Copyright
©2014 Baishideng Publishing Group Inc.
World J Gastroenterol. May 21, 2014; 20(19): 5708-5720
Published online May 21, 2014. doi: 10.3748/wjg.v20.i19.5708
Published online May 21, 2014. doi: 10.3748/wjg.v20.i19.5708
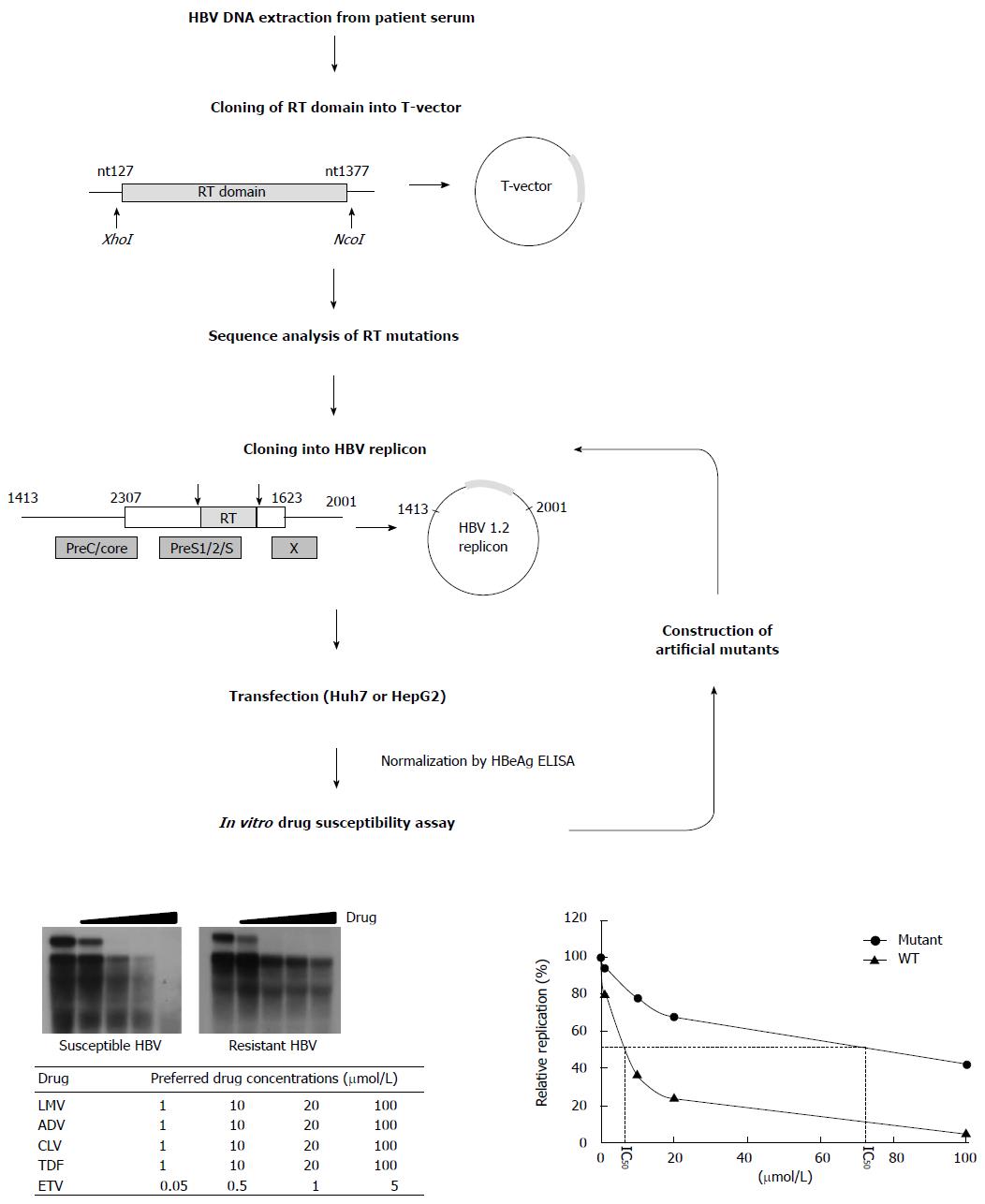
Figure 3 Scheme for in vitro phenotypic validation of drug-resistant hepatitis B virus.
Hepatitis B virus (HBV) DNA is purified from patient serum, and the sequence of RT mutations is analyzed. After cloning into replication-competent HBV replicons, each mutant is transfected into hepatoma cell lines followed by Southern blot (or real time polymerase chain reaction) analysis. The IC50 (μmol/L) value is obtained by quantification of replication ability and curve-fitting. To characterize the specific mutation(s) conferring resistance to antiviral drugs, each artificial mutant must be constructed and individually tested.
- Citation: Kim JH, Park YK, Park ES, Kim KH. Molecular diagnosis and treatment of drug-resistant hepatitis B virus. World J Gastroenterol 2014; 20(19): 5708-5720
- URL: https://www.wjgnet.com/1007-9327/full/v20/i19/5708.htm
- DOI: https://dx.doi.org/10.3748/wjg.v20.i19.5708