolog; CA: Constitutively active; PH: Pleckstrin homology; PKB: Protein kinase B; SGK: Serum and glucocorticoid-inducible kinase; Hsp90: Heat shock protein 90.
- Citation: Li Y, Yang KJ, Park J. Multiple implications of 3-phosphoinositide-dependent protein kinase 1 in human cancer. World J Biol Chem 2010; 1(8): 239-247
- URL: https://www.wjgnet.com/1949-8454/full/v1/i8/239.htm
- DOI: https://dx.doi.org/10.4331/wjbc.v1.i8.239