Copyright
©The Author(s) 2023.
World J Gastroenterol. Mar 14, 2023; 29(10): 1614-1626
Published online Mar 14, 2023. doi: 10.3748/wjg.v29.i10.1614
Published online Mar 14, 2023. doi: 10.3748/wjg.v29.i10.1614
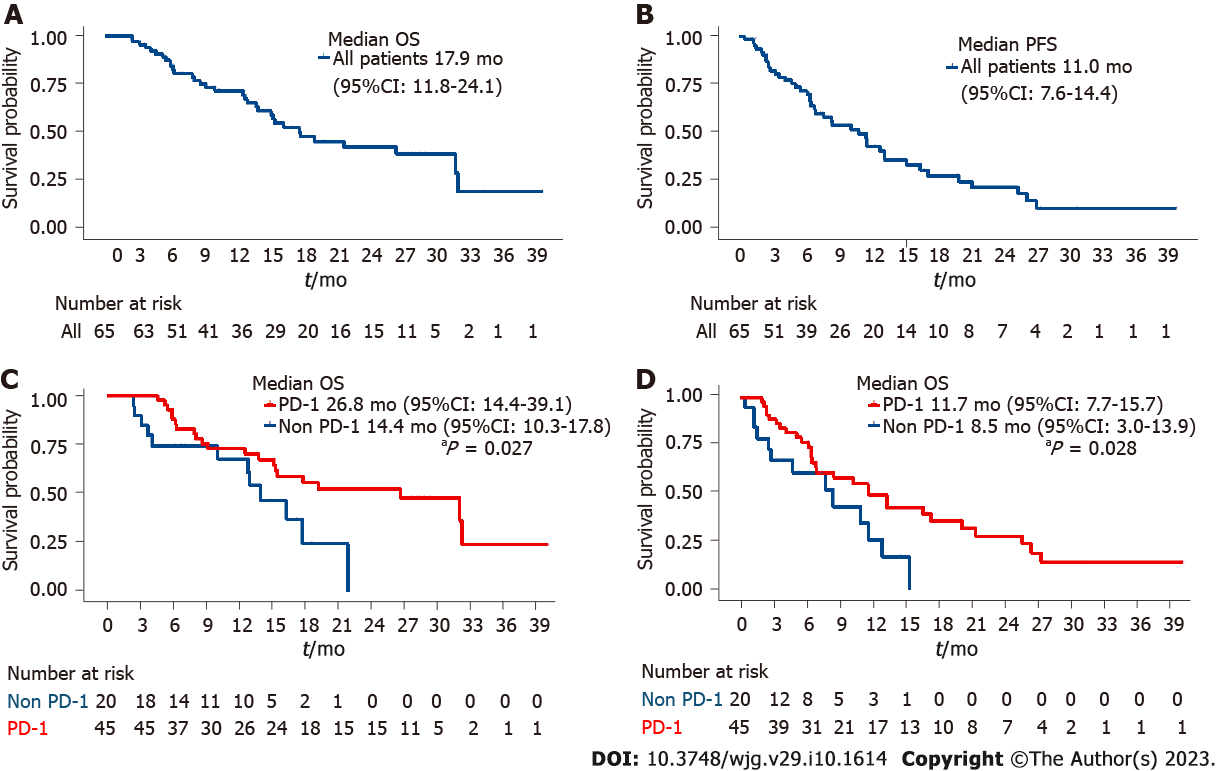
Figure 2 The overall survival and progression-free survival times of all included patients and programmed death receptor-1-lenvatinib-transarterial chemoembolization group and lenvatinib-transarterial chemoembolization group.
A: The overall survival (OS) times of all included patients; B: The progression-free survival (PFS) times of all included patients; C: The OS times of programmed death receptor-1 (PD-1)-lenvatinib-transarterial chemoembolization (TACE) group and lenvatinib-TACE group; D: The PFS times of PD-1-lenvatinib-TACE group and lenvatinib-TACE group. OS: Overall survival; PFS: Progression-free survival; CI: Confidence interval; PD-1: Programmed death receptor-1.
- Citation: Wang YY, Yang X, Wang YC, Long JY, Sun HS, Li YR, Xun ZY, Zhang N, Xue JN, Ning C, Zhang JW, Zhu CP, Zhang LH, Yang XB, Zhao HT. Clinical outcomes of lenvatinib plus transarterial chemoembolization with or without programmed death receptor-1 inhibitors in unresectable hepatocellular carcinoma. World J Gastroenterol 2023; 29(10): 1614-1626
- URL: https://www.wjgnet.com/1007-9327/full/v29/i10/1614.htm
- DOI: https://dx.doi.org/10.3748/wjg.v29.i10.1614